Sodium chloride
 Sodium chloride crystals in a form of halite
| |
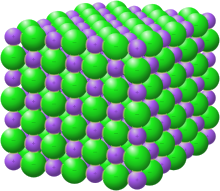 Crystal structure with sodium in purple and chloride in green[1]
| |
| Names | |
|---|---|
| IUPAC name
Sodium chloride
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 3534976 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.028.726 |
| EC Number |
|
| 13673 | |
| KEGG | |
| MeSH | Sodium+chloride |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NaCl | |
| Molar mass | 58.443 g/mol[2] |
| Appearance | Colorless cubic crystals[2] |
| Odor | Odorless |
| Density | 2.17 g/cm3[2] |
| Melting point | 800.7 °C (1,473.3 °F; 1,073.8 K)[2] |
| Boiling point | 1,465 °C (2,669 °F; 1,738 K)[2] |
| 360 g/L (25°C)[2] | |
| Solubility in ammonia | 21.5 g/L |
| Solubility in methanol | 14.9 g/L |
| −30.2·10−6 cm3/mol[3] | |
Refractive index (nD)
|
1.5441 (at 589 nm)[4] |
| Structure[5] | |
| Face-centered cubic (see text), cF8 | |
| Fm3m (No. 225) | |
a = 564.02 pm
| |
Formula units (Z)
|
4 |
| octahedral at Na+ octahedral at Cl− | |
| Thermochemistry[6] | |
Heat capacity (C)
|
50.5 J/(K·mol) |
Std molar
entropy (S⦵298) |
72.10 J/(K·mol) |
Std enthalpy of (ΔfH⦵298)formation |
−411.120 kJ/mol |
| Pharmacology | |
| A12CA01 (WHO) B05CB01 (WHO), B05XA03 (WHO), S01XA03 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3 g/kg (oral, rats)[7] |
| Related compounds | |
Other anions
|
Sodium fluoride Sodium bromide Sodium iodide Sodium astatide |
Other cations
|
Lithium chloride Potassium chloride Rubidium chloride Caesium chloride Francium chloride |
| Supplementary data page | |
| Sodium chloride (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium chloride
Uses
In addition to the familiar domestic uses of salt, more dominant applications of the approximately 250 million tonnes per year production (2008 data) include chemicals and de-icing.[9]
Chemical functions
Salt is used, directly or indirectly, in the production of many chemicals, which consume most of the world's production.[10]
Chlor-alkali industry
It is the starting point for the chloralkali process, the industrial process to produce chlorine and sodium hydroxide, according to the chemical equation
This electrolysis is conducted in either a mercury cell, a diaphragm cell, or a membrane cell. Each of those uses a different method to separate the chlorine from the sodium hydroxide. Other technologies are under development due to the high energy consumption of the electrolysis, whereby small improvements in the efficiency can have large economic paybacks. Some applications of chlorine include
Sodium hydroxide is extensively used in many different industries enabling production of paper, soap, and aluminium etc.
Soda-ash industry
Sodium chloride is used in the Solvay process to produce sodium carbonate and calcium chloride. Sodium carbonate, in turn, is used to produce glass, sodium bicarbonate, and dyes, as well as a myriad of other chemicals. In the Mannheim process, sodium chloride is used for the production of sodium sulfate and hydrochloric acid.
Miscellaneous industrial uses
Sodium chloride is heavily used, so even relatively minor applications can consume massive quantities. In
In textiles and dyeing, salt is used as a brine rinse to separate organic contaminants, to promote "salting out" of dyestuff precipitates, and to blend with concentrated dyes to increase yield in dyebaths and make the colors look sharper. One of its main roles is to provide the positive ion charge to promote the absorption of negatively charged ions of dyes.[10]
For use in the pulp and paper industry, it is used to manufacture sodium chlorate, which is then reacted with sulfuric acid and a reducing agent such as methanol to manufacture chlorine dioxide, a bleaching chemical that is widely used to bleach wood pulp.
In tanning and leather treatment, salt is added to animal hides to inhibit microbial activity on the underside of the hides and to attract moisture back into the hides.[10]
In rubber manufacture, salt is used to make buna, neoprene, and white rubber types. Salt brine and sulfuric acid are used to coagulate an emulsified latex made from chlorinated butadiene.[10][9]
Salt also is added to secure the soil and to provide firmness to the foundation on which highways are built. The salt acts to minimize the effects of shifting caused in the subsurface by changes in humidity and traffic load.[10]
Water softening
Hard water contains calcium and magnesium ions that interfere with action of soap and contribute to the buildup of a scale or film of alkaline mineral deposits in household and industrial equipment and pipes. Commercial and residential water-softening units use ion-exchange resins to remove ions that cause the hardness. These resins are generated and regenerated using sodium chloride.[10][9]
Road salt
The second major application of salt is for deicing and anti-icing of roads, both in grit bins and spread by winter service vehicles. In anticipation of snowfall, roads are optimally "anti-iced" with brine (concentrated solution of salt in water), which prevents bonding between the snow-ice and the road surface. This procedure obviates the heavy use of salt after the snowfall. For de-icing, mixtures of brine and salt are used, sometimes with additional agents such as calcium chloride and/or magnesium chloride. The use of salt or brine becomes ineffective below −10 °C (14 °F).

Salt for de-icing in the United Kingdom predominantly comes from a single mine in Winsford in Cheshire. Prior to distribution it is mixed with <100 ppm of sodium ferrocyanide as an anticaking agent, which enables rock salt to flow freely out of the gritting vehicles despite being stockpiled prior to use. In recent years this additive has also been used in table salt. Other additives had been used in road salt to reduce the total costs. For example, in the US, a byproduct carbohydrate solution from sugar-beet processing was mixed with rock salt and adhered to road surfaces about 40% better than loose rock salt alone. Because it stayed on the road longer, the treatment did not have to be repeated several times, saving time and money.[10]
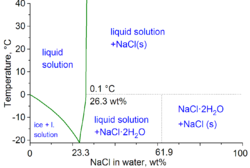
In the technical terms of physical chemistry, the minimum freezing point of a water-salt mixture is −21.12 °C (−6.02 °F) for 23.31 wt% of salt. Freezing near this concentration is however so slow that the
Environmental effects
Road salt ends up in fresh-water bodies and could harm aquatic plants and animals by disrupting their
In highway de-icing, salt has been associated with
Substitution
Some agencies are substituting
Food industry and agriculture
Salt is added to food, either by the food producer or by the consumer, as a flavor enhancer, preservative, binder,
It is used as a cheap and safe
In many dairy industries, salt is added to cheese as a color-, fermentation-, and texture-control agent. The dairy subsector includes companies that manufacture creamery butter, condensed and evaporated milk, frozen desserts, ice cream, natural and processed cheese, and specialty dairy products. In canning, salt is primarily added as a flavor enhancer and preservative. It also is used as a carrier for other ingredients, dehydrating agent, enzyme inhibitor and tenderizer. In baking, salt is added to control the rate of fermentation in bread dough. It also is used to strengthen the gluten (the elastic protein-water complex in certain doughs) and as a flavor enhancer, such as a topping on baked goods. The food-processing category also contains grain mill products. These products consist of milling flour and rice and manufacturing cereal breakfast food and blended or prepared flour. Salt is also used a seasoning agent, e.g. in potato chips, pretzels, and cat and dog food.[10]
Sodium chloride is used in veterinary medicine as
Medicine
Sodium chloride is used together with water as one of the primary solutions for intravenous therapy. Nasal spray often contains a saline solution.
Sodium chloride is also available as an oral tablet, and is taken to treat low sodium levels.[17]
Firefighting

Sodium chloride is the principal extinguishing agent in fire extinguishers (Met-L-X, Super D) used on combustible metal fires such as magnesium, potassium, sodium, and NaK alloys (Class D). Thermoplastic powder is added to the mixture, along with waterproofing (metal stearates) and anticaking agents (tricalcium phosphate) to form the extinguishing agent. When it is applied to the fire, the salt acts like a heat sink, dissipating heat from the fire, and also forms an oxygen-excluding crust to smother the fire. The plastic additive melts and helps the crust maintain its integrity until the burning metal cools below its ignition temperature. This type of extinguisher was invented in the late 1940s as a cartridge-operated unit, although stored pressure versions are now popular. Common sizes are 30 pounds (14 kg) portable and 350 pounds (160 kg) wheeled.[citation needed]
Cleanser
Since at least
Optical usage
Defect-free NaCl crystals have an optical transmittance of about 90% for infrared light, specifically between 200
Chemistry

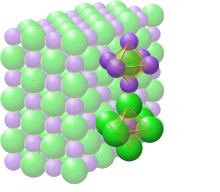
Solid sodium chloride
In solid sodium chloride, each ion is surrounded by six ions of the opposite charge as expected on electrostatic grounds. The surrounding ions are located at the vertices of a regular
Solid sodium chloride has a melting point of 801 °C and liquid sodium chloride boils at 1465 °C. Atomic-resolution real-time video imaging allows visualization of the initial stage of crystal nucleation of sodium chloride.[19]
The
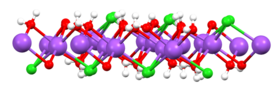
From cold (sub-freezing) solutions, salt crystallises with
In 2023, it was discovered that under pressure, sodium chloride can form the hydrates NaCl·8.5H2O and NaCl·13H2O.[23]
Aqueous solutions
The attraction between the Na+ and Cl− ions in the solid is so strong that only highly
When dissolved in water, the sodium chloride framework disintegrates as the Na+ and Cl− ions become surrounded by polar water molecules. These solutions consist of
pH of sodium chloride solutions
The pH of a sodium chloride solution remains ≈7 due to the extremely weak basicity of the Cl− ion, which is the conjugate base of the strong acid HCl. In other words, NaCl has no effect on system pH[25] in diluted solutions where the effects of ionic strength and activity coefficients are negligible.
| Solubility of NaCl (g NaCl / 1 kg of solvent at 25 °C (77 °F))[26] | |
|---|---|
Water |
360 |
| Formamide | 94 |
Glycerin |
83 |
| Propylene glycol | 71 |
| Formic acid | 52 |
| Liquid ammonia | 30.2 |
| Methanol | 14 |
| Ethanol | 0.65 |
| Dimethylformamide | 0.4 |
Propan-1-ol |
0.124 |
| Sulfolane | 0.05 |
Butan-1-ol |
0.05 |
Propan-2-ol |
0.03 |
Pentan-1-ol |
0.018 |
| Acetonitrile | 0.003 |
| Acetone | 0.00042 |
Stoichiometric and structure variants
Common salt has a 1:1 molar ratio of sodium and chlorine. In 2013, compounds of sodium and chloride of different stoichiometries have been discovered; five new compounds were predicted (e.g., Na3Cl, Na2Cl, Na3Cl2, NaCl3, and NaCl7). The existence of some of them has been experimentally confirmed at high pressures and other conditions: cubic and orthorhombic NaCl3, two-dimensional metallic tetragonal Na3Cl and exotic hexagonal NaCl.[27] This indicates that compounds violating chemical intuition are possible, in simple systems under non-ambient conditions.[28]
Occurrence
Salt is found in the
Production
Salt is currently
-
Modern rock salt mine nearMount Morris, New York, United States
-
Mounds of salt, Salar de Uyuni, Bolivia.
See also
- Biosalinity
- Edible salt (table salt)
- Halite, the mineral form of sodium chloride
- Health effects of salt
- Salinity
- Salting the earth
- Salt poisoning
References
- ^ "Sodium Chloride (NaCl) Crystal". PhysicsOpenLab. Retrieved 23 August 2021.
- ^ a b c d e f Haynes, 4.89
- ^ Haynes, 4.135
- ^ Haynes, 10.241
- ^ Haynes, 4.148
- ^ Haynes, 5.8
- PMID 5110827.
- ISBN 9781405881180
- ^ .
- ^ a b c d e f g h i j k l Kostick, Dennis S. (October 2010). "Salt" (PDF). U.S. Geological Survey, 2008 Minerals Yearbook.
- ^ ISBN 978-3-527-20124-2.
- ISSN 1091-2339. Retrieved 11 March 2023.
- ^ "Saltier waterways are creating dangerous 'chemical cocktails'". phys.org.
- ^ "Most Road Salt Is Making It into Lakes And Rivers". www.sciencedaily.com. University of Minnesota. 20 February 2009. Retrieved 27 September 2015.
- ^ Casey, Michael. "Turning to beet juice and beer to address road salt danger". phys.org.
- ^ "EASA Cautions on Organic Salt Deicing Fluid". MRO Network. 9 December 2016.
- ^ "Sodium Chloride for oral solution". Cleveland Clinic. Retrieved 9 March 2024.
- ^ .
- PMID 33475359.
- ISBN 978-3-540-42180-1.
- .
- ^ Water-NaCl phase diagram. Lide, CRC Handbook of Chemistry and Physics, 86 ed (2005-2006), CRC pages 8-71, 8-116
- ^ University of Washington. "Newly discovered form of salty ice could exist on surface of extraterrestrial moons". Phys.org.
- .
- ^ "Acidic, Basic, and Neutral Salts". Flinn Scientific Chem Fax. 2016. Retrieved 18 September 2018.
Neutralization of a strong acid and a strong base gives a neutral salt.
- ISBN 978-0-85312-027-8.
- S2CID 216130640.
- S2CID 15298372.
- S2CID 121846285.
- ^ Bolen, Wallace P. (January 2018). "Salt" (PDF). U.S. Geological Survey, 2018 Mineral Commodity Summaries.
 This article incorporates public domain material from Salt (PDF). United States Geological Survey.
This article incorporates public domain material from Salt (PDF). United States Geological Survey.
Cited sources
- Haynes, William M., ed. (2011). ISBN 978-1439855119.
External links
- Salt United States Geological Survey Statistics and Information
- "Using Salt and Sand for Winter Road Maintenance". Road Management Journal. December 1997. Archived from the original on 21 September 2016. Retrieved 13 February 2007.
- Calculators: surface tensions, and densities, molarities and molalities of aqueous NaCl (and other salts)
- JtBaker MSDS


![{\displaystyle {\ce {2 NaCl + 2 H2O ->[electrolysis] Cl2 + H2 + 2 NaOH}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/6175336b0ecf3b2032d6a60ac2a534b10e6f61f3)


