Sodium dodecyl sulfate
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium dodecyl sulfate | |
| Other names
Sodium monododecyl sulfate; Sodium lauryl sulfate; Sodium monolauryl sulfate; Sodium dodecanesulfate; dodecyl alcohol, hydrogen sulfate, sodium salt; n-dodecyl sulfate sodium; Sulfuric acid monododecyl ester sodium salt
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.005.263 |
| E number | E487 (thickeners, ...) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H25NaSO4 | |
| Molar mass | 288.372 g/mol |
| Appearance | white or cream-colored solid |
| Odor | odorless |
| Density | 1.01 g/cm3 |
| Melting point | 206 °C (403 °F; 479 K) |
| Surface tension: | |
| 8.2 mM at 25 °C[1] | |
Refractive index (nD)
|
1.461 |
| Pharmacology | |
| A06AG11 (WHO) | |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
1288 mg/kg (rat, oral) |
| Related compounds | |
Other anions
|
Sodium laureth sulfate Sodium myreth sulfate |
Other cations
|
Ammonium lauryl sulfate Potassium lauryl sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium dodecyl sulfate (SDS) or sodium lauryl sulfate (SLS), sometimes written sodium laurilsulfate, is an
Physicochemical properties

The critical micelle concentration (CMC) in water at 25 °C is 8.2 mM,[1] and the aggregation number at this concentration is usually considered to be about 62.[3] The micelle ionization fraction (α) is around 0.3 (or 30%).[4]
Applications
Cleaning and hygiene
SDS is mainly used in detergents for laundry with many cleaning applications.[5] It is a highly effective surfactant and is used in any task requiring the removal of oily stains and residues. For example, it is found in higher concentrations with industrial products including engine degreasers, floor cleaners, and car exterior cleaners.[citation needed]
It is a component in hand soap, toothpastes, shampoos, shaving creams, and bubble bath formulations, for its ability to create a foam (lather), for its surfactant properties, and in part for its thickening effect.[6]
Food additive
Sodium dodecyl sulfate, appearing as its synonym sodium lauryl sulfate (SLS), is considered a generally recognized as safe (GRAS) ingredient for food use according to the USFDA (21 CFR 172.822).[7] It is used as an emulsifying agent and whipping aid.[8] As an emulsifier in or with egg whites the United States Code of Federal Regulations require that it must not exceed 1,000 parts per million (0.1%) in egg white solids or 125 parts per million (0.0125%) in frozen or liquid egg whites and as a whipping agent for the preparation of marshmallows it must not exceed 0.5% of the weight of gelatine.[9] SLS is reported to temporarily diminish perception of sweetness.[10]
Laboratory applications
SDS is used in cleaning procedures,[11] and is commonly used as a component for lysing cells during RNA extraction or DNA extraction, inhibiting the activity of nucleases, enzymes that can degrade DNA, protecting the integrity of the isolated genetic material, and for denaturing proteins in preparation for electrophoresis in the SDS-PAGE technique.[12]
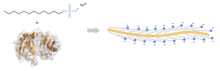
In the case of SDS-PAGE, the compound works by disrupting non-covalent bonds in the proteins, and so denaturing them, i.e. causing the protein molecules to lose their native conformations and shapes. By binding to proteins at a ratio of one SDS molecule per 2 amino acid residues, the negatively charged detergent provides all proteins with a similar net negative charge and therefore a similar charge-to-mass ratio.[13] In this way, the difference in mobility of the polypeptide chains in the gel can be attributed solely to their length as opposed to both their native charge and shape.[13][14] This separation based on the size of the polypeptide chain simplifies the analysis of protein molecules.[15]
Pharmaceutical applications
Sodium lauryl sulfate is a widely used in the pharmaceutical field as an ionic solubilizer and emulsifier that is suitable for applications in liquid dispersions, solutions, emulsions and micro emulsions, tablets, foams and semi-solids such as creams, lotions and gels.[16] Additionally, SLS aids in tablet wettability, as well as lubrication during manufacturing. Brand names of pharma-grade SLS include Kolliphor SLS and Kolliphor SLS Fine.[17]
Miscellaneous applications
SLS is used in an improved technique for preparing brain tissues for study by optical microscopy. The technique, which has been branded as CLARITY, was the work of Karl Deisseroth and coworkers at
Along with
Other uses
SLS has been proposed as a potentially effective topical microbicide, for intravaginal use, to inhibit and possibly prevent infection by various enveloped and non-enveloped viruses such as the herpes simplex viruses, HIV, and the Semliki Forest virus.[21][22]
Liquid membranes formed from SDS in water have been demonstrated to work as unusual particle separators.[23] The device acts as a reverse filter, allowing large particles to pass while capturing smaller particles.
Production
SDS is
Safety
SDS is not
Oral concerns
SDS is a common ingredient in toothpastes due to its low cost,[33] its lack of impact on taste,[33] and its desirable action as a foaming agent.[33]
VSCs
SDS may reduce the amount of
Dry mouth
Primary sources from the group of Irma Rantanen at University of Turku, Finland claim that SLS-containing pastes cause more dry mouth (xerostomia) than their proposed alternative. However, a 2011 Cochrane review of these studies, and of the more general area, concludes that there "is no strong evidence... that any topical therapy is effective for relieving the symptom of dry mouth."[35]
Mouth ulceration
A safety concern has been raised on the basis of several studies regarding the effect of toothpaste SDS on
See also
- Sodium tetradecyl sulfate, another anionic surfactant in common use
- Mouth ulcer
References
- ^ a b P. Mukerjee, P. & Mysels, K. J. (1971), "Critical Micelle Concentration of Aqueous Surfactant Systems," NSRDS-NBS 36, Washington, DC: US. Government Printing Office.[full citation needed][page needed]
- ISBN 978-3527306732.
- .
- .
- ]
- ^ "Household Products Database – Health and Safety Information on Household Products". nih.gov. Archived from the original on 12 June 2018. Retrieved 13 March 2016.
- ^ "21 CFR 172.822 – Sodium lauryl sulfate". gpo.gov. Retrieved 13 March 2016.
- ^ Igoe, R. S. (1983). Dictionary of food ingredients. New York: Van Nostrand Reinhold Co.[page needed]
- ^ "21 CFR 172.822 – Sodium lauryl sulfate". Retrieved 19 August 2021.
- ISBN 9780841209442.
- ^ "Sodium Lauryl Sulfate – National Library of Medicine HSDB Database". toxnet.nlm.nih.gov. Retrieved 2017-02-16.
- ^ The acronym expands to "sodium dodecyl sulfate-polyacrylamide gel electrophoresis."
- ^ OCLC 794620168.
- ISBN 978-0470087664.
- ISBN 978-1-891-78600-6.
- ^ "Pharmaceuticals". pharmaceutical.basf.com. Retrieved 2021-04-27.
- ^ "Kolliphor® SLS". pharmaceutical.basf.com. Retrieved 2021-04-27.
- better source needed]
- PMID 23575631.
Obtaining high-resolution information from a complex system, while maintaining the global perspective needed to understand system function, represents a key challenge in biology. Here we address this challenge with a method (termed CLARITY) for the transformation of intact tissue into a nanoporous hydrogel-hybridized form (crosslinked to a three-dimensional network of hydrophilic polymers) that is fully assembled but optically transparent and macromolecule-permeable.
- .
- PMID 11899262.
- PMID 10618073.
- PMID 30151426.
- S2CID 84286689.
- ^ Gloxhuber, C., & Kunster, K. (1992). Anionic Surfactants: Biochemistry, toxicology, dermatology (2nd ed.). New York.
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: multiple names: authors list (link)[page needed] - ^ US 3,491,033, "Process of making solid foams from polymer emulsions", published 1970
- S2CID 34123578. Retrieved 13 March 2016.
[Quoting:] Carcinogenesis. A one-year chronic oral study using beagles showed that Sodium Lauryl Sulfate at concentrations up to 2% in the diet was not tumorigenic or carcinogenic. [p. 157] / Summary… In mutagenesis studies, rats fed 1.13% and 0.56% Sodium Lauryl Sulfate in the diet for 90 days produced no more chromosomal aberrations or clastogenic effects than did a control diet. [p. 175]. / Conclusion. Sodium Lauryl Sulfate and Ammonium Lauryl Sulfate appear to be safe in formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. In products intended for prolonged contact with skin, concentrations should not exceed 1%. [p. 176.]
. - ^ Wilma F. Bergfeld, Chair, and the Cosmetic Ingredient Review (CIR) program Expert Panel (2005). "Final report on the safety assessment of sodium lauryl sulfate and ammonium lauryl sulfate" (PDF). Int. J. Toxicol. 24 (1): 1–102, esp. 89–98. Retrieved 13 March 2016.
[Quoting:] Sodium Lauryl Sulfate and Ammonium Lauryl Sulfate appear to be safe in formulations designed for discontinuous, brief use followed by thorough rinsing from the surface of the skin. In products intended for prolonged contact with skin, concentrations should not exceed 1%… New studies confirmed the irritant properties of these ingredients and reinforced the concentration limit of 1% or leave-on uses established by the [earlier] Panel. [p. 89] / The available studies that looked for carcinogenesis failed to find evidence that Ammonium Lauryl Sulfate are [sic.] carcinogenic. None of the available data suggested that SLS or Ammonium Lauryl Sulfate could be carcinogenic. Despite assertions to the contrary on the Internet, the carcinogenicity of these ingredients is only a rumor. [pp. 89ff]
{{cite journal}}: CS1 maint: multiple names: authors list (link). - S2CID 35890797.
- S2CID 37806228.
- PMID 7979441.
- S2CID 10409476.
- ^ PMID 23817056.
- ^ PMID 23817059.
- , referenced therein.
- ^ "Mouth ulcers". NHS. 18 October 2017.
do not use toothpaste containing sodium lauryl sulphate
- PMID 7825393.
