Sodium nitrite
| |||
| |||
 Unit cell of sodium nitrite under standard conditions
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Sodium nitrite
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.028.687 | ||
| EC Number |
| ||
| E number | E250 (preservatives) | ||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1500 3287 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
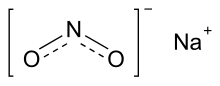
| NaNO2 | |||
| Molar mass | 68.9953 g/mol | ||
| Appearance | white or slightly yellowish solid | ||
| Density | 2.168 g/cm3 | ||
| Melting point | 271 °C (520 °F; 544 K) (decomposes at 320 °C) | ||
| 71.4 g/100 mL (0 °C) 84.8 g/100 mL (25 °C) 160 g/100 mL (100 °C) | |||
| Solubility | soluble in methanol (4.4 g/100 mL) ethanol slightly soluble in diethyl ether (0.3 g/100 mL) very soluble in ammonia | ||
| Acidity (pKa) | ~9 | ||
| −14.5·10−6 cm3/mol | |||
Refractive index (nD)
|
1.65 | ||
| Structure[1] | |||
| orthorhombic | |||
| Im2m | |||
a = 3.5653(8) Å, b = 5.5728(7) Å, c = 5.3846(13) Å
| |||
Formula units (Z)
|
2 | ||
| Thermochemistry | |||
Std molar
entropy (S⦵298) |
106 J/mol K | ||
Std enthalpy of (ΔfH⦵298)formation |
−359 kJ/mol[2] | ||
Gibbs free energy (ΔfG⦵)
|
−295 kJ/mol | ||
| Pharmacology | |||
| V03AB08 (WHO) | |||
| Hazards | |||
| GHS labelling:[3] | |||
  
| |||
| Danger | |||
| H272, H301, H319, H400 | |||
| P220, P273, P301+P310, P305+P351+P338 | |||
| NFPA 704 (fire diamond) | |||
| 489 °C (912 °F; 762 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
180 mg/kg (rats, oral) | ||
| Safety data sheet (SDS) | "Sodium nitrite". Safety Data Sheet. Sigma-Aldrich. 28 December 2022. | ||
| Related compounds | |||
Other anions
|
Sodium nitrate | ||
Other cations
|
Potassium nitrite Ammonium nitrite Lithium nitrite | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Sodium nitrite is an
Uses
Industrial chemistry
The main use of sodium nitrite is for the industrial production of organonitrogen compounds. It is a reagent for conversion of
It is used in a variety of metallurgical applications, for phosphatizing and detinning.[4]
Sodium nitrite is an effective corrosion inhibitor and is used as an additive in industrial greases,[5] as an aqueous solution in closed loop cooling systems, and in a molten state as a heat transfer medium.[6]
Food additive and preservative
Sodium nitrite is used to speed up the curing of meat, inhibit the germination of Clostridium botulinum spores, and also impart an attractive pink color.[7][8][9] Nitrite reacts with the meat myoglobin to cause color changes, first converting to nitrosomyoglobin (bright red), then, on heating, to nitrosohemochrome (a pink pigment).[10]
Historically, salt has been used for the preservation of meat. The salt-preserved meat product was usually brownish-gray in color. When sodium nitrite is added with the salt, the meat develops a red, then pink color, which is associated with cured meats such as ham, bacon, hot dogs, and bologna.[11]
In the early 1900s, irregular curing was commonplace. This led to further research surrounding the use of sodium nitrite as an
Nitrite has the E number E250. Potassium nitrite (E249) is used in the same way. It is approved for usage in the European Union,[14][15] USA,[16] and Australia and New Zealand.[17]
In meat processing, sodium nitrite is never used in a pure state but always mixed with common salt. This mixture is known as nitrited salt, curing salt or nitrited curing salt. In Europe, nitrited curing salt contains between 99.1% and 99.5% common salt and between 0.5% and 0.9% nitrite. In the US, nitrited curing salt is dosed at 6% and must be remixed with salt before use.[18]
Color and taste
The appearance and taste of meat is an important component of consumer acceptance.[12] Sodium nitrite is responsible for the desirable red color (or shaded pink) of meat.[12] Very little nitrite is needed to induce this change.[12] It has been reported that as little as 2 to 14 parts per million (ppm) is needed to induce this desirable color change.[19] However, to extend the lifespan of this color change, significantly higher levels are needed.[19] The mechanism responsible for this color change is the formation of nitrosylating agents by nitrite, which has the ability to transfer nitric oxide that subsequently reacts with myoglobin to produce the cured meat color.[19] The unique taste associated with cured meat is also affected by the addition of sodium nitrite.[12] However, the mechanism underlying this change in taste is still not fully understood.[19]
Inhibition of microbial pathogens
In conjunction with salt and pH levels, sodium nitrite reduces the ability of
Sodium nitrite has shown varying degrees of effectiveness for controlling growth of other spoilage or disease causing microorganisms.
Other food additives (such as
Inhibition of lipid peroxidation
Sodium nitrite is also able to effectively delay the development of oxidative
Medication
 Chemical structure | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | FDA Professional Drug Information |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
JSmol) | |
| |
| |
Sodium nitrite is used as a medication together with sodium thiosulfate to treat cyanide poisoning.[26] It is recommended only in severe cases of cyanide poisoning and has largely been replaced by use of hydroxocobalamin.[27]
NaNO₂ side effects are chiefly related to creation of methemoglobinemia and vasodilation. Side effects can include
Sodium nitrite came into medical use in the 1920s and 1930s.[32][33] It is on the World Health Organization's List of Essential Medicines.[34]
Suicide
Several academic publications in 2020 and 2021 have discussed the toxicity of sodium nitrite, and an apparent recent increase in suicides using sodium nitrite which had been ordered online.
Toxicity
Sodium nitrite is toxic. signs of sodium nitrite poisoning are as follows:
"Symptoms of [nitrite] poisoning can vary depending on the amount and duration of the exposure. Those with very mild methemoglobinemia might not have any symptoms at all, or might appear a little pale and feel tired. Moderate-to-severe poisoning is associated with cyanosis (blueness of the skin), confusion, loss of consciousness, seizures, abnormal heart rhythms, and death."
With prompt action, sodium nitrite poisoning is reversible using an antidote, methylene blue.[44] It has been reported[52] that sodium nitrite poisoning can also be detected post-mortem:
"Postmortem detection of [methemoglobinemia] is typically established via screening techniques such as scene evidence suggesting fatal consumption of a toxic salt in addition to the characteristic grey-purple lividity observed upon the body. The diagnosis can be established via postmortem blood testing demonstrating elevated methemoglobin saturation. Additionally, we have confirmed that postmortem MRI in cases of [methemoglobinemia] demonstrates a T1-bright (hyperintense) signal of the blood; both within intracardiac blood on chest MRIs and postmortem blood samples in tubes."
Death by sodium nitrite ingestion can happen at lower doses than the
Occurrence in vegetables
Nitrites do not occur naturally in vegetables in significant quantities,[60] but deliberate fermentation of celery juice, for instance, with a naturally high level of nitrates, can produce nitrite levels sufficient for commercial meat curing.[61] Boiling vegetables does not affect nitrite levels.[62]
The presence of nitrite in animal tissue is a consequence of metabolism of nitric oxide, an important neurotransmitter.[63] Nitric oxide can be created de novo from nitric oxide synthase utilizing arginine or from ingested nitrite.[64]
Pigs
Because of sodium nitrite's high level of toxicity to swine (Sus scrofa) it is now being developed in Australia to control feral pigs and wild boar.[65][66] The sodium nitrite induces methemoglobinemia in swine, i.e. it reduces the amount of oxygen that is released from hemoglobin, so the animal will feel faint and pass out, and then die in a humane manner after first being rendered unconscious.[67] The Texas Parks and Wildlife Department operates a research facility at Kerr Wildlife Management Area, where they examine feral pig feeding preferences and bait tactics to administer sodium nitrite.[68]
Cancer
Adding nitrites to meat has been shown to generate known carcinogens such as
Nitrosamines can be formed during the curing process used to preserve meats, when sodium nitrite-treated meat is cooked, and also from the reaction of nitrite with secondary amines under acidic conditions (such as occurs in the human stomach). Dietary sources of nitrosamines include US cured meats preserved with sodium nitrite as well as the dried salted fish eaten in Japan. In the 1920s, a significant change in US meat curing practices resulted in a 69% decrease in average nitrite content. This event preceded the beginning of a dramatic decline in gastric cancer mortality.
Ingesting nitrite under conditions that result in endogenous nitrosation has been classified as "probably carcinogenic to humans" by International Agency for Research on Cancer (IARC).[77][78]
Sodium nitrite consumption has also been linked to the triggering of migraines in individuals who already experience them.[79]
One study has found a correlation between highly frequent ingestion of meats cured with
Production
Industrial production of sodium nitrite follows one of two processes, the reduction of nitrate salts, or the oxidation of lower nitrogen oxides.
One method uses molten sodium nitrate as the salt, and lead which is oxidized, while a more modern method uses scrap iron filings to reduce the nitrate.[4][84]
A more commonly used method involves the general reaction of nitrogen oxides in alkaline aqueous solution, with the addition of a catalyst. The exact conditions depend on which nitrogen oxides are used, and what the oxidant is, as the conditions need to be carefully controlled to avoid over oxidation of the nitrogen atom.[4]
Sodium nitrite has also been produced by reduction of nitrate salts by exposure to heat, light, ionizing radiation, metals, hydrogen, and electrolytic reduction.[85]
Chemical reactions
In the laboratory, sodium nitrite can be used to destroy excess sodium azide.[86][87]
Above 330 °C sodium nitrite decomposes (in air) to sodium oxide, nitric oxide and nitrogen dioxide.[88]
Sodium nitrite can also be used in the production of nitrous acid:
The nitrous acid then, under normal conditions, decomposes:
The resulting nitrogen dioxide hydrolyzes to a mixture of nitric and nitrous acids:
Isotope labelling 15N

In organic synthesis isotope enriched sodium nitrite-15N can be used instead of normal sodium nitrite as their reactivity is nearly identical in most reactions.
The obtained products carry isotope 15N and hence nitrogen NMR can be efficiently carried out.[89]
References
- ^ Gohda T, Ichikawa M (November 1996). "The Refinement of the Structure of Ferroelectric Sodium Nitrite". Journal of the Korean Physical Society. 29: 551–554.
- ISBN 978-0-618-94690-7.
- ^ "GESTIS-Stoffdatenbank sodium nitrite". gestis.dguv.de. Retrieved 10 December 2021.
- ^ ISBN 978-3-527-30673-2.
- S2CID 94383988.
- ^ "Sodium Nitrite". General Chemical. Archived from the original on 22 May 2013. Retrieved 28 September 2012.
- ^ from the original on 10 February 2021. Retrieved 14 February 2021.
In trade journals of the 1960s, the firms who sold nitrite powders to ham-makers spoke quite openly about how the main advantage was to increase profit margins by speeding up production.
- .
- ^ a b "Use and Removal of Nitrite in Meat Products". Archived from the original on 9 March 2023. Retrieved 23 April 2023.
- .
- ^ ""Meat Pigment Chemistry", taken from IFT Mini-Experiments in Food Science Series" (PDF). Archived from the original (PDF) on 3 December 2019. Retrieved 28 January 2019.
- ^ PMID 22487433.
- ^ "Science Says: Are hot dogs healthier without added nitrites? | Lifestyle from CTV News". www.ctvnews.ca. 30 June 2017.
- ^ "Approved additives and E numbers". Food Standards Agency. Retrieved 9 January 2023.
- ^ "Opinions of the Scientific Committee for Food on: Nitrates and Nitrite" (PDF). Food Science and Techniques. European Commission. Retrieved 1 April 2018.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". Food and Drug Administration. Retrieved 27 October 2011.
- ^ "Australia New Zealand Food Standards Code - Standard 1.2.4 - Labelling of Ingredients". www.legislation.gov.au. Retrieved 9 January 2023.
- ISBN 978-1785786112.
- ^ a b c d e f g h i j k l Sindelar J, Milkowski A (November 2011). "Sodium Nitrite in Processed Meat and Poultry Meats: A Review of Curing and Examining the Risk/Benefit of Its Use" (PDF). American Meat Science Association. 3: 1–14.
- PMID 29531192.
- PMID 6758699.
- .
- ^ https://www.tga.gov.au/therapeutic-goods-exempted-pregnancy-categorisation
- FDA. Retrieved 22 October 2023.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ ISBN 9789241547659.
- ^ "Sodium Nitrite Solution for Injection - Summary of Product Characteristics (SPC) - (eMC)". www.medicines.org.uk. Archived from the original on 18 September 2017. Retrieved 15 January 2017.
- S2CID 21116328.
- PMID 29939573, retrieved 7 December 2023
- .
- ^ a b "Sodium Nitrite Injection - FDA prescribing information, side effects and uses". www.drugs.com. Archived from the original on 18 January 2017.
- .
- .
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Durão C, et al., 2020.; Durão C, et al., 2021.; Tomsia M, et al., 2021.; McCann SD, Kennedy JM, et al., 2021.; Dean DE, et al., 2021.; Hickey TB, et al., 2021.; Harvey M, et al., 2010.; McCann SD, Tweet MS, Wahl MS, 2021.; Sedhai YR, et al., 2022.; Mudan A, et al., 2020.
- ^ Twohey M (9 December 2021). "Where the Despairing Log On, and Learn Ways to Die". The New York Times. Retrieved 24 July 2022.
- ^ Belanger A (7 October 2022). "Amazon 'suicide kits' have led to teen deaths, according to new lawsuit". Ars Technica. Retrieved 26 August 2023.
- ^ Stempel J (28 June 2023). "Judge dismisses lawsuit claiming Amazon sold 'suicide kits' to teenagers". Reuters. Retrieved 3 November 2023.
- ^ Director, eBay (UK) Limited (8 January 2021). "Re Jason Thompson (deceased) – Sodium Nitrite" (PDF). Retrieved 26 August 2023 – via Judiciaries of the United Kingdom.
- ^ Murphy A (25 July 2023). "Here's why the world will be watching Kenneth Law's court battle". CTV Toronto. Retrieved 26 August 2023.
- ^ Crawford A (25 August 2023). "88 UK deaths linked to Canada 'poison seller'". BBC News. Retrieved 26 August 2023.
- ^ "Chemical Used in Suicides Faces Sales Ban Under Proposed Law". news.bloomberglaw.com. Retrieved 3 November 2023.
- ISSN 0041-008X.
- ^ S2CID 258324908.
- ^ Su M, McRae M (8 September 2022). "2022 Health Advisory #22 Poisonings and Death Related to Intentional Sodium Nitrite Ingestions" (PDF). New York City Department of Health and Mental Hygiene. Retrieved 4 November 2023.
- .
- ^ Crellin J. "How toxic is it?" (PDF). The Association for Science Educators. Retrieved 6 February 2022.
- Oxford University. Archived from the originalon 10 April 2008.
- PMID 27563472.
- PMID 36360874.
- ^ "Nitrate and Nitrite Poisoning". www.poison.org. Retrieved 4 November 2023.
- PMID 34298207.
- PMID 2271282.
- PMID 541641.
- ^ "Chinese teacher sentenced to death for poisoning nursery children". BBC News. 29 September 2020.
- ^ "Teacher in China sentenced to death for poisoning children's porridge". The Guardian. Agence France-Presse in Beijing. 29 September 2020.
- ^ "'General Hospital' Actress Lindsey Pearlman's Cause of Death Revealed". The Hollywood Reporter. 16 August 2022.
- ^ "Police charge Mississauga man in connection with sale, distribution of sodium nitrite in GTA". CTV News. 3 May 2023.
- ^ "The Use and Removal of Nitrite in Meat Products | FAQs". The Food Safety Authority of Ireland. Archived from the original on 27 June 2022. Retrieved 10 February 2021.
- ISBN 978-0-12-227055-0.
- ^ "Is celery juice a viable alternative to nitrites in cured meats?". Office for Science and Society. Retrieved 14 September 2022.
- .
- PMID 7866533.
- PMID 9851368.
- ^ Lapidge S, Wishart J, Smith M, Staples L (4 May 2009). Is America Ready for a Humane Feral Pig Toxicant?. Wildlife Damage Management Conference. Saratoga Springs, NY.
- ^ WO 2008/104028, Cowled BD, Lapidge SJ, Humphrys S, Staples L, "Nitrite Salts as Poisons in Baits for Omnivores", published 2008
- ^ Porter S, Kuchel T (2010). Assessing the humaness and efficacy of a new feral pig bait in domestic pigs. Study PC0409 (PDF). Canberra, South Australia: Veterinary Services Division, Institute of Medical and Veterinary Science. p. 11.
- ^ "Hogs Wild – Fighting the Feral Pig Problem". Texas Parks and Wildlife. 21 February 2013. Archived from the original on 12 December 2021. Retrieved 1 April 2018 – via YouTube.
- ^ "Known and Probable Human Carcinogens". www.cancer.org. Retrieved 28 January 2019.
- ^ "IARC Monographs evaluate consumption of red meat and processed meat" (PDF). International Agency for Research on Cancer. 26 October 2015. Archived from the original (PDF) on 18 January 2021. Retrieved 14 February 2021.
Processed meat was classified as carcinogenic to humans (Group 1), based on sufficient evidence in humans that the consumption of processed meat causes colorectal cancer.
- PMID 11171883.
- PMID 2492212.
- ^ "Research Newsletter". Linus Pauling Institute. 1 July 2014. Retrieved 1 April 2018.
- NAID 80001710206.
- ISBN 978-1-4822-9784-3.[page needed]
- ISBN 978-0-444-51721-0.
- ^ "List of classifications, Volumes 1–116 – IARC Monographs on the Evaluation of Carcinogenic Risks to Humans". International Agency for Research on Cancer (IARC) – World Health Organization (WHO). 2010. Retrieved 25 September 2016.
- ISBN 978-92-832-1294-2. Retrieved 25 September 2016.
- U.S. Food and Drug Administration. 1998.
- ^ Hitti M (17 April 2007). "Study: Cured Meats, COPD May Be Linked". WebMD Medical News.
- PMID 17255565.
- ^ Hitti M (17 April 2007). "Study: Cured Meats, COPD May Be Linked". WebMD Medical News.
- PMID 17255565.
- PMID 15682502.
- ISBN 978-0-471-23896-6.
- ^ "Sodium Azide". Hazardous Waste Management. Northeastern University. March 2003. Archived from the original on 4 November 2007.
- ISBN 978-0-309-05229-0.
- S2CID 95532988.
- S2CID 105312344.
Sources
- Durão C, Pedrosa F, Dinis-Oliveira RJ (July 2020). "A fatal case by a suicide kit containing sodium nitrite ordered on the internet". Journal of Forensic and Legal Medicine. 73: 101989. S2CID 219909654.
- Durão C, Pedrosa F, Dinis-Oliveira RJ (June 2021). "Another suicide by sodium nitrite and multiple drugs: an alarming trend for "exit"?". Forensic Science, Medicine, and Pathology. 17 (2): 362–366. S2CID 227180461.
- Tomsia M, Głaz M, Nowicka J, Szczepański M (July 2021). "Sodium nitrite detection in costal cartilage and vitreous humor - Case report of fatal poisoning with sodium nitrite". Journal of Forensic and Legal Medicine. 81: 102186. S2CID 235268052.
- McCann SD, Kennedy JM, Tweet MS, Bryant SM (March 2021). "Sodium Nitrite Ingestion: an Emerging Trend in Suicide Attempts Shared via Online Communities". The Journal of Emergency Medicine. 60 (3): 409–412. S2CID 232218908.
- Dean DE, Looman KB, Topmiller RG (July 2021). "Fatal methemoglobinemia in three suicidal sodium nitrite poisonings". Journal of Forensic Sciences. 66 (4): 1570–1576. S2CID 231952466.
- Hickey TB, MacNeil JA, Hansmeyer C, Pickup MJ (September 2021). "Fatal methemoglobinemia: A case series highlighting a new trend in intentional sodium nitrite or sodium nitrate ingestion as a method of suicide". Forensic Science International. 326: 110907. PMID 34298207.
- Harvey M, Cave G, Chanwai G (October 2010). "Fatal methaemoglobinaemia induced by self-poisoning with sodium nitrite". Emergency Medicine Australasia. 22 (5): 463–465. S2CID 38780081.
- McCann SD, Tweet MS, Wahl MS (December 2021). "Rising incidence and high mortality in intentional sodium nitrite exposures reported to US poison centers". Clinical Toxicology. 59 (12): 1264–1269. S2CID 232431594.
- Sedhai YR, Atreya A, Basnyat S, Phuyal P, Pokhrel S (June 2022). "The use of sodium nitrite for deliberate self-harm, and the online suicide market: Should we care?". The Medico-Legal Journal. 90 (2): 79–80. S2CID 233429578.
- Mudan A, Repplinger D, Lebin J, Lewis J, Vohra R, Smollin C (September 2020). "Severe Methemoglobinemia and Death From Intentional Sodium Nitrite Ingestions". The Journal of Emergency Medicine. 59 (3): e85–e88. S2CID 220797852.
Further reading
- National Toxicology Program (May 2001). "Toxicology and carcinogenesis studies of sodium nitrite (CAS NO. 7632-00-0) in F344/N rats and B6C3F1 mice (drinking water studies)". National Toxicology Program Technical Report Series. 495: 7–273. PMID 12563346.
External links
- "Sodium nitrite". International Chemical Safety Card. IPCS INCHEM. 1120.
- Epley RJ, Addis PB, Warthesen JJ (2011). "Nitrite in Meat". University of Minnesota. Archived from the original on 1 January 2011.
| HNO2 | He | ||||||||||||||||||
| LiNO3 | Be(NO3)2 | B | C(NO2)4, CH(NO2)3, CH2(NO2)2, CH3(NO2) |
N(NO2)3 H2NNO2 xNO3NO2 |
N2O3 | NO2F | Ne | ||||||||||||
| NaNO2 | Mg(NO2)2 | Al(NO2)3 | Si | P | S | NO2Cl | Ar | ||||||||||||
| KNO2 | Ca(NO2)2 | Sc(NO2)3 | Ti | VO(NO2)3 | Cr(NO2)3 | Mn(NO2)2 | Fe(NO2)3 | Co(NO2)2, Co(NO2)3 |
Ni(NO2)2 | Cu(NO2)2 | Zn(NO2)2 | Ga(NO2)3 | Ge | As | Se | NO2Br | Kr | ||
| RbNO2 | Sr(NO2)2 | Y(NO2)3 | Zr | Nb | Mo | Tc | Ru | Rh | Pd(NO2)2 | AgNO2 | Cd(NO2)2 | In | Sn | Sb | Te | NO2I | Xe | ||
| CsNO2 | Ba(NO2)2 | Hf | Ta | W | Re | Os | Ir | Pt(NO2)2, [Pt(NO2)4]2− |
Au | Hg2(NO2)2, Hg(NO2)2 |
TlNO2 | Pb(NO2)2 | Bi(NO2)3 BiO(NO2) |
Po | At | Rn | |||
| Fr | Ra | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| ↓ | |||||||||||||||||||
| La(NO2)3 | Ce(NO2)3 | Pr(NO2)3 | Nd(NO2)3 | Pm | Sm(NO2)3 | Eu(NO2)2 | Gd(NO2)3 | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||||
| Ac | Th | Pa | UO2(NO2)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||||












