Solubility


In
. Insolubility is the opposite property, the inability of the solute to form such a solution.The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a saturated solution, one in which no more solute can be dissolved.[1] At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscible in all proportions" (or just "miscible").[2]
The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,[3] and a solid or liquid can be "dissolved" in a gas only by passing into the gaseous state first.
The solubility mainly depends on the composition of solute and solvent (including their pH and the presence of other dissolved substances) as well as on temperature and pressure. The dependency can often be explained in terms of interactions between the particles (atoms, molecules, or ions) of the two substances, and of thermodynamic concepts such as enthalpy and entropy.
Under certain conditions, the concentration of the solute can exceed its usual solubility limit. The result is a supersaturated solution, which is metastable and will rapidly exclude the excess solute if a suitable nucleation site appears.[4]
The concept of solubility does not apply when there is an irreversible
The concept and measure of solubility are extremely important in many sciences besides chemistry, such as geology, biology, physics, and oceanography, as well as in engineering, medicine, agriculture, and even in non-technical activities like painting, cleaning, cooking, and brewing. Most chemical reactions of scientific, industrial, or practical interest only happen after the reagents have been dissolved in a suitable solvent. Water is by far the most common such solvent.
The term "soluble" is sometimes used for materials that can form colloidal suspensions of very fine solid particles in a liquid.[5] The quantitative solubility of such substances is generally not well-defined, however.
Quantification of solubility
The solubility of a specific solute in a specific solvent is generally expressed as the concentration of a saturated solution of the two.[1] Any of the several ways of expressing concentration of solutions can be used, such as the mass, volume, or amount in moles of the solute for a specific mass, volume, or mole amount of the solvent or of the solution.
Per quantity of solvent
In particular, chemical
Alternatively, the solubility of a solute can be expressed in moles instead of mass. For example, if the quantity of solvent is given in
Per quantity of solution
The solubility of a substance in a liquid may also be expressed as the quantity of solute per quantity of solution, rather than of solvent. For example, following the common practice in
In more specialized contexts the solubility may be given by the
Liquid and gaseous solutes
For solutions of liquids or gases in liquids, the quantities of both substances may be given volume rather than mass or mole amount; such as litre of solute per litre of solvent, or litre of solute per litre of solution. The value may be given as a percentage, and the abbreviation "v/v" for "volume per volume" may be used to indicate this choice.
Conversion of solubility values
Conversion between these various ways of measuring solubility may not be trivial, since it may require knowing the density of the solution — which is often not measured, and cannot be predicted. While the total mass is conserved by dissolution, the final volume may be different from both the volume of the solvent and the sum of the two volumes.[7]
Moreover, many solids (such as acids and salts) will dissociate in non-trivial ways when dissolved; conversely, the solvent may form coordination complexes with the molecules or ions of the solute. In those cases, the sum of the moles of molecules of solute and solvent is not really the total moles of independent particles solution. To sidestep that problem, the solubility per mole of solution is usually computed and quoted as if the solute does not dissociate or form complexes -- that is, by pretending that the mole amount of solution is the sum of the mole amounts of the two substances.
Qualifiers used to describe extent of solubility
The extent of solubility ranges widely, from infinitely soluble (without limit, i.e.
| Term | Range (msv/msu) | Example | g/dL | msv/msu |
|---|---|---|---|---|
| Very soluble | <1 | calcium nitrate | 158.7 | 0.63 |
| Freely soluble | 1 to 10 | calcium chloride | 65 | 1.54 |
| Soluble | 10 to 30 | sodium oxalate | 3.9 | 26 |
| Sparingly soluble | 30 to 100 | |||
| Slightly soluble | 100 to 1000 | calcium sulfate | 0.21 | 490 |
| Very slightly soluble | 1000 to 10,000 | dicalcium phosphate | 0.02 | 5000 |
| Practically insoluble or insoluble | ≥ 10,000 | barium sulfate | 0.000245 | 409000 |
The thresholds to describe something as insoluble, or similar terms, may depend on the application. For example, one source states that substances are described as "insoluble" when their solubility is less than 0.1 g per 100 mL of solvent.[9]
Molecular view
Solubility occurs under dynamic equilibrium, which means that solubility results from the simultaneous and opposing processes of
The term solubility is also used in some fields where the solute is altered by solvolysis. For example, many metals and their oxides are said to be "soluble in hydrochloric acid", although in fact the aqueous acid irreversibly degrades the solid to give soluble products. It is also true that most ionic solids are dissolved by polar solvents, but such processes are reversible. In those cases where the solute is not recovered upon evaporation of the solvent, the process is referred to as solvolysis. The thermodynamic concept of solubility does not apply straightforwardly to solvolysis.
When a solute dissolves, it may form several species in the solution. For example, an
2, will contain the series [Fe(H2O)x(OH)x](2x)+ as well as other species. Furthermore, the solubility of ferrous hydroxide and the composition of its soluble components depend on pH
Factors affecting solubility
Solubility is defined for specific
]The solubility of one substance in another is determined by the balance of intermolecular forces between the solvent and solute, and the entropy change that accompanies the solvation. Factors such as temperature and pressure will alter this balance, thus changing the solubility.
Solubility may also strongly depend on the presence of other species dissolved in the solvent, for example,
For a solid that dissolves in a redox reaction, solubility is expected to depend on the potential (within the range of potentials under which the solid remains the thermodynamically stable phase). For example, solubility of gold in high-temperature water is observed to be almost an order of magnitude higher (i.e. about ten times higher) when the redox potential is controlled using a highly oxidizing Fe3O4-Fe2O3
Solubility (metastable, at concentrations approaching saturation) also depends on the physical size of the crystal or droplet of solute (or, strictly speaking, on the specific surface area or molar surface area of the solute).[11] For quantification, see the equation in the article on solubility equilibrium. For highly defective crystals, solubility may increase with the increasing degree of disorder. Both of these effects occur because of the dependence of solubility constant on the Gibbs energy of the crystal. The last two effects, although often difficult to measure, are of practical importance.[citation needed] For example, they provide the driving force for precipitate aging (the crystal size spontaneously increasing with time).
Temperature
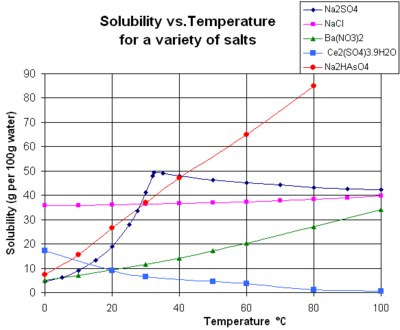
The solubility of a given solute in a given solvent is function of temperature. Depending on the change in
Gaseous solutes exhibit more complex behavior with temperature. As the temperature is raised, gases usually become less soluble in water (exothermic dissolution reaction related to their hydration) (to a minimum, which is below 120 °C for most permanent gases[13]), but more soluble in organic solvents (endothermic dissolution reaction related to their solvation).[12]
The chart shows solubility curves for some typical solid inorganic
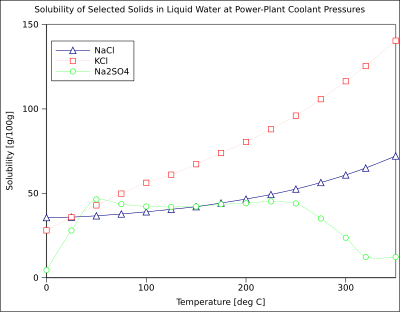
The solubility of
Pressure
For condensed phases (solids and liquids), the pressure dependence of solubility is typically weak and usually neglected in practice. Assuming an ideal solution, the dependence can be quantified as:
where the index iterates the components, is the mole fraction of the -th component in the solution, is the pressure, the index refers to constant temperature, is the
The pressure dependence of solubility does occasionally have practical significance. For example, precipitation fouling of oil fields and wells by calcium sulfate (which decreases its solubility with decreasing pressure) can result in decreased productivity with time.
Solubility of gases
Henry's law is used to quantify the solubility of gases in solvents. The solubility of a gas in a solvent is directly proportional to the partial pressure of that gas above the solvent. This relationship is similar to Raoult's law and can be written as:
where is a temperature-dependent constant (for example, 769.2
(O2) in water at 298 K), is the partial pressure (in atm), and is the concentration of the dissolved gas in the liquid (in mol/L).The solubility of gases is sometimes also quantified using
In the presence of small
Henry's law is valid for gases that do not undergo change of chemical speciation on dissolution. Sieverts' law shows a case when this assumption does not hold.
The
Polarity
A popular aphorism used for predicting solubility is "like dissolves like" also expressed in the Latin language as "Similia similibus solventur".[19] This statement indicates that a solute will dissolve best in a solvent that has a similar chemical structure to itself, based on favorable entropy of mixing. This view is simplistic, but it is a useful rule of thumb. The overall solvation capacity of a solvent depends primarily on its polarity.[a] For example, a very polar (hydrophilic) solute such as urea is very soluble in highly polar water, less soluble in fairly polar methanol, and practically insoluble in non-polar solvents such as benzene. In contrast, a non-polar or lipophilic solute such as naphthalene is insoluble in water, fairly soluble in methanol, and highly soluble in non-polar benzene.[20]

In even more simple terms a simple
The solubility is favored by
Chemists often exploit differences in solubilities to separate and purify compounds from reaction mixtures, using the technique of
Rate of dissolution
Dissolution is not an instantaneous process. The rate of solubilization (in kg/s) is related to the solubility product and the surface area of the material. The speed at which a solid dissolves may depend on its crystallinity or lack thereof in the case of
The rate of dissolution can be often expressed by the
where:
- = mass of dissolved material
- = time
- = surface area of the interface between the dissolving substance and the solvent
- = diffusion coefficient
- = thickness of the boundary layer of the solvent at the surface of the dissolving substance
- = mass concentration of the substance on the surface
- = mass concentration of the substance in the bulk of the solvent
For dissolution limited by diffusion (or mass transfer if mixing is present), is equal to the solubility of the substance. When the dissolution rate of a pure substance is normalized to the surface area of the solid (which usually changes with time during the dissolution process), then it is expressed in kg/m2s and referred to as "intrinsic dissolution rate". The intrinsic dissolution rate is defined by the United States Pharmacopeia.
Dissolution rates vary by orders of magnitude between different systems. Typically, very low dissolution rates parallel low solubilities, and substances with high solubilities exhibit high dissolution rates, as suggested by the Noyes-Whitney equation.
Theories of solubility
Solubility product
Other theories
The
The
The energy change associated with dissolving is usually given per mole of solute as the
Applications
Solubility is of fundamental importance in a large number of scientific disciplines and practical applications, ranging from ore processing and nuclear reprocessing to the use of medicines, and the transport of pollutants.
Solubility is often said to be one of the "characteristic properties of a substance", which means that solubility is commonly used to describe the substance, to indicate a substance's polarity, to help to distinguish it from other substances, and as a guide to applications of the substance. For example, indigo is described as "insoluble in water, alcohol, or ether but soluble in chloroform, nitrobenzene, or concentrated sulfuric acid".[22]
Solubility of a substance is useful when separating mixtures. For example, a mixture of salt (sodium chloride) and silica may be separated by dissolving the salt in water, and filtering off the undissolved silica. The synthesis of chemical compounds, by the milligram in a laboratory, or by the ton in industry, both make use of the relative solubilities of the desired product, as well as unreacted starting materials, byproducts, and side products to achieve separation.
Another example of this is the synthesis of
Differential solubility
In flowing systems, differences in solubility often determine the dissolution-precipitation driven transport of species. This happens when different parts of the system experience different conditions. Even slightly different conditions can result in significant effects, given sufficient time.
For example, relatively low solubility compounds are found to be soluble in more extreme environments, resulting in geochemical and geological effects of the activity of hydrothermal fluids in the Earth's crust. These are often the source of high quality economic mineral deposits and precious or semi-precious gems. In the same way, compounds with low solubility will dissolve over extended time (geological time), resulting in significant effects such as extensive cave systems or Karstic land surfaces.
Solubility of ionic compounds in water
Some ionic compounds (
- AgCl(s) ⇌ Ag+(aq) + Cl−(aq)
However, there is a limit to how much salt can be dissolved in a given volume of water. This concentration is the solubility and related to the
One can calculate the amount of AgCl that will dissolve in 1 liter of pure water as follows:
- Ksp = [Ag+] × [Cl−] / M2 (definition of solubility product; M = mol/L)
- Ksp = 1.8 × 10−10 (from a table of solubility products)
[Ag+] = [Cl−], in the absence of other silver or chloride salts, so
- [Ag+]2 = 1.8 × 10−10 M2
- [Ag+] = 1.34 × 10−5 mol/L
The result: 1 liter of water can dissolve 1.34 × 10−5 moles of AgCl at room temperature. Compared with other salts, AgCl is poorly soluble in water. For instance, table salt (NaCl) has a much higher Ksp = 36 and is, therefore, more soluble. The following table gives an overview of solubility rules for various ionic compounds.
| Soluble | Insoluble[23] |
|---|---|
| Group I and NH4+ compounds (except lithium phosphate) | Carbonates (except Group I, NH4+ and uranyl compounds) |
| Nitrates | Sulfites (except Group I and NH4+ compounds) |
| Acetates (ethanoates) (except Ag+ compounds) | Phosphates (except Group I and NH4+ compounds (excluding Li+)) |
| Chlorides (chlorates and perchlorates), bromides and iodides (except Ag+, Pb2+, Cu+ and Hg22+) | Hydroxides and oxides (except Group I, NH4+, Ba2+, Sr2+ and Tl+) |
| Sulfates (except Ag+, Pb2+, Ba2+, Sr2+ and Ca2+) | Sulfides (except Group I, Group II and NH4+ compounds) |
Solubility of organic compounds
The principle outlined above under
Solid solution
This term is often used in the field of
In microelectronic fabrication, solid solubility refers to the maximum concentration of impurities one can place into the substrate.
In solid compounds (as opposed to elements), the solubility of a solute element can also depend on the phases separating out in equilibrium. For example, amount of Sn soluble in the ZnSb phase can depend significantly on whether the phases separating out in equilibrium are (Zn4Sb3+Sn(L)) or (ZnSnSb2+Sn(L))[24]. Besides these, the ZnSb compound with Sn as a solute can separate out into other combinations of phases after the solubility limit is reached depending on the initial chemical composition during synthesis. Each combination produces a different solubility of Sn in ZnSb. Hence solubility studies in compounds, concluded upon the first instance of observing secondary phases separating out might underestimate solubility.[25] While the maximum number of phases separating out at once in equilibrium can be determined by the Gibb's phase rule, for chemical compounds there is no limit on the number of such phase separating combinations itself. Hence, establishing the "maximum solubility" in solid compounds experimentally can be difficult, requiring equilibration of many samples. If the dominant crystallographic defect (mostly interstitial or substitutional point defects) involved in the solid-solution can be chemically intuited beforehand, then using some simple thermodynamic guidelines can considerably reduce the number of samples required to establish maximum solubility. [26]
Incongruent dissolution
Many substances dissolve congruently (i.e. the composition of the solid and the dissolved solute stoichiometrically match). However, some substances may dissolve incongruently, whereby the composition of the solute in solution does not match that of the solid. This solubilization is accompanied by alteration of the "primary solid" and possibly formation of a secondary solid phase. However, in general, some primary solid also remains and a complex solubility equilibrium establishes. For example, dissolution of albite may result in formation of gibbsite.[27]
- NaAlSi3O8(s) + H+ + 7H2O ⇌ Na+ + Al(OH)3(s) + 3H4SiO4.
In this case, the solubility of albite is expected to depend on the solid-to-solvent ratio. This kind of solubility is of great importance in geology, where it results in formation of metamorphic rocks.
In principle, both congruent and incongruent dissolution can lead to the formation of secondary solid phases in equilibrium. So, in the field of
Solubility prediction
Solubility is a property of interest in many aspects of science, including but not limited to: environmental predictions, biochemistry, pharmacy, drug-design, agrochemical design, and protein ligand binding. Aqueous solubility is of fundamental interest owing to the vital biological and transportation functions played by water.[28][29][30] In addition, to this clear scientific interest in water solubility and solvent effects; accurate predictions of solubility are important industrially. The ability to accurately predict a molecule's solubility represents potentially large financial savings in many chemical product development processes, such as pharmaceuticals.[31] In the pharmaceutical industry, solubility predictions form part of the early stage lead optimisation process of drug candidates. Solubility remains a concern all the way to formulation.[31] A number of methods have been applied to such predictions including quantitative structure–activity relationships (QSAR), quantitative structure–property relationships (QSPR) and data mining. These models provide efficient predictions of solubility and represent the current standard. The draw back such models is that they can lack physical insight. A method founded in physical theory, capable of achieving similar levels of accuracy at an sensible cost, would be a powerful tool scientifically and industrially.[32][33][34][35]
Methods founded in physical theory tend to use thermodynamic cycles, a concept from classical thermodynamics. The two common thermodynamic cycles used involve either the calculation of the free energy of sublimation (solid to gas without going through a liquid state) and the free energy of solvating a gaseous molecule (gas to solution), or the free energy of fusion (solid to a molten phase) and the free energy of mixing (molten to solution). These two process are represented in the following diagrams.


These cycles have been used for attempts at first principles predictions (solving using the fundamental physical equations) using physically motivated
Well known fitted equations for solubility prediction are the general solubility equations. These equations stem from the work of Yalkowsky et al.[37][38] The original formula is given first, followed by a revised formula which takes a different assumption of complete miscibility in octanol.[38]
These equations are founded on the principles of the fusion cycle.
See also
- Apparent molar property – Difference in properties of one mole of substance in a mixture vs. an ideal solution
- Biopharmaceutics Classification System – System to differentiate drugs on the basis of their solubility and permeability
- Dühring's rule – Linear relationship between the temperatures at which two solutions exert the same vapour pressure
- Fajans–Paneth–Hahn Law – chemistry rule concerning co-precipitation and adsorption
- Flexible SPC water model– Aspect of computational chemistry
- Henry's law – Gas law regarding proportionality of dissolved gas
- Hot water extraction – method of carpet cleaning
- Hydrotrope – chemical substance
- Micellar solubilization – Process of incorporating the solubilizate into or onto micelles
- Raoult's law – Law of thermodynamics for vapour pressure of a mixture
- Rate of solution– Capacity of a substance to dissolve in a solvent in a homogeneous way
- Solubility equilibrium – Thermodynamic equilibrium between a solid and a solution of the same compound
- van 't Hoff equation – Relation between temperature and the equilibrium constant of a chemical reaction
Notes
- ^ The solvent polarity is defined as its solvation power according to Reichardt.
References
- ^
- ^ a b Clugston, M.; Fleming, R. (2000). Advanced Chemistry (1st ed.). Oxford: Oxford Publishing. p. 108.
- S2CID 110079029.
- ^ Abler (2021): "W/W (Weight/Weight)". Online page at Abler.com website. Accessed on 2021-11-26.
- .
- ^ "Pharmacopeia of the United States of America, 32nd revision, and the National Formulary, 27th edition," 2009, pp.1 to 12.
- ^ Rogers, Elizabeth; Stovall, Iris (2000). "Fundamentals of Chemistry: Solubility". Department of Chemistry. University of Wisconsin. Archived from the original on 13 April 2015. Retrieved 22 April 2015.
- ISBN 978-90-5410-723-1.
- ISBN 978-0-471-49708-0.
- ^ a b John W. Hill, Ralph H. Petrucci, General Chemistry, 2nd edition, Prentice Hall, 1999.
- ^ P. Cohen, ed. (1989). The ASME Handbook on Water Technology for Thermal Power Systems. The American Society of Mechanical Engineers. p. 442.
- ^ Handbook of Chemistry and Physics (27th ed.). Cleveland, Ohio: Chemical Rubber Publishing Co. 1943.
- ^ "What substances, such as cerium sulfate, have a lower solubility when they are heated?". Scientific American. Retrieved 28 May 2014.
- PMID 12189867.
- ^ E.M. Gutman (1994). Mechanochemistry of Solid Surfaces. World Scientific Publishing Co.
- S2CID 93098036.
- ISBN 978-0-669-19429-6.
- ^ Merck Index (7th ed.). Merck & Co. 1960.
- PMID 16920290.
- ^ El-Mansy, Mohamed & Yahia, Ibrahim S. & Alfaify, Sa. (2015). Conformational and Vibrational Properties of Indigo Dye: DFT Approach. Organo Opto-Electronics An International Journal. 3. 1-9.
- ISBN 978-0-471-12120-6.
- S2CID 234807088.
- PMID 26189943.
- S2CID 246516386.
- ISBN 978-90-5410-641-8.
- PMID 25660403.
- PMID 16092826.
- PMID 11849023.
- ^ PMID 25880026.
- hdl:10023/6534.
- ^ S2CID 26334468.
- ^ PMID 24564264.
- PMID 23795551.
- PMID 11604020.
- PMID 7400936.
- ^ PMID 11169540.























