Spermatozoon
| Spermatozoon | |
|---|---|
ovum coat to fertilize it. | |
 Diagram of a human spermatozoon | |
| Details | |
| Identifiers | |
| Latin | spermatozoon |
| MeSH | D013094 |
| Anatomical terms of microanatomy] | |
A spermatozoon (
Sperm cells contribute approximately half of the nuclear
Mammalian spermatozoon structure, function, and size
Humans
The human
A human sperm cell consists of a flat, disc shaped head 5.1 µm by 3.1 µm and a tail known as a flagellum 50 µm long.[4] The flagellum propels the sperm cell (at about 1–3 mm/minute in humans) by whipping in an elliptical cone.[5] Sperm have an olfactory guidance mechanism, and after reaching the fallopian tubes, must undergo a period of capacitation before penetration of the ovum.[6]
Head: It has a compact nucleus with only chromatic substance and is surrounded by only a thin rim of cytoplasm. Above the nucleus lies a cap-like structure called the acrosome, formed by modification of the Golgi body, which secretes the enzyme spermlysin (hyaluronidase, corona-penetrating enzyme, zona lysin, or acrosin), that is necessary for fertilization. As the spermatozoon approaches the egg, it undergoes the acrosome reaction in which the membrane surrounding the acrosome fuses with the plasma membrane of the sperm's head, exposing the contents of the acrosome.[7]
Neck: It is the smallest part (.03 µm), and has a proximal centriole parallel to the base of the nucleus and distal centriole perpendicular to the previous one. The proximal centriole is present also in the mature spermatozoon; the distal centriole disappears after axoneme assembly. The proximal centriole enters into the egg during fertilisation and starts the first cleavage division of the egg, which has no centriole. The distal centriole gives rise to the axial filament which forms the tail and has a (9+2) arrangement. A transitory membrane called the Manchette lies in the middle piece.
Middle piece: It has 10–14 spirals of mitochondria surrounding the axial filament in the cytoplasm. It provides motility, and hence is called the powerhouse of the sperm. It also has a ring centriole (annulus) that form a diffusion barrier between the middle piece and the principal piece and serve as a stabilizing structure for tail rigidity.[8]
Tail: The flagellum is the longest part (50 µm), having an axial filament surrounded by cytoplasm and plasma membrane, but at the posterior end the axial filament is naked. The flagellum gives movement to the cell.
The spermatozoon is characterized by a minimum of cytoplasm and the most densely packed DNA known in eukaryotes. Compared to mitotic chromosomes in somatic cells, sperm DNA is at least sixfold more highly condensed.[9]
The specimen contributes with DNA/chromatin, a centriole, and perhaps also an oocyte-activating factor (OAF).[10] It may also contribute with paternal messenger RNA (mRNA), also contributing to embryonic development.[10]
-
Electron micrograph of human spermatozoa magnified 3140 times.
-
Sperm cells in the urine sample of a 45-year-old male patient who is being followed with the diagnosis ofbenign prostate hyperplasia.
-
Dimensions of the human sperm head measured from a 39-year-old healthy subject.
The human spermatozoon contains at least 7500 different proteins.[11]
Human sperm genetics has been associated with human evolution, per a 2020 study.[12][13][14]
DNA damage and repair
DNA damages present in spermatozoa in the period after meiosis but before fertilization may be repaired in the fertilized egg, but if not repaired, can have serious deleterious effects on fertility and the developing embryo. Human spermatozoa are particularly vulnerable to free radical attack and the generation of oxidative DNA damage.[15][16] (see e.g. 8-Oxo-2'-deoxyguanosine)
Exposure of males to certain lifestyle, environmental or occupational hazards may increase the risk of aneuploid spermatozoa.[17] In particular, risk of aneuploidy is increased by tobacco smoking,[18][19] and occupational exposure to benzene,[20] insecticides,[21][22] and perfluorinated compounds.[23] Increased aneuploidy of spermatozoa often occurs in association with increased DNA damage. DNA fragmentation and increased in situ DNA susceptibility to denaturation, the features similar to these seen during apoptosis of somatic cells, characterize abnormal spermatozoa in cases of male infertility.[24][25]
Avoidance of immune system response
The
Spermatozoa in other organisms

Animals
Fertilization relies on spermatozoa for most sexually reproductive animals.
Some species of fruit fly produce the largest known spermatozoon found in nature.[27][28] Drosophila melanogaster produces sperm that can be up to 1.8 mm,[29] while its relative Drosophila bifurca produces the largest known spermatozoon, measuring over 58 mm in length.[27] In Drosophila melanogaster, the entire sperm, tail included, gets incorporated into the oocyte cytoplasm, however, for Drosophila bifurca only a small portion of the tail enters the oocyte.[30]
The wood mouse
The postmeiotic phase of mouse spermatogenesis is very sensitive to environmental genotoxic agents, because as male germ cells form mature spermatozoa they progressively lose the ability to repair DNA damage.[32] Irradiation of male mice during late spermatogenesis can induce damage that persists for at least 7 days in the fertilizing spermatozoa, and disruption of maternal DNA double-strand break repair pathways increases spermatozoa-derived chromosomal aberrations.[33] Treatment of male mice with melphalan, a bifunctional alkylating agent frequently employed in chemotherapy, induces DNA lesions during meiosis that may persist in an unrepaired state as germ cells progress through DNA repair-competent phases of spermatogenic development.[34] Such unrepaired DNA damages in spermatozoa, after fertilization, can lead to offspring with various abnormalities.
Sea urchins such as Arbacia punctulata are ideal organisms to use in sperm research, they spawn large numbers of sperm into the sea, making them well-suited as model organisms for experiments.[35]
The spermatozoa of
Plants, algae and fungi
The
Some algae and fungi produce non-motile sperm cells, called spermatia. In higher plants and some algae and fungi, fertilization involves the migration of the sperm nucleus through a fertilization tube (e.g. pollen tube in higher plants) to reach the egg cell.[citation needed]
Spermatozoa production in mammals
Spermatozoa are produced in the
In assisted reproductive technology, normozoospermia is referred to a total amount of >39 mill ejaculated, >32% with progressive motility and >4% normal morphology. Also, a normal ejaculation in humans must have a volume over 1.5 ml, being an excessive volume 6 ml per ejaculation (hyperspermia). An insufficient volume is called hypospermia. These problems are related to several complications in spermatozoa production, for example:
- Hyperspermia: usually provoked because of prostate inflammation.
- Hypospermia: an incomplete ejaculation, usually referred to an androgen deficit (hypoandrogenism) or obstruction in some part of the ejaculatory duct. In laboratory conditions, is also due to a partial loss of the sample.
- Aspermia: there is no ejaculation. It could happen due to retrograde ejaculation, anatomical or neurological diseases or anti-hypertensive drugs.
Spermatozoa activation
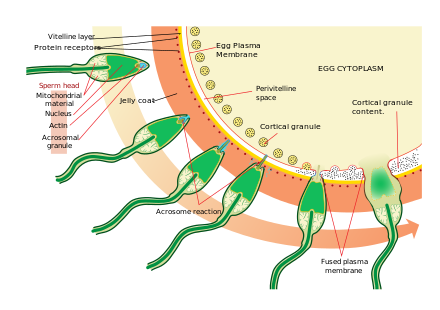
Approaching the egg cell is a rather complex, multistep process of chemotaxis guided by different chemical substances/stimuli on individual levels of phylogeny. One of the most significant, common signaling characters of the event is that a prototype of professional chemotaxis receptors, formyl peptide receptor (60,000 receptor/cell) as well as the activator ability of its ligand formyl Met-Leu-Phe have been demonstrated in the surface membrane even in the case of human sperms.[37] Mammalian sperm cells become even more active when they approach an egg cell in a process called sperm activation. Sperm activation has been shown to be caused by
The initial change is called "hyperactivation", which causes a change in spermatozoa motility. They swim faster and their tail movements become more forceful and erratic.
A recent discovery links hyperactivation to a sudden influx of calcium ion into the tails. The whip-like tail (flagellum) of the sperm is studded with
The second process in sperm activation is the acrosome reaction. This involves releasing the contents of the acrosome, which disperse, and the exposure of enzymes attached to the inner acrosomal membrane of the sperm. This occurs after the sperm first meets the egg. This lock-and-key type mechanism is species-specific and prevents the sperm and egg of different species from fusing. There is some evidence that this binding is what triggers the acrosome to release the enzymes that allow the sperm to fuse with the egg.
ZP3, one of the proteins that make up the zona pellucida, then binds to a partner molecule on the sperm. Enzymes on the inner acrosomal membrane digest the zona pellucida. After the sperm penetrates the zona pellucida, part of the sperm's cell membrane then
Upon penetration, the oocyte is said to have become
Artificial storage
Spermatozoa can be stored in diluents such as the Illini Variable Temperature (IVT) diluent, which have been reported to be able to preserve high fertility of spermatozoa for over seven days.[38] The IVT diluent is composed of several salts, sugars and antibacterial agents and gassed with CO2.[38]
Semen cryopreservation can be used for far longer storage durations. For human spermatozoa, the longest reported successful storage with this method is 21 years.[39]
MMP and capacitation
The capacitation is the final phase of spermatozoa development, when they acquire the capability to fertilize the oocyte. In vivo, it happens during ejaculation, when spermatozoa leave the vagina and come in the superior female reproductive tract. In vitro, it happens when the spermatozoa is washed and purified. Almost 30-40% of infertility is due to male factor, so several strategies have been created in order to recover the functional spermatozoa. The MMP (Million Motile Progressive cells per milliliter) measure is synonymous with capacitation, and is very useful parameter to decide, along with a spermiogram, the kind of treatment needed. it represents the ratio between the % of progressive motile sperm obtained in capacitated and the % of progressive motile sperm obtained in ejaculated. It is based on the recovery percentage. Depending on the percentage, we will decide the quality of the motile spermatozoa recovery: 15 to 25 million sperm/ml is considered optimal, between 5 and 15 million is considered enough and less than 5 million is considered sub-optimal or not sufficient. Regarding the values that we have obtained, along with the spermiogram results, different techniques will be displayed.
For example, if more than 1.0×106 progressive motile sperm per milliliter are found, it will be recommended to have sexual intercourse, and if that fails, the next step will be
With less than 1.0×106 progressive motile sperm per milliliter, we will perform intracytoplasmic sperm injection. In case of azoospermia (no spermatozoa in the ejaculate), we will do a testicular biopsy in order to check if there are spermatozoa in the testes or if no spermatozoa are being produced.
History
- In 1677 microbiologist Antonie van Leeuwenhoek discovered spermatozoa.
- In 1841 the Swiss anatomist Albert von Kölliker wrote about spermatozoon in his work Untersuchungen über die Bedeutung der Samenfäden (Studies on the importance of spermatozoa).
See also
- Aneuploidy
- Non-disjunction
References
- ^ "spermatozoon". Dictionary.com Unabridged (Online). n.d.
- ^ "Timeline: Assisted reproduction and birth control". CBC News. Retrieved 2006-04-06.
- ^ Scheinfeld, Amram (1939). You and Heredity. New York: Frederick A. Stokes Company. p. 39.
- S2CID 3942426.
- .
- S2CID 32567894.
- .
- ^ "sperm annulus | SGD". www.yeastgenome.org. Retrieved 2019-02-22.
- PMID 2043729.
- ^ PMID 19631936.
- PMID 24082039.
- PMID 31978344.
- ^ "Scanning system in sperm may control rate of human evolution".
- ^ "Genetic Scanning System in Sperm May Control Rate of Human Evolution".
- PMID 26178844.
- ^ Lozano, G.M.; Bejarano, I.; Espino, J.; González, D.; Ortiz, A.; García, J.F.; Rodríguez, A.B.; Pariente, J.A. (2009). "Density gradient capacitation is the most suitable method to improve fertilization and to reduce DNA fragmentation positive spermatozoa of infertile men". Anatolian Journal of Obstetrics & Gynecology. 3 (1): 1–7.
- PMID 23720770.
- S2CID 35230655.
- PMID 9797104.
- PMID 20418200.
- S2CID 36073841.
- PMID 15615886.
- S2CID 13484513.
- PMID 8391465.
- PMID 7444440.
- ^ "Sperm clue to 'disease immunity'". BBC News. 2007-12-17.
- ^ S2CID 4368953.
- PMID 7937755.
- ^ Cooper, K.W. (1950). Demerec, M. (ed.). Biology of Drosophila. New York: Wiley. pp. 1–61.
- S2CID 4368953.
- S2CID 4413444.
- S2CID 1316244.
- PMID 17978187.
- PMID 25567288.
- S2CID 13452188.
- ISBN 978-1-4863-0752-4.
- PMID 3018025.
- ^ PMID 9627729.
- ^ Planer NEWS and Press Releases > Child born after 21 year semen storage using Planer controlled rate freezer 14/10/2004



