Sodium dithionite
| |

| |

| |
| Names | |
|---|---|
| Other names
D-Ox, Hydrolin, Reductone
sodium hydrosulfite, sodium sulfoxylate, Sulfoxylate Vatrolite, Virtex L Hydrosulfit, Prayon Blankit, Albite A, Konite Zepar, Burmol, Arostit | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.028.991 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1384 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Na2S2O4 | |
| Molar mass | 174.107 g/mol (anhydrous) 210.146 g/mol (dihydrate) |
| Appearance | white to grayish crystalline powder light-lemon colored flakes |
| Odor | faint sulfur odor |
| Density | 2.38 g/cm3 (anhydrous) 1.58 g/cm3 (dihydrate) |
| Melting point | 52 °C (126 °F; 325 K) |
| Boiling point | Decomposes |
| 18.2 g/100 mL (anhydrous, 20 °C) 21.9 g/100 mL (Dihydrate, 20 °C) | |
| Solubility | slightly soluble in alcohol |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H251, H302 | |
| P235+P410, P264, P270, P280, P301+P312, P330, P407, P413, P420, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 100 °C (212 °F; 373 K) |
| 200 °C (392 °F; 473 K) | |
| Related compounds | |
Other anions
|
Sodium sulfite Sodium sulfate |
Related compounds
|
Sodium thiosulfate Sodium bisulfite Sodium metabisulfite Sodium bisulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium dithionite (also known as sodium hydrosulfite) is a white crystalline powder with a sulfurous odor. Although it is stable in dry air, it decomposes in hot water and in acid solutions.
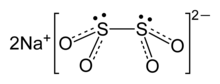
Structure
The structure has been examined by
A weak S-S bond is indicated by the S-S distance of 239 pm, which is elongated by ca. 30 pm relative to a typical S-S bond.
Preparation
Sodium dithionite is produced industrially by reduction of sulfur dioxide. Approximately 300,000 tons were produced in 1990.[4] The route using zinc powder is a two-step process:
- 2 SO2 + Zn → ZnS2O4
- ZnS2O4 + 2 NaOH → Na2S2O4 + Zn(OH)2
The sodium borohydride method obeys the following stoichiometry:
- NaBH4 + 8 NaOH + 8 SO2 → 4 Na2S2O4 + NaBO2 + 6 H2O
Each equivalent of H− reduces two equivalents of sulfur dioxide. Formate has also been used as the reductant.
Properties and reactions
Hydrolysis
Sodium dithionite is stable when dry, but aqueous solutions deteriorate due to the following reaction:
- 2 S2O42− + H2O → S2O32− + 2 HSO3−
This behavior is consistent with the instability of dithionous acid. Thus, solutions of sodium dithionite cannot be stored for a long period of time.[3]
Anhydrous sodium dithionite decomposes to sodium sulfate and sulfur dioxide above 90 °C in the air. In absence of air, it decomposes quickly above 150 °C to sodium sulfite, sodium thiosulfate, sulfur dioxide and trace amount of sulfur.
Redox reactions
Sodium dithionite is a reducing agent. At pH 7, the potential is -0.66 V compared to the normal hydrogen electrode. Redox occurs with formation of bisulfite:[5]
- S2O42- + 2 H2O → 2 HSO3− + 2 e− + 2 H+
Sodium dithionite reacts with oxygen:
- Na2S2O4 + O2 + H2O → NaHSO4 + NaHSO3
These reactions exhibit complex pH-dependent equilibria involving bisulfite, thiosulfate, and sulfur dioxide.
With organic carbonyls
In the presence of aldehydes, sodium dithionite reacts either to form α-hydroxy-sulfinates at room temperature or to reduce the aldehyde to the corresponding alcohol above a temperature of 85 °C.[6][7] Some ketones are also reduced under similar conditions.
Applications
Industry
Being water-soluble, sodium dithionite is used as a reducing agent in some industrial dyeing processes. In the case of sulfur dyes and vat dyes, an otherwise water-insoluble dye can be reduced into a water-soluble alkali metal salt (e.g. indigo dye).[8]
Sodium dithionite can also be used for water treatment, aquarium water conditioners, gas purification, cleaning, and stripping. It has also been applied as a sulfonating agent. In addition to the textile industry, this compound is used in industries concerned with leather, foods, polymers, photography, and many others, often as a decolourising agent. It is even used domestically as a decoloring agent for white laundry, when it has been accidentally stained by way of a dyed item slipping into the high temperature washing cycle. It is usually available in 5 gram sachets termed hydrosulfite after the antiquated name of the salt.
It is the active ingredient in "Iron Out Rust Stain Remover", a commercial rust product.[9]
Laboratory
Sodium dithionite is often used in
Aqueous solutions of sodium dithionite were once used to produce '
Photography
It is used in Kodak fogging developer, FD-70. This is used in the second step in processing black and white positive images, for making slides. It is part of the Kodak Direct Positive Film Developing Outfit.[13]
Safety
The wide use of sodium dithionite is attributable in part to its low toxicity LD50 at 2.5 g/kg (rats, oral).[4]
See also
References
- S2CID 97124638.
- ISBN 978-0-08-037941-8.
- ^ ISBN 978-0-13-175553-6.
- ^ ISBN 978-3527306732.
- PMID 648533.
- ^ J. Org. Chem., 1980, 45 (21), pp 4126–4129, http://pubs.acs.org/doi/abs/10.1021/jo01309a011
- ^ "Aldehyde sulfoxylate systemic fungicides". google.com. Archived from the original on 27 April 2018. Retrieved 27 April 2018.
- .
- ^ "The Best Rust Removers for Restoring Every Surface". 23 March 2023.
- PMID 648533.
- ISBN 978-0471936237.
- ^ "Kodak Direct Positive Film 5246" (PDF). 125px.com. Kodak. Retrieved 6 November 2019.
External links
- Sodium dithionite - ipcs inchem[1]
- ^ "Sodium dithionite - ipcs inchem" (PDF). www.inchem.org. Berliln, Germany. 2004. Archived from the original (PDF) on 17 April 2018. Retrieved 15 June 2018.

