Speciation
| Part of a series on |
| Evolutionary biology |
|---|
 |
Speciation is the evolutionary process by which populations evolve to become distinct species. The biologist Orator F. Cook coined the term in 1906 for cladogenesis, the splitting of lineages, as opposed to anagenesis, phyletic evolution within lineages.[1][2][3] Charles Darwin was the first to describe the role of natural selection in speciation in his 1859 book On the Origin of Species.[4] He also identified sexual selection as a likely mechanism, but found it problematic.
There are four geographic modes of speciation in nature, based on the extent to which speciating populations are isolated from one another: allopatric, peripatric, parapatric, and sympatric. Whether genetic drift is a minor or major contributor to speciation is the subject of much ongoing discussion.[5]
Rapid sympatric speciation can take place through
Historical background
In addressing the origin of species, there are two key issues:
- the evolutionary mechanisms of speciation
- how the separateness and individuality of species is maintained
Since Charles Darwin's time, efforts to understand the nature of species have primarily focused on the first aspect, and it is now widely agreed that the critical factor behind the origin of new species is reproductive isolation.[6]
Darwin's dilemma: why do species exist?
In On the Origin of Species (1859), Darwin interpreted biological evolution in terms of natural selection, but was perplexed by the clustering of organisms into species.[7] Chapter 6 of Darwin's book is entitled "Difficulties of the Theory". In discussing these "difficulties" he noted
Firstly, why, if species have descended from other species by insensibly fine gradations, do we not everywhere see innumerable transitional forms? Why is not all nature in confusion instead of the species being, as we see them, well defined?
— On the Origin of Species (1859), chapter 6[7]
This dilemma can be described as the absence or rarity of transitional varieties in habitat space.[8]
Another dilemma,[9] related to the first one, is the absence or rarity of transitional varieties in time. Darwin pointed out that by the theory of natural selection "innumerable transitional forms must have existed", and wondered "why do we not find them embedded in countless numbers in the crust of the earth". That clearly defined species actually do exist in nature in both space and time implies that some fundamental feature of natural selection operates to generate and maintain species.[7]
Effect of sexual reproduction on species formation
It has been argued that the resolution of Darwin's first dilemma lies in the fact that

Rarity brings with it other costs. Rare and unusual features are very seldom advantageous. In most instances, they indicate a (non-silent) mutation, which is almost certain to be deleterious. It therefore behooves sexual creatures to avoid mates sporting rare or unusual features (koinophilia).[16][17] Sexual populations therefore rapidly shed rare or peripheral phenotypic features, thus canalizing the entire external appearance, as illustrated in the accompanying image of the African pygmy kingfisher, Ispidina picta. This uniformity of all the adult members of a sexual species has stimulated the proliferation of field guides on birds, mammals, reptiles, insects, and many other taxa, in which a species can be described with a single illustration (or two, in the case of sexual dimorphism). Once a population has become as homogeneous in appearance as is typical of most species (and is illustrated in the photograph of the African pygmy kingfisher), its members will avoid mating with members of other populations that look different from themselves.[18] Thus, the avoidance of mates displaying rare and unusual phenotypic features inevitably leads to reproductive isolation, one of the hallmarks of speciation.[19][20][21][22]
In the contrasting case of organisms that
Modes

All forms of natural speciation have taken place over the course of evolution; however, debate persists as to the relative importance of each mechanism in driving biodiversity.[26]
One example of natural speciation is the diversity of the
Allopatric
During allopatric (from the ancient Greek allos, "other" + patrā, "fatherland") speciation, a population splits into two geographically isolated populations (for example, by
Peripatric
In peripatric speciation, a subform of allopatric speciation, new species are formed in isolated, smaller peripheral populations that are prevented from exchanging genes with the main population. It is related to the concept of a founder effect, since small populations often undergo bottlenecks. Genetic drift is often proposed to play a significant role in peripatric speciation.[29][30]
Case studies include Mayr's investigation of bird fauna;
Parapatric
In parapatric speciation, there is only partial separation of the zones of two diverging populations afforded by geography; individuals of each species may come in contact or cross habitats from time to time, but reduced fitness of the heterozygote leads to selection for behaviours or mechanisms that prevent their interbreeding. Parapatric speciation is modelled on continuous variation within a "single", connected habitat acting as a source of natural selection rather than the effects of isolation of habitats produced in peripatric and allopatric speciation.[33]
Parapatric speciation may be associated with differential landscape-dependent
Ecologists refer to[clarification needed] parapatric and peripatric speciation in terms of ecological niches. A niche must be available in order for a new species to be successful. Ring species such as Larus gulls have been claimed to illustrate speciation in progress, though the situation may be more complex.[36] The grass Anthoxanthum odoratum may be starting parapatric speciation in areas of mine contamination.[37]
Sympatric

Sympatric speciation is the formation of two or more descendant species from a single ancestral species all occupying the same geographic location.
Often-cited examples of sympatric speciation are found in insects that become dependent on different host plants in the same area.[38][39]

The best known example of sympatric speciation is that of the cichlids of East Africa inhabiting the Rift Valley lakes, particularly Lake Victoria, Lake Malawi and Lake Tanganyika. There are over 800 described species, and according to estimates, there could be well over 1,600 species in the region. Their evolution is cited as an example of both natural and sexual selection.[40][41] A 2008 study suggests that sympatric speciation has occurred in Tennessee cave salamanders.[42] Sympatric speciation driven by ecological factors may also account for the extraordinary diversity of crustaceans living in the depths of Siberia's Lake Baikal.[43]
Budding speciation has been proposed as a particular form of sympatric speciation, whereby small groups of individuals become progressively more isolated from the ancestral stock by breeding preferentially with one another. This type of speciation would be driven by the conjunction of various advantages of inbreeding such as the expression of advantageous recessive phenotypes, reducing the recombination load, and reducing the cost of sex.[44]
The hawthorn fly (
Methods of selection
Reinforcement

Reinforcement, also called the
One reasoning behind this is that if the parents of the hybrid offspring each have naturally selected traits for their own certain environments, the hybrid offspring will bear traits from both, therefore would not fit either ecological niche as well as either parent (ecological speciation). The low fitness of the hybrids would cause selection to favor assortative mating, which would control hybridization. This is sometimes called the Wallace effect after the evolutionary biologist Alfred Russel Wallace who suggested in the late 19th century that it might be an important factor in speciation.[48] Conversely, if the hybrid offspring are more fit than their ancestors, then the populations will merge back into the same species within the area they are in contact.[citation needed]
Another important theoretical mechanism is the arise of intrinsic genetic incompatibilities, addressed in the Bateson-Dobzhansky-Muller model.[49] Genes from allopatric populations will have different evolutionary backgrounds and are never tested together until hybridization at secondary contact, when negative epistatic interactions will be exposed. In other words, new alleles will emerge in a population and only pass through selection if they work well together with other genes in the same population, but it may not be compatible with genes in an allopatric population, be those other newly derived alleles or retained ancestral alleles. This is only revealed through new hybridization.[49][50] Such incompatibilities cause lower fitness in hybrids regardless of the ecological environment, and are thus intrinsic, although they can originate from the adaptation to different environments.[51] The accumulation of such incompatibilities increases faster and faster with time, creating a "snowball" effect.[52] There is a large amount of evidence supporting this theory, primarily from laboratory populations such as Drosophila and Mus, and some genes involved in incompatibilities have been identified.[50]
Reinforcement favoring reproductive isolation is required for both parapatric and sympatric speciation. Without reinforcement, the geographic area of contact between different forms of the same species, called their "hybrid zone", will not develop into a boundary between the different species. Hybrid zones are regions where diverged populations meet and interbreed. Hybrid offspring are common in these regions, which are usually created by diverged species coming into secondary contact. Without reinforcement, the two species would have uncontrollable inbreeding.[citation needed] Reinforcement may be induced in artificial selection experiments as described below.
Ecological
Ecological selection is "the interaction of individuals with their environment during resource acquisition".[53] Natural selection is inherently involved in the process of speciation, whereby, "under ecological speciation, populations in different environments, or populations exploiting different resources, experience contrasting natural selection pressures on the traits that directly or indirectly bring about the evolution of reproductive isolation".[54] Evidence for the role ecology plays in the process of speciation exists. Studies of stickleback populations support ecologically-linked speciation arising as a by-product,[55] alongside numerous studies of parallel speciation, where isolation evolves between independent populations of species adapting to contrasting environments than between independent populations adapting to similar environments.[56] Ecological speciation occurs with much of the evidence, "...accumulated from top-down studies of adaptation and reproductive isolation".[56]
Sexual selection
Sexual selection can drive speciation in a clade, independently of natural selection.[57] However the term "speciation", in this context, tends to be used in two different, but not mutually exclusive senses. The first and most commonly used sense refers to the "birth" of new species. That is, the splitting of an existing species into two separate species, or the budding off of a new species from a parent species, both driven by a biological "fashion fad" (a preference for a feature, or features, in one or both sexes, that do not necessarily have any adaptive qualities).[57][58][59][60] In the second sense, "speciation" refers to the wide-spread tendency of sexual creatures to be grouped into clearly defined species,[61][20] rather than forming a continuum of phenotypes both in time and space – which would be the more obvious or logical consequence of natural selection. This was indeed recognized by Darwin as problematic, and included in his On the Origin of Species (1859), under the heading "Difficulties with the Theory".[7] There are several suggestions as to how mate choice might play a significant role in resolving Darwin's dilemma.[20][10][16][17][18][62] If speciation takes place in the absence of natural selection, it might be referred to as nonecological speciation.[63][64]
Artificial speciation


New species have been created by animal husbandry, but the dates and methods of the initiation of such species are not clear. Often, the domestic counterpart can still interbreed and produce fertile offspring with its wild ancestor. This is the case with domestic cattle, which can be considered the same species as several varieties of wild ox, gaur, and yak; and with domestic sheep that can interbreed with the mouflon.[65][66]
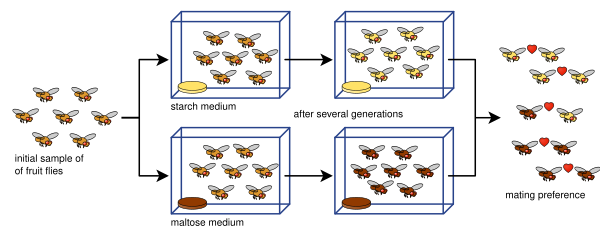
The best-documented creations of new species in the laboratory were performed in the late 1980s. William R. Rice and George W. Salt bred Drosophila melanogaster fruit flies using a maze with three different choices of habitat such as light/dark and wet/dry. Each generation was placed into the maze, and the groups of flies that came out of two of the eight exits were set apart to breed with each other in their respective groups. After thirty-five generations, the two groups and their offspring were isolated reproductively because of their strong habitat preferences: they mated only within the areas they preferred, and so did not mate with flies that preferred the other areas.[67] The history of such attempts is described by Rice and Elen E. Hostert (1993).[68][69] Diane Dodd used a laboratory experiment to show how reproductive isolation can develop in Drosophila pseudoobscura fruit flies after several generations by placing them in different media, starch- and maltose-based media.[70]
Dodd's experiment has been replicated many times, including with other kinds of fruit flies and foods.[71] Such rapid evolution of reproductive isolation may sometimes be a relic of infection by Wolbachia bacteria.[72]
An alternative explanation is that these observations are consistent with sexually-reproducing animals being inherently reluctant to mate with individuals whose appearance or behavior is different from the norm. The risk that such deviations are due to heritable maladaptations is high. Thus, if an animal, unable to predict natural selection's future direction, is conditioned to produce the fittest offspring possible, it will avoid mates with unusual habits or features.[73][74][16][17][18] Sexual creatures then inevitably group themselves into reproductively isolated species.[17]
Genetics
Few speciation genes have been found. They usually involve the reinforcement process of late stages of speciation. In 2008, a speciation gene causing reproductive isolation was reported.[75] It causes hybrid sterility between related subspecies. The order of speciation of three groups from a common ancestor may be unclear or unknown; a collection of three such species is referred to as a "trichotomy".[citation needed]
Speciation via polyploidy
It has been suggested that many of the existing plant and most animal species have undergone an event of polyploidization in their evolutionary history.[77][78] Reproduction of successful polyploid species is sometimes asexual, by parthenogenesis or apomixis, as for unknown reasons many asexual organisms are polyploid. Rare instances of polyploid mammals are known, but most often result in prenatal death. [citation needed]
Hybrid speciation
Hybridization between two different species sometimes leads to a distinct phenotype. This phenotype can also be fitter than the parental lineage and as such natural selection may then favor these individuals. Eventually, if reproductive isolation is achieved, it may lead to a separate species. However, reproductive isolation between hybrids and their parents is particularly difficult to achieve and thus hybrid speciation is considered an extremely rare event. The Mariana mallard is thought to have arisen from hybrid speciation.[citation needed]
Hybridization is an important means of speciation in plants, since
Hybridization without change in chromosome number is called homoploid hybrid speciation. It is considered very rare but has been shown in
Gene transposition
Theodosius Dobzhansky, who studied fruit flies in the early days of genetic research in 1930s, speculated that parts of chromosomes that switch from one location to another might cause a species to split into two different species. He mapped out how it might be possible for sections of chromosomes to relocate themselves in a genome. Those mobile sections can cause sterility in inter-species hybrids, which can act as a speciation pressure. In theory, his idea was sound, but scientists long debated whether it actually happened in nature. Eventually a competing theory involving the gradual accumulation of mutations was shown to occur in nature so often that geneticists largely dismissed the moving gene hypothesis.[83] However, 2006 research shows that jumping of a gene from one chromosome to another can contribute to the birth of new species.[84] This validates the reproductive isolation mechanism, a key component of speciation.[85]
Rates
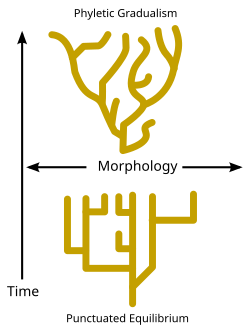
There is debate as to the rate at which speciation events occur over geologic time. While some evolutionary biologists claim that speciation events have remained relatively constant and gradual over time (known as "Phyletic gradualism" – see diagram), some palaeontologists such as Niles Eldredge and Stephen Jay Gould[86] have argued that species usually remain unchanged over long stretches of time, and that speciation occurs only over relatively brief intervals, a view known as punctuated equilibrium. (See diagram, and Darwin's dilemma.) [citation needed]
Punctuated evolution
Evolution can be extremely rapid, as shown in the creation of domesticated animals and plants in a very short geological space of time, spanning only a few tens of thousands of years. Maize (Zea mays), for instance, was created in Mexico in only a few thousand years, starting about 7,000 to 12,000 years ago.[87] This raises the question of why the long term rate of evolution is far slower than is theoretically possible.[88][89][90][91]
Evolution is imposed on species or groups. It is not planned or striven for in some
as selectively disadvantageous to the individual.The resolution to Darwin's second dilemma might thus come about as follows:
If sexual individuals are disadvantaged by passing mutations on to their offspring, they will avoid mutant mates with strange or unusual characteristics.[74][16][17][62] Mutations that affect the external appearance of their carriers will then rarely be passed on to the next and subsequent generations. They would therefore seldom be tested by natural selection. Evolution is, therefore, effectively halted or slowed down considerably. The only mutations that can accumulate in a population, on this punctuated equilibrium view, are ones that have no noticeable effect on the outward appearance and functionality of their bearers (i.e., they are "silent" or "neutral mutations", which can be, and are, used to trace the relatedness and age of populations and species.[16][99])
This argument implies that evolution can only occur if mutant mates cannot be avoided, as a result of a severe scarcity of potential mates. This is most likely to occur in small, isolated communities. These occur most commonly on small islands, in remote valleys, lakes, river systems, or caves,[100] or during the aftermath of a mass extinction.[99] Under these circumstances, not only is the choice of mates severely restricted but population bottlenecks, founder effects, genetic drift and inbreeding cause rapid, random changes in the isolated population's genetic composition.[100] Furthermore, hybridization with a related species trapped in the same isolate might introduce additional genetic changes. If an isolated population such as this survives its genetic upheavals, and subsequently expands into an unoccupied niche, or into a niche in which it has an advantage over its competitors, a new species, or subspecies, will have come into being. In geological terms, this will be an abrupt event. A resumption of avoiding mutant mates will thereafter result, once again, in evolutionary stagnation.[86][89]
In apparent confirmation of this punctuated equilibrium view of evolution, the fossil record of an evolutionary progression typically consists of species that suddenly appear, and ultimately disappear, hundreds of thousands or millions of years later, without any change in external appearance.[86][99][101] Graphically, these fossil species are represented by lines parallel with the time axis, whose lengths depict how long each of them existed. The fact that the lines remain parallel with the time axis illustrates the unchanging appearance of each of the fossil species depicted on the graph. During each species' existence new species appear at random intervals, each also lasting many hundreds of thousands of years before disappearing without a change in appearance. The exact relatedness of these concurrent species is generally impossible to determine. This is illustrated in the diagram depicting the distribution of hominin species through time since the hominins separated from the line that led to the evolution of their closest living primate relatives, the chimpanzees.[101]
For similar evolutionary time lines see, for instance, the paleontological list of African dinosaurs, Asian dinosaurs, the Lampriformes and Amiiformes. [citation needed]
See also
References
- ^ Berlocher 1998, p. 3
- PMID 17789700.
- S2CID 84565616.
- PMID 19528641.
- PMID 11058173.
- ^ Mayr 1982, p. 273
- ^ a b c d Darwin 1859
- ISBN 978-0-226-74858-0.
One of his greatest anxieties was that the "incompleteness" of the fossil record would be used to criticize his theory: that the apparent "gaps" in fossil succession could be cited as negative evidence, at the very least, for his proposal that all organisms have descended by minute and gradual modifications from a common ancestor.
- S2CID 45302603.
The near-simultaneous appearance of most modern animal body plans in the Cambrian explosion suggests a brief interval of rapid phenotypic and genetic evolution, which Darwin believed were too fast to be explained by natural selection.
- ^ PMID 4094459.
- ^ .
- ^ Bernstein & Bernstein 1991
- ^ a b Michod 1995
- ^ Michod 1999
- ^ Hockey, Dean & Ryan 2005, pp. 176, 193
- ^ PMID 2200930.
- ^ ISSN 0022-5193.
- ^ S2CID 14431499.
The Phenotypic Differences between Carrion and Hooded Crows across the Hybridization Zone in Europe are Unlikely to be due to Assortative Mating.
— Commentary by Mazhuvancherry K. Unnikrishnan and H. S. Akhila - ^ ISBN 978-1-4051-0345-9.
- ^ a b c Maynard Smith 1989, pp. 275–280
- ^ Mayr 1988
- ^ Williams 1992, p. 118
- S2CID 3901837. Archived from the original(PDF) on 2019-03-05.
- ^ Clapham, Tutin & Warburg 1952
- ^ Grant 1971
- S2CID 3555049.
- PMID 19186749.
- ^ Sulloway, Frank J. (September 30, 1982). "The Beagle collections of Darwin's finches (Geospizinae)". Bulletin of the British Museum (Natural History), Zoology. 43 (2): 49–58.
- ^ Coyne & Orr 2004, p. 105.
- PMID 26126573.
- ^ Mayr 1992, pp. 21–53
- OCLC 47011551.
- ^ "Speciation: The Origin of New Species | Learn Science at Scitable". Nature. Retrieved 2020-02-16.
- ^ Endler 1977
- .
- PMID 15255043.
- ^ "Parapatric speciation". University of California Berkeley. Retrieved 3 April 2017.
- PMID 15851672.
- S2CID 9677456.
- PMID 21665847.
- PMID 22201168.
- S2CID 20761880.
- PMID 21238028.
- PMID 22152499.
- PMID 12663534. Retrieved 2015-09-07.
- JSTOR 2413033.
- ISBN 978-0470016176.
- S2CID 13300641. Archived from the original(PDF) on 2007-06-05. Retrieved 2015-09-07.
- ^ PMID 8978022.
- ^ S2CID 5592958.
- S2CID 41904934.
- PMID 7789779.
- S2CID 9845298
- S2CID 2744267
- ^ S2CID 307207
- ^ PMID 11403869.
- .
- ^ Darwin 1859, p. 89, "IV. Natural Selection".
- ^ Eberhard, W. G. (1985). Sexual Selection and Animal Genitalia. Harvard University Press, Cambridge, Massachusetts
- ^ Gould 1980, pp. 204–213, "A Quahog is a Quahog".
- ^ a b Miller 2013, pp. 177, 395–396
- PMID 19409647.
- S2CID 73494468.
- ^ Nowak 1999
- PMID 12028771.
- S2CID 84876223.
- PMID 28568007.
- S2CID 198158082.
- PMID 28564510.
- S2CID 16516804.
- S2CID 198153238.
- ^ Symons 1979
- ^ S2CID 18557871.
- PMID 19074311.
- S2CID 31637733. Archived from the original(PDF) on 2020-06-08.
- PMID 11092833.
- ^ S2CID 3329282.
- S2CID 14856314.
- PMID 18006297.
- PMID 10860970.
- S2CID 2457445.
- ^ Sherwood, Jonathan (September 8, 2006). "Genetic Surprise Confirms Neglected 70-Year-Old Evolutionary Theory" (Press release). University of Rochester. Retrieved 2015-09-10.
- S2CID 23462115.
- ^ Minkel, J. R. (September 8, 2006). "Wandering Fly Gene Supports New Model of Speciation". Scientific American. Retrieved 2015-09-11.
- ^ S2CID 83492071. Archived from the original(PDF) on 2014-06-24. Retrieved 2015-09-15.
- ^ Laws 2010, pp. 210–215
- ^ Williams 1992, chpt. 9
- ^ a b Eldredge & Gould 1972, chpt. 5
- ^ Mayr 1954, pp. 157–180
- ^ Maynard Smith 1989, p. 281
- ^ Gould 1980, pt. 4, chpt. 18
- ^ Williams 1974
- S2CID 4177102.
- chpt. 4
- S2CID 143378724. Archived from the original on 2006-09-15. Retrieved 2015-09-15. "Remarks on an earlier article by [Elliot] Sober [sic] and David Sloan Wilson, who made a more extended argument in their recent book Unto Others : The Evolution and Psychology of Unselfish Behavior"
- S2CID 146359497. Archived from the originalon 2007-12-27. "Commentary on Wilson & Sober: Group Selection."
- ^ Pinker, Steven (June 18, 2012). "The False Allure of Group Selection". edge.org. Edge Foundation, Inc. Retrieved 2015-09-15.
- ^ a b c Campbell 1990, pp. 450–451, 487–490, 499–501
- ^ a b Ayala 1982, pp. 73–83, 182–190, 198–215
- ^ a b McCarthy & Rubidge 2005
Bibliography
- OCLC 8034790.
- Berlocher, Stewart H. (1998). "Origins: A Brief History of Research on Speciation". In Howard, Daniel J.; Berlocher, Stewart H. (eds.). Endless Forms: Species and Speciation. New York: OCLC 37545522.
- Bernstein, Carol; Bernstein, Harris (1991). Aging, Sex, and DNA Repair. San Diego, CA: OCLC 22542921.
- OCLC 20352649.
- OCLC 1084058.
- OCLC 55078441.
- OCLC 741260650. The book is available from John van Wyhe, ed. (2002), The Complete Work of Charles Darwin Online, retrieved 2015-09-12.
- OCLC 31376584.
- OCLC 572084. Reprinted in Eldredge 1985, pp. 193–223
- Eldredge, Niles (1985). Time Frames: The Rethinking of Darwinian Evolution and the Theory of Punctuated Equilibria. New York: OCLC 11443805.
- PMID 409931.
- OCLC 6331415. 1982 editionvia Archive.org.
- Grant, Verne (1971). Plant Speciation. New York: OCLC 139834.
- Hockey, Phil A. R.; Dean, W. Richard J.; Ryan, Peter G., eds. (2005). Roberts Birds of Southern Africa (7th ed.). Cape Town, South Africa: Trustees of the J. Voelcker Bird Book Fund. OCLC 65978899.
- Laws, Bill (2010). Fifty Plants that Changed the Course of History. Buffalo, NY: Firefly Books. OCLC 711609823.
- OCLC 18069049.
- OCLC 974739.
- Mayr, Ernst (1982). OCLC 7875904.
- Mayr, Ernst (1988). OCLC 17108004.
- Mayr, Ernst (1992). "Speciational Evolution or Punctuated Equilibrium". In Somit, Albert; Peterson, Steven A. (eds.). Dynamics of Evolution: The Punctuated Equilibrium Debate in the Natural and Social Sciences. Ithaca, NY: OCLC 24374091.
- McCarthy, Terence; Rubidge, Bruce (2005). The Story of Earth & Life: A Southern African Perspective on a 4.6-Billion-Year Journey. Cape Town, South Africa: Struik Publishers. OCLC 62098231.
- Michod, Richard E. (1995). Eros and Evolution: A Natural Philosophy of Sex. Helix Books. Reading, MA: OCLC 30625193.
- Michod, Richard E. (1999). Darwinian Dynamics: Evolutionary Transitions in Fitness and Individuality. Princeton, NJ: Princeton University Press. OCLC 38948118.
- Miller, William B. Jr. (2013). The Microcosm Within: Evolution and Extinction in the Hologenome. Boca Raton, FL: OCLC 859168474.
- Nowak, Ronald M. (1999). Walker's Mammals of the World (6th ed.). Baltimore, MD: OCLC 39045218.
- OCLC 4494283.
- OCLC 8500898.
- Williams, George C. (1992). Natural Selection: Domains, Levels, and Challenges. Oxford Series in Ecology and Evolution. New York: Oxford University Press. OCLC 228136567.
Further reading
External links
- Boxhorn, Joseph (September 1, 1995). "Observed Instances of Speciation". TalkOrigins Archive. Houston, TX: The TalkOrigins Foundation, Inc.
- Hawks, John D. (February 9, 2005). "Speciation". John Hawks Weblog.
- "Speciation". University of California, Berkeley. 13 March 2021.