Sodium naphthalene
(Redirected from
Sodium naphthalenide
)
| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium naphthalenide | |
| Systematic IUPAC name
Sodium naphthalen-1-ide | |
| Other names
sodium naphthalenide, sodium naphthalide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.020.420 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
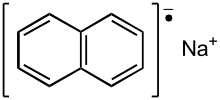
| Na+[C10H8]− | |
| Molar mass | 151.164 g·mol−1 |
| Appearance | Deep green crystals |
| Related compounds | |
Other anions
|
Lithium naphthalene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium naphthalene is an organic
Preparation and properties

The alkali metal naphthalene salts are prepared by stirring the metal with
signal near g = 2.0. Its deep green color arises from absorptions centered at 463 and 735 nm.Several solvates of sodium naphthalenide have been characterized by
pm and the other nine C−C bonds elongate by 2–3 pm. The net effect is that reduction weakens the bonding.[5][6]
Reactions
Redox
With a
NHE, the naphthalene radical anion is a strong reducing agent.[1]
Protonation
The anion is strongly
alcohols. These reactions afford dihydronaphthalene
:
As a ligand
Alkali metal salts of the naphthalene radical anion are used to prepare complexes of naphthalene.[7]
Related reagents
References
- ^ PMID 11848774.
- .
- ISBN 0-471-84997-9
- ISBN 978-0-08-022057-4.
- .
- .
- S2CID 73436073.
