Sphingolipid
This article needs additional citations for verification. (January 2023) |
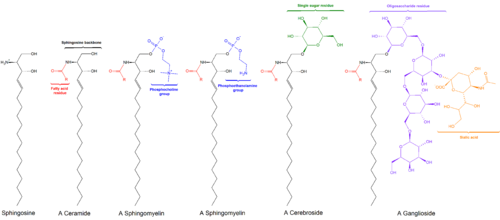
Sphingolipids are a class of
Structure
The long-chain bases, sometimes simply known as sphingoid bases, are the first non-transient products of de novo sphingolipid synthesis in both yeast and mammals. These compounds, specifically known as phytosphingosine and dihydrosphingosine (also known as sphinganine,[2] although this term is less common), are mainly C18 compounds, with somewhat lower levels of C20 bases.[3] Ceramides and glycosphingolipids are N-acyl derivatives of these compounds.[4]
The sphingosine backbone is O-linked to a (usually) charged head group such as ethanolamine, serine, or choline.[citation needed]
The backbone is also amide-linked to an acyl group, such as a fatty acid.[citation needed]
Types
Simple sphingolipids, which include the sphingoid bases and ceramides, make up the early products of the sphingolipid synthetic pathways.
- Sphingoid bases are the fundamental building blocks of all sphingolipids. The main mammalian sphingoid bases are dihydrosphingosine and sphingosine, while dihydrosphingosine and phytosphingosine are the principal sphingoid bases in yeast.[5][6] Sphingosine, dihydrosphingosine, and phytosphingosine may be phosphorylated.
- Ceramides, as a general class, are N-acylated sphingoid bases lacking additional head groups.
- Dihydroceramide is produced by N-acylation of dihydrosphingosine. Dihydroceramide is found in both yeast and mammalian systems.
- Ceramide is produced in mammalian systems by desaturation of dihydroceramide by dihydroceramide desaturase 1 (DES1). This highly bioactive molecule may also be phosphorylated to form ceramide-1-phosphate.
- Phytoceramideis produced in yeast by hydroxylation of dihydroceramide at C-4.
Complex sphingolipids may be formed by addition of head groups to ceramide or phytoceramide:
- phosphoethanolamine molecule with an ester linkageto the 1-hydroxy group of a ceramide.
- glycosidiclinkage at the 1-hydroxyl position (see image).
- Cerebrosides have a single glucose or galactose at the 1-hydroxy position.
- Sulfatides are sulfated cerebrosides.
- Gangliosides have at least three sugars, one of which must be sialic acid.
- Cerebrosides have a single glucose or galactose at the 1-hydroxy position.
- Inositol-containing ceramides, which are derived from phytoceramide, are produced in yeast. These include inositol phosphorylceramide, mannose inositol phosphorylceramide, and mannose diinositol phosphorylceramide.
Mammalian sphingolipid metabolism
De novo sphingolipid synthesis begins with formation of 3-keto-dihydrosphingosine by

De novo generated
Breakdown pathways allow the reversion of these metabolites to ceramide. The complex glycosphingolipids are hydrolyzed to glucosylceramide and galactosylceramide. These lipids are then hydrolyzed by beta-glucosidases and beta-galactosidases to regenerate ceramide. Similarly, sphingomyelin may be broken down by sphingomyelinase to form ceramide.[citation needed]
The only route by which sphingolipids are converted to non-sphingolipids is through sphingosine-1-phosphate lyase. This forms ethanolamine phosphate and hexadecenal.[14]
Functions of mammalian sphingolipids
Sphingolipids are commonly believed to protect the cell surface against harmful environmental factors by forming a mechanically stable and chemically resistant outer leaflet of the
Recently, simple sphingolipid metabolites, such as ceramide and sphingosine-1-phosphate, have been shown to be important mediators in the signaling cascades involved in apoptosis, proliferation, stress responses, necrosis, inflammation, autophagy, senescence, and differentiation.[15][16][17][18][19][20] Ceramide-based lipids self-aggregate in cell membranes and form separate phases less fluid than the bulk phospholipids. These sphingolipid-based microdomains, or "lipid rafts" were originally proposed to sort membrane proteins along the cellular pathways of membrane transport. At present, most research focuses on the organizing function during signal transduction.[21]
Sphingolipids are synthesized in a pathway that begins in the
In experimental animals, feeding sphingolipids inhibits colon carcinogenesis, reduces LDL cholesterol and elevates HDL cholesterol.[24]
Other sphingolipids
Sphingolipids are universal in
Yeast sphingolipids
Because of the incredible complexity of mammalian systems, yeast are often used as a model organism for working out new pathways. These single-celled organisms are often more genetically tractable than mammalian cells, and strain libraries are available to supply strains harboring almost any non-lethal open reading frame single deletion. The two most commonly used yeasts are Saccharomyces cerevisiae and Schizosaccharomyces pombe, although research is also done in the pathogenic yeast Candida albicans.[citation needed]
In addition to the important structural functions of complex sphingolipids (inositol phosphorylceramide and its mannosylated derivatives), the sphingoid bases
Plant sphingolipids
Higher plants contain a wider variety of sphingolipids than animals and fungi.[citation needed]
Disorders
There are several disorders of sphingolipid metabolism, known as
Sphingolipids have also been implicated with the frataxin protein (Fxn), the deficiency of which is associated with Friedreich's ataxia (FRDA). Loss of Fxn in the nervous system in mice also activates an iron/sphingolipid/PDK1/Mef2 pathway, indicating that the mechanism is evolutionarily conserved. Furthermore, sphingolipid levels and PDK1 activity are also increased in hearts of FRDA patients, suggesting that a similar pathway is affected in FRDA.[30] Other research has demonstrated that iron accumulation in the nervous systems of flies enhances the synthesis of sphingolipids, which in turn activates 3-phosphoinositide dependent protein kinase-1 (Pdk1) and myocyte enhancer factor-2 (Mef2) to trigger neurodegeneration of adult photoreceptors.[31]
Sphingolipids play a key role in neuronal survival in Parkinson's Disease (PD) and their catabolic pathway alteration in the brain is partly represented in cerebrospinal fluid and blood tissues (Table1) and have the diagnostic potential.[32]
Additional images
See also
References
- PMID 20061941.
- ^ |SIGMA&N5=SEARCH_CONCAT_PNO|BRAND_KEY&F=SPEC Product page at Sigma Aldrich
- PMID 9759481.
- ISBN 0-7514-0253-2
- PMID 18296751.
- PMID 19017611.
- PMID 6652105.
- PMID 6707526.
- PMID 19095642.
- PMID 18499644.
- PMID 16793762.
- S2CID 3962533.
- S2CID 8692993.
- PMID 17416206.
- PMID 12011103.
- PMID 12011102.
- PMID 16415355.
- PMID 8530509.
- PMID 12415011.
- PMID 20156522.
- PMID 10770957.
- PMID 16996025.
- PMID 12011105.
- PMID 10395583.
- S2CID 26181993.
- PMID 11468289.
- PMID 16997623.
- PMID 18296751.
- PMID 19139096.
- PMID 27901468.
- PMID 27343351.
- PMID 35456032.
External links
- Sphingolipids at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Laurila, Pirkka-Pekka; et al. (December 2022). "Sphingolipids accumulate in aged muscle, and their reduction counteracts sarcopenia". Nature Aging. 2 (12): 1159–1175. S2CID 254819305.
