Spectroelectrochemistry
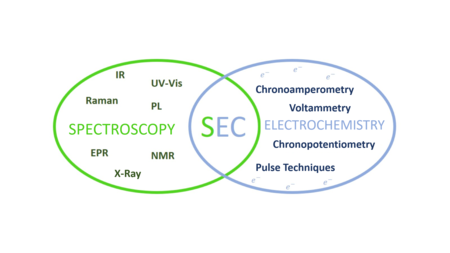
Spectroelectrochemistry (SEC) is a set of multi-response analytical techniques in which complementary chemical information (
The main objective of spectroelectrochemical experiments is to obtain simultaneous, time-resolved and in-situ
The techniques on which the spectroelectrochemistry is based are:
- Electrochemistry, which studies the interaction between electrical energy and chemical changes. This technique allows us to analyse reactions that involve electron transfer processes (redox reactions).[7]
- Spectroscopy, which studies the interaction between electromagnetic radiation and matter (absorption, dispersion or emission).[8][9]
Spectroelectrochemistry provides molecular,
Classification of spectroelectrochemical techniques
There are different spectroelectrochemical techniques based on the combination of spectroscopic and electrochemical techniques. Regarding electrochemistry, the most common techniques used are:
- Chronoamperometry, which measures current intensity as a function of time by applying a constant difference of potential to the working electrode.
- Chronopotentiometry, which measures the difference of potential as a function of time by applying a constant current.
- Voltammetry, which measures the change of current as a function of the linear change of the working electrode potential.
- Pulse techniques, which measure the change of current as a function of difference of potential, applying pulse potential functions to the working electrode.
The general classification of the spectroelectrochemical techniques is based on the spectroscopic technique chosen.
Ultraviolet-visible absorption spectroelectrochemistry
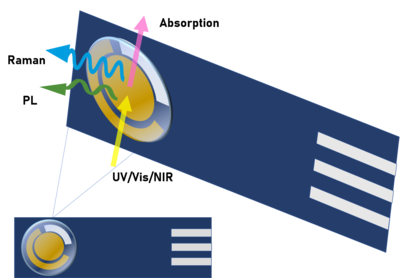
Ultraviolet-visible(UV-Vis) absorption spectroelectrochemistry is a technique that studies the absorption of electromagnetic radiation in the UV-Vis regions of the spectrum, providing molecular information related to the electronic levels of molecules.[10] It provides qualitative as well as quantitative information. UV-Vis spectroelectrochemistry helps to characterize compounds and materials, determines concentrations and different parameters such as absorptivity coefficients, diffusion coefficients, formal potentials or electron transfer rates.[11][12]
Photoluminescence spectroelectrochemistry
Infrared spectroelectrochemistry
Infrared spectroscopy is based on the fact that molecules absorb electromagnetic radiation at characteristic frequencies related to their vibrational structure. Infrared (IR) spectroelectrochemistry is a technique that allows the characterization of molecules based on the resistance, stiffness and number of bonds present. It also detects the presence of compounds, determines the concentration of species during a reaction, the structure of compounds, the properties of the chemical bonds, etc.[10]
Raman spectroelectrochemistry
Raman spectroelectrochemistry is based on the
X-ray spectroelectrochemistry
X-ray spectroelectrochemistry is a technique that studies the interaction of high-energy radiation with matter during an electrode process.
Nuclear magnetic resonance spectroelectrochemistry
Electron paramagnetic resonance spectroelectrochemistry
Advantages and applications
The versatility of spectroelectrochemistry is increasing due to the possibility of using several
The main advantages of spectroelectrochemical techniques are:
- The simultaneous information is obtained by different techniques in a single experiment, increasing the selectivity and the sensitivity.
- Both qualitative and quantitative information can be obtained.
- The possibility of working with a small amount of sample, saving it for future analysis.[1]
Due to the high versatility of the technique, the field of applications is considerably wide.[1][2][3][4][5][16]
- Study of reaction intermediates.
- Characterization of organic and inorganic materials, that allow to understand the structure and properties of the material when is perturbed by a signal (electric, light, etc.).
- Development of spectroelectrochemical sensors, which are based on optical and electrical responses, capable of providing two independent signals about the same sample and offering a self-validated determination.
- Study of spectroscopicproperties and their photochemical and photophysical behaviour.
- Study different processes and molecules in biotechnology, biochemistry or medicine.
- Determine specific properties and characteristics of new materials in fields such as energy or nanotechnology.
References
- ^ S2CID 133304199.
- ^ ISBN 9780444519580.
- ^ PMID 20449056.
- ^ PMID 31998878.
- ^ PMID 29379916.
- .
- .
- ^ ISBN 978-8188449156.
- ^ ISBN 9788578110796.
- ^ ISBN 978-1429209656.
- S2CID 155767924.
- ^ PMID 27427898.
- PMID 19645423.
- PMID 20931995.
- PMID 7752937.
- ISBN 9780128032244.
