Staphylococcus aureus
| Staphylococcus aureus | |
|---|---|
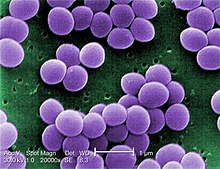
| |
| Scanning electron micrograph of S. aureus; false color added | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Bacillota |
| Class: | Bacilli |
| Order: | Bacillales |
| Family: | Staphylococcaceae |
| Genus: | Staphylococcus |
| Species: | S. aureus
|
| Binomial name | |
| Staphylococcus aureus Rosenbach 1884
| |
| Staphylococcus aureus | |
|---|---|
| Other names | Staph aureus, S. aureus |
Infectious disease | |
| Types | Methicillin-susceptible Staphylococcus aureus, Methicillin-resistant Staphylococcus aureus |
| Causes | Staphylococcus aureus bacteria |
| Differential diagnosis | other bacterial, viral and fungal infections, |
| Prevention | hand washing, cleaning surfaces |
| Medication | Antibiotics |
| Frequency | 20% to 30% of the human population often without symptoms |

Staphylococcus aureus is a
An estimated 21% to 30% of the human population are long-term carriers of S. aureus,
History
Discovery
In 1880,
Evolution
Staphylococcus aureus can be sorted into ten dominant human lineages.
It has been proposed that one possible reason for the great deal of heterogeneity within the species could be due to its reliance on heterogeneous infections. This occurs when multiple different types of S. aureus cause an infection within a host. The different strains can secrete different enzymes or bring different antibiotic resistances to the group, increasing its pathogenic ability.[13] Thus, there is a need for a large number of mutations and acquisitions of mobile genetic elements.[citation needed]
Another notable evolutionary process within the S. aureus species is its co-evolution with its human hosts. Over time, this parasitic relationship has led to the bacterium's ability to be carried in the
Though S. aureus has quick reproductive and micro-evolutionary rates, there are multiple barriers that prevent evolution with the species. One such barrier is AGR, which is a global accessory gene regulator within the bacteria. This such regulator has been linked to the virulence level of the bacteria. Loss of function mutations within this gene have been found to increase the fitness of the bacterium containing it. Thus, S. aureus must make a trade-off to increase their success as a species, exchanging reduced virulence for increased drug resistance. Another barrier to evolution is the Sau1 Type I restriction modification (RM) system. This system exists to protect the bacterium from foreign DNA by digesting it. Exchange of DNA between the same lineage is not blocked, since they have the same enzymes and the RM system does not recognize the new DNA as foreign, but transfer between different lineages is blocked.[13]
Microbiology

S. aureus (
S. aureus is catalase-positive (meaning it can produce the enzyme catalase). Catalase converts hydrogen peroxide (H
2O
2) to water and oxygen. Catalase-activity tests are sometimes used to distinguish staphylococci from enterococci and streptococci. Previously, S. aureus was differentiated from other staphylococci by the coagulase test. However, not all S. aureus strains are coagulase-positive[20][22] and incorrect species identification can impact effective treatment and control measures.[23]
Natural genetic transformation is a reproductive process involving DNA transfer from one bacterium to another through the intervening medium, and the integration of the donor sequence into the recipient genome by homologous recombination. S. aureus was found to be capable of natural genetic transformation, but only at low frequency under the experimental conditions employed.[24] Further studies suggested that the development of competence for natural genetic transformation may be substantially higher under appropriate conditions, yet to be discovered.[25]
Role in health
In humans, S. aureus can be present in the upper respiratory tract, gut mucosa, and skin as a member of the normal microbiota.[26][27][28] However, because S. aureus can cause disease under certain host and environmental conditions, it is characterized as a "pathobiont".[26]
Role in disease

While S. aureus usually acts as a
S. aureus infections can
S. aureus is a significant cause of chronic biofilm infections on
S. aureus can lay dormant in the body for years undetected. Once symptoms begin to show, the host is contagious for another two weeks, and the overall illness lasts a few weeks. If untreated, though, the disease can be deadly.[31] Deeply penetrating S. aureus infections can be severe.[citation needed]
Skin infections
S. aureus is extremely prevalent in persons with
The presence of S. aureus in persons with atopic dermatitis is not an indication to treat with oral antibiotics, as evidence has not shown this to give benefit to the patient.
Food poisoning
S. aureus is also responsible for food poisoning and achieves this by generating toxins in the food, which is then ingested.[39] Its incubation period lasts one to six hours,[40] with the illness itself lasting from 30 minutes to 3 days.[41] Preventive measures one can take to help prevent the spread of the disease include washing hands thoroughly with soap and water before preparing food. The
Bone and joint infections
S. aureus is a common cause of major bone and joint infections, including osteomyelitis, septic arthritis, and infections following joint replacement surgeries.[43][3][44]
Bacteremia
S. aureus is a leading cause of
Without antibiotic treatment, S. aureus bacteremia has a case fatality rate around 80%.[3] With antibiotic treatment, case fatality rates range from 15% to 50% depending on the age and health of the patient, as well as the antibiotic resistance of the S. aureus strain.[3]
Medical implant infections
S. aureus is often found in biofilms formed on medical devices implanted in the body or on human tissue. It is commonly found with another pathogen, Candida albicans, forming multispecies biofilms. The latter is suspected to help S. aureus penetrate human tissue.[9] A higher mortality is linked with multispecies biofilms.[45]
S. aureus biofilm is the predominant cause of orthopedic implant-related infections, but is also found on cardiac implants, vascular grafts, various catheters, and cosmetic surgical implants.[46][47] After implantation, the surface of these devices becomes coated with host proteins, which provide a rich surface for bacterial attachment and biofilm formation. Once the device becomes infected, it must be completely removed, since S. aureus biofilm cannot be destroyed by antibiotic treatments.[47]
Current therapy for S. aureus biofilm-mediated infections involves surgical removal of the infected device followed by antibiotic treatment. Conventional antibiotic treatment alone is not effective in eradicating such infections.[46] An alternative to postsurgical antibiotic treatment is using antibiotic-loaded, dissolvable calcium sulfate beads, which are implanted with the medical device. These beads can release high doses of antibiotics at the desired site to prevent the initial infection.[47]
Novel treatments for S. aureus biofilm involving nano silver particles, bacteriophages, and plant-derived antibiotic agents are being studied. These agents have shown inhibitory effects against S. aureus embedded in biofilms.[48] A class of enzymes have been found to have biofilm matrix-degrading ability, thus may be used as biofilm dispersal agents in combination with antibiotics.[49]
Animal infections
S. aureus can survive on dogs,[50] cats,[51] and horses,[52] and can cause bumblefoot in chickens.[53] Some believe health-care workers' dogs should be considered a significant source of antibiotic-resistant S. aureus, especially in times of outbreak.[50] In a 2008 study by Boost, O’Donoghue, and James, it was found that just about 90% of S. aureus colonized within pet dogs presented as resistant to at least one antibiotic. The nasal region has been implicated as the most important site of transfer between dogs and humans.[54]
S. aureus is one of the causal agents of
Virulence factors
Enzymes
S. aureus produces various enzymes such as coagulase (bound and free coagulases) which facilitates the conversion of fibrinogen to fibrin to cause clots which is important in skin infections.[56]
Toxins
Depending on the strain, S. aureus is capable of secreting several exotoxins, which can be categorized into three groups. Many of these toxins are associated with specific diseases.[60]
- Superantigens
- TSST-1 which causes TSS associated with tampon use. Toxic shock syndrome is characterized by fever, erythematous rash, low blood pressure, shock, multiple organ failure, and skin peeling. Lack of antibody to TSST-1 plays a part in the pathogenesis of TSS. Other strains of S. aureus can produce an enterotoxin that is the causative agent of a type of gastroenteritis. This form of gastroenteritis is self-limiting, characterized by vomiting and diarrhea 1–6 hours after ingestion of the toxin, with recovery in 8 to 24 hours. Symptoms include nausea, vomiting, diarrhea, and major abdominal pain.[62][63]
- Exfoliative toxins
- Exfoliative toxins are exotoxins implicated in the disease staphylococcal scalded skin syndrome (SSSS), which occurs most commonly in infants and young children. It also may occur as epidemics in hospital nurseries. The protease activity of the exfoliative toxins causes peeling of the skin observed with SSSS.[63]
- Other toxins
- Staphylococcal toxins that act on cell membranes include Panton-Valentine leukocidin (PVL), to increase virulence. The bicomponent toxin PVL is associated with severe necrotizing pneumonia in children.[64][65] The genes encoding the components of PVL are encoded on a bacteriophage found in community-associated MRSA strains.[citation needed]
Type VII Secretion system
A secretion system is a highly specialised multi-protein unit that is embedded in the cell envelope with the function of translocating effector proteins from inside of the cell to the extracellular space or into a target host cytosol. The exact structure and function of T7SS is yet to be fully elucidated. Currently, four proteins are known components of S. aureus type VII secretion system; EssC is a large integral membrane ATPase - which most likely powers the secretion systems and has been hypothesised forming part of the translocation channel. The other proteins are EsaA, EssB, EssA, that are membrane proteins that function alongside EssC to mediate protein secretion. The exact mechanism of how substrates reach the cell surface is unknown, as is the interaction of the three membrane proteins with each other and EssC.[66]
T7 dependent effector proteins
EsaD is DNA endonuclease toxin secreted by S. aureus, has been shown to inhibit growth of competitor S. aureus strain in vitro.[67] EsaD is cosecreted with chaperone EsaE, which stabilises EsaD structure and brings EsaD to EssC for secretion.[67][66] Strains that produce EsaD also co-produce EsaG, a cytoplasmic anti-toxin that protects the producer strain from EsaD's toxicity.[67]
TspA is another toxin that mediates intraspecies competition. It is a bacteriostatic toxin that has a membrane depolarising activity facilitated by its
Toxins that play a role in intraspecies competition confers an advantage by promoting successful colonisation in polymicrobial communities such as the nasopharynx and lung by outcompeting lesser strains.[68]
There are also T7 effector proteins that play role a in pathogenesis, for example mutational studies of S. aureus have suggested that EsxB and EsxC contribute to persistent infection in a murine abscess model.[69]
EsxX has been implicated in neutrophil lysis, therefore suggested as contributing to the evasion of host immune system. Deletion of essX in S. aureus resulted in significantly reduced resistance to neutrophils and reduced virulence in murine skin and blood infection models.[70]
Altogether, T7SS and known secreted effector proteins are a strategy of pathogenesis by improving fitness against competitor S. aureus species as well as increased virulence via evading the innate immune system and optimising persistent infections.[citation needed]
Small RNA
The list of small RNAs involved in the control of bacterial virulence in S. aureus is growing. This can be facilitated by factors such as increased biofilm formation in the presence of increased levels of such small RNAs.[71] For example, RNAIII,[72] SprD,[73] SprC,[74][75] RsaE,[76] SprA1,[77] SSR42,[78] ArtR,[79] SprX, and Teg49.[80]
DNA repair
Host neutrophils cause DNA double-strand breaks in S. aureus through the production of reactive oxygen species.[81] For infection of a host to be successful, S. aureus must survive such damages caused by the hosts’ defenses. The two protein complex RexAB encoded by S. aureus is employed in the recombinational repair of DNA double-strand breaks.[81]
Strategies for post-transcriptional regulation by 3'untranslated region
Many mRNAs in S. aureus carry three prime untranslated regions (3'UTR) longer than 100 nucleotides, which may potentially have a regulatory function.[82]
Further investigation of icaR mRNA (mRNA coding for the repressor of the main expolysaccharidic compound of the bacteria biofilm matrix) demonstrated that the 3'UTR binding to the
Biofilm
Studies in biofilm development have shown to be related to changes in gene expression. There are specific genes that were found to be crucial in the different biofilm growth stages. Two of these genes include rocD and gudB, which encode for the enzyme's
Other immunoevasive strategies
- Protein A
Protein A in various recombinant forms has been used for decades to bind and purify a wide range of antibodies by

- Staphylococcal pigments
Some strains of S. aureus are capable of producing staphyloxanthin — a golden-coloured carotenoid pigment. This pigment acts as a virulence factor, primarily by being a bacterial antioxidant which helps the microbe evade the reactive oxygen species which the host immune system uses to kill pathogens.[91][92]
These tests suggest the Staphylococcus strains use staphyloxanthin as a defence against the normal human immune system. Drugs designed to inhibit the production of staphyloxanthin may weaken the bacterium and renew its susceptibility to antibiotics.
Classical diagnosis

Depending upon the type of infection present, an appropriate specimen is obtained accordingly and sent to the laboratory for definitive identification by using biochemical or enzyme-based tests. A
Furthermore, for differentiation on the species level,
Rapid diagnosis and typing
When observing the evolvement of S. aureus and its ability to adapt to each modified antibiotic, two basic methods known as "band-based" or "sequence-based" are employed.
With MLST, this technique of typing uses fragments of several housekeeping genes known as aroE, glpF, gmk, pta, tip, and yqiL. These sequences are then assigned a number which give to a string of several numbers that serve as the allelic profile. Although this is a common method, a limitation about this method is the maintenance of the microarray which detects newly allelic profiles, making it a costly and time-consuming experiment.[99]
With PFGE, a method which is still very much used dating back to its first success in 1980s, remains capable of helping differentiate MRSA isolates.[101] To accomplish this, the technique uses multiple gel electrophoresis, along with a voltage gradient to display clear resolutions of molecules. The S. aureus fragments then transition down the gel, producing specific band patterns that are later compared with other isolates in hopes of identifying related strains. Limitations of the method include practical difficulties with uniform band patterns and PFGE sensitivity as a whole.[citation needed]
Spa locus typing is also considered a popular technique that uses a single locus zone in a polymorphic region of S. aureus to distinguish any form of mutations.[101] Although this technique is often inexpensive and less time-consuming, the chance of losing discriminatory power making it hard to differentiate between MLST clonal complexes exemplifies a crucial limitation.[citation needed]
Treatment
For susceptible strains, the treatment of choice for S. aureus infection is
Antibiotic resistance in S. aureus was uncommon when penicillin was first introduced in 1943. Indeed, the original Petri dish on which Alexander Fleming of Imperial College London observed the antibacterial activity of the Penicillium fungus was growing a culture of S. aureus. By 1950, 40% of hospital S. aureus isolates were penicillin-resistant; by 1960, this had risen to 80%.[106]
Methicillin-resistant Staphylococcus aureus (MRSA, often pronounced /ˈmɜːrsə/ or /ɛm ɑːr ɛs eɪ/), is one of a number of greatly feared strains of S. aureus which have become resistant to most β-lactam antibiotics. For this reason, vancomycin, a glycopeptide antibiotic, is commonly used to combat MRSA. Vancomycin inhibits the synthesis of peptidoglycan, but unlike β-lactam antibiotics, glycopeptide antibiotics target and bind to amino acids in the cell wall, preventing peptidoglycan cross-linkages from forming. MRSA strains are most often found associated with institutions such as hospitals, but are becoming increasingly prevalent in community-acquired infections.[citation needed]
Minor skin infections can be treated with
Antibiotic resistance
S. aureus was found to be the second leading pathogen for deaths associated with antimicrobial resistance in 2019.[108]
Staphylococcal resistance to penicillin is mediated by

Resistance to methicillin is mediated via the mec
Aminoglycoside-modifying enzymes inactivate the aminoglycoside by covalently attaching either a
Glycopeptide resistance is typically mediated by acquisition of the vanA gene, which originates from the Tn1546 transposon found in a plasmid in enterococci and codes for an enzyme that produces an alternative peptidoglycan to which vancomycin will not bind.[113]
Today, S. aureus has become
Despite this, MRSA generally remained an uncommon finding, even in hospital settings, until the 1990s, when the MRSA prevalence in hospitals exploded, and it is now endemic.[115] Now, methicillin-resistant Staphylococcus aureus (MRSA) is not only a human pathogen causing a variety of infections, such as skin and soft tissue infection (SSTI), pneumonia, and sepsis, but it also can cause disease in animals, known as livestock-associated MRSA (LA-MRSA).[116]
MRSA infections in both the hospital and community setting are commonly treated with non-β-lactam antibiotics, such as clindamycin (a lincosamine) and co-trimoxazole (also commonly known as trimethoprim/sulfamethoxazole). Resistance to these antibiotics has also led to the use of new, broad-spectrum anti-Gram-positive antibiotics, such as linezolid, because of its availability as an oral drug. First-line treatment for serious invasive infections due to MRSA is currently glycopeptide antibiotics (vancomycin and teicoplanin). A number of problems with these antibiotics occur, such as the need for intravenous administration (no oral preparation is available), toxicity, and the need to monitor drug levels regularly by blood tests. Also, glycopeptide antibiotics do not penetrate very well into infected tissues (this is a particular concern with infections of the brain and meninges and in endocarditis). Glycopeptides must not be used to treat methicillin-sensitive S. aureus (MSSA), as outcomes are inferior.[117]
Because of the high level of resistance to penicillins and because of the potential for MRSA to develop resistance to vancomycin, the
Vancomycin-resistant S. aureus (VRSA) is a strain of S. aureus that has become resistant to the glycopeptides. The first case of vancomycin-intermediate S. aureus (VISA) was reported in Japan in 1996;[118] but the first case of S. aureus truly resistant to glycopeptide antibiotics was only reported in 2002.[119] Three cases of VRSA infection had been reported in the United States as of 2005.[120] At least in part the antimicrobial resistance in S. aureus can be explained by its ability to adapt. Multiple two component signal transduction pathways helps S. aureus to express genes that are required to survive under antimicrobial stress.[121]
Efflux pumps
Among the various mechanisms that MRSA acquires to elude antibiotic resistance (e.g., drug inactivation, target alteration, reduction of permeability) there is also the overexpression of
These pumps are overexpressed by MDR S. aureus (Multidrug resistant S. aureus) and the result is an excessive expulsion of the antibiotic outside the cell, which makes its action ineffective. Efflux pumps also contribute significantly to the development of impenetrable biofilms.[citation needed]
By directly modulating efflux pumps' activity or decreasing their expression, it may be possible to modify the resistant phenotype and restore the effectiveness of existing antibiotics.[122]
Carriage
About 33% of the U.S. population are carriers of S. aureus and about 2% carry
The carriage of S. aureus is an important source of hospital-acquired infection (also called nosocomial) and community-acquired MRSA. Although S. aureus can be present on the skin of the host, a large proportion of its carriage is through the anterior nares of the nasal passages[2] and can further be present in the ears.[126] The ability of the nasal passages to harbour S. aureus results from a combination of a weakened or defective host immunity and the bacterium's ability to evade host innate immunity.[127] Nasal carriage is also implicated in the occurrence of staph infections.[128]
Infection control
Spread of S. aureus (including MRSA) generally is through human-to-human contact, although recently some veterinarians have discovered the infection can be spread through pets,
Recently,[when?] myriad cases of S. aureus have been reported in hospitals across America. Transmission of the pathogen is facilitated in medical settings where healthcare worker hygiene is insufficient. S. aureus is an incredibly hardy bacterium, as was shown in a study where it survived on polyester for just under three months;[132] polyester is the main material used in hospital privacy curtains.
The bacteria are transported on the hands of healthcare workers, who may pick them up from a seemingly healthy patient carrying a benign or commensal strain of S. aureus, and then pass it on to the next patient being treated. Introduction of the bacteria into the bloodstream can lead to various complications, including endocarditis, meningitis, and, if it is widespread, sepsis.[citation needed]
An important and previously unrecognized means of community-associated MRSA colonization and transmission is during sexual contact.[133]
S. aureus is killed in one minute at 78 °C and in ten minutes at 64 °C but is resistant to freezing.[134][135]
Certain strains of S. aureus have been described as being resistant to chlorine disinfection.[136][137]
The use of mupirocin ointment can reduce the rate of infections due to nasal carriage of S. aureus.[138] There is limited evidence that nasal decontamination of S. aureus using antibiotics or antiseptics can reduce the rates of surgical site infections.[139]
| Top common bacterium in each industry | |
|---|---|
| Catering industry | Vibrio parahaemolyticus, S. aureus, Bacillus cereus |
Medical industry
|
Escherichia coli, S. aureus, Pseudomonas aeruginosa[140] |
Research
As of 2021, no approved vaccine exists against S. aureus. Early clinical trials have been conducted for several vaccines candidates such as Nabi's StaphVax and PentaStaph, Intercell's / Merck's V710, VRi's SA75, and others.[141]
While some of these vaccines candidates have shown immune responses, others aggravated an infection by S. aureus. To date, none of these candidates provides protection against a S. aureus infection. The development of Nabi's StaphVax was stopped in 2005 after phase III trials failed.[142] Intercell's first V710 vaccine variant was terminated during phase II/III after higher mortality and morbidity were observed among patients who developed S. aureus infection.[143]
Nabi's enhanced S. aureus vaccines candidate PentaStaph was sold in 2011 to GlaxoSmithKline Biologicals S.A.[144] The current status of PentaStaph is unclear. A WHO document indicates that PentaStaph failed in the phase III trial stage.[145]
In 2010,
Pfizer's S. aureus four-antigen vaccine SA4Ag was granted fast track designation by the U.S. Food and Drug Administration in February 2014.[148] In 2015, Pfizer has commenced a phase 2b trial regarding the SA4Ag vaccine.[149] Phase 1 results published in February 2017 showed a very robust and secure immunogenicity of SA4Ag.[150] The vaccine underwent clinical trial until June 2019, with results published in September 2020, that did not demonstrate a significant reduction in Postoperative Bloodstream Infection after Surgery.[149]
In 2015, Novartis Vaccines and Diagnostics, a former division of Novartis and now part of GlaxoSmithKline, published promising pre-clinical results of their four-component Staphylococcus aureus vaccine, 4C-staph.[151]
In addition to vaccine development, research is being performed to develop alternative treatment options that are effective against antibiotic resistant strains including MRSA. Examples of alternative treatments are phage therapy, antimicrobial peptides and host-directed therapy.[152][153]
Standard strains
A number of standard strains of S. aureus (called "type cultures") are used in research and in laboratory testing, such as:
| Name | NCTC | ATCC | Year of deposit | Comment |
|---|---|---|---|---|
| Oxford H | 6571 | 9144 | 1943 | Standard strain used for testing penicillin potency and by which the penicillin unit was originally defined.[154][155] |
| Rosenbach | 12973 | 29213 | 1884 | Standard strain for EUCAST antimicrobial resistance testing.[156] |
See also
- Bundaberg tragedy, deaths of 12 children inoculated with an S. aureus-contaminated vaccine
References
- PMID 11717286.
- ^ PMID 9227864.
- ^ PMID 26016486.
- PMID 11687441.
- PMID 19821358.
- ISBN 978-0071716727.
- ^ a b "Staphylococcal Infections". MedlinePlus [Internet]. Bethesda, MD: National Library of Medicine, US.
Skin infections are the most common. They can look like pimples or boils.
- ^ Bowersox J (27 May 1999). "Experimental Staph Vaccine Broadly Protective in Animal Studies". NIH. Archived from the original on 5 May 2007. Retrieved 28 July 2007.
- ^ PMID 25332378.
- ^ Orent W (2006). "A Brief History of Staph". Proto Magazine.
- ^ "S. aureus clonal complex designation". PubMLST. Retrieved 28 February 2024.
- PMID 11447287.
- ^ PMID 19811948.
- PMID 23624187.
- PMID 19000784.
- ^ "Staphylococcus". Dictionary.com Unabridged (Online). n.d. "aureus". Dictionary.com Unabridged (Online). n.d.
- OxfordDictionaries.com. Archived from the originalon 16 July 2012. Retrieved 20 January 2016.
- ^ "PATHOGEN SAFETY DATA SHEET – INFECTIOUS SUBSTANCES." Staphylococcus cells have a diameter of 0.7–1.2 um. Staphylococcus Aureus. Public Health Agency of Canada, 2011. Web
- ^ "Canadian Centre for Occupational Health and Safety". Retrieved 8 April 2016.
- ^ ISBN 978-0-8385-8529-0.
- PMID 24992290.
- ^ PreTest, Surgery, 12th ed., p.88
- PMID 31195568.
- PMID 23133387.
- S2CID 1568023.
- ^ PMID 27730630.
- PMID 28260936.
- PMID 20473345.
- PMID 15963281.
- PMID 27129223.
- ^ "Staphylococcus aureus in Healthcare Settings | HAI". CDC. Retrieved 19 April 2017.
- ^ S2CID 255039948.
- S2CID 21783186.
- ABIM Foundation, American Academy of Dermatology, retrieved 5 December 2013
- PMID 31684694.
- PMID 22777230.
- PMID 25902485.
- PMID 27381887.
- ^ "Staphylococcal Food Poisoning". cdc.gov. hhs.gov. 4 October 2016. Retrieved 23 October 2016.
- ^ "Staphylococcus." Foodsafety.gov, U.S. Department of Health and Human Services, https://www.foodsafety.gov/poisoning/causes/bacteriaviruses/staphylococcus/.
- ^ "Staphylococcal Food Poisoning." Food Safety, Centers for Disease Control and Prevention, 4 October 2016, https://www.cdc.gov/foodsafety/diseases/staphylococcal.html.
- ^ Woodson J. "Centers for disease control and prevention". Food Safety. Archived from the original on 8 February 2016. Retrieved 24 October 2017.
- ^ PMID 21162635.
- ^ Latha T, Anil B, Manjunatha H, Chiranjay M, Elsa D, Baby N, Anice G. MRSA: the leading pathogen of orthopedic infection in a tertiary care hospital, South India. African health sciences. 2019 Apr 16;19(1):1393-401.
- PMID 25875834.
- ^ .
- ^ PMID 21921685.
- PMID 24453168.
- PMID 28351512.
- ^ PMID 17678561.
- PMID 19949556.
- PMID 18978975.
- ^ "Staphylococcosis, Staphylococcal Arthritis, Bumble Foot". The Poultry Site. Retrieved 22 October 2013.
- PMID 17678561.
- PMID 14503727.
- PMID 33522395.
- PMID 20829609.
- PMID 34516771.
- ^ Medical Laboratory Manual For Tropical Countries vol two
- PMID 10627489.
- PMID 32927913.
- PMID 11123352.
- ^ PMID 12682126.
- PMID 10524952.
- ^ S2CID 236915377.
- ^ PMID 27723728.
- ^ PMID 32769205.
- PMID 15657139.
- PMID 28529509.
- PMID 25092913.
- PMID 20300607.
- PMID 20532214.
- PMID 26240382.
- PMID 27596601.
- PMID 20511587.
- S2CID 8217681.
- PMID 22493015.
- S2CID 18371872.
- PMID 25092913.
- ^ a b Ha KP, Clarke RS, Kim GL, Brittan JL, Rowley JE, Mavridou DAI, Parker D, Clarke TB, Nobbs AH, Edwards AM. Staphylococcal DNA Repair Is Required for Infection. mBio. 2020 Nov 17;11(6):e02288-20. doi: 10.1128/mBio.02288-20. PMID: 33203752; PMCID: PMC7683395
- ^ PMID 24367275.
- PMID 22695065.
- ^ a b Vidyasagar, A. (2016). What Are Biofilms? Live Science.
- PMID 23880136.
- PMID 16769197.
- PMID 33584569.
- PMID 7701329.
- PMID 3679545.
- PMID 18193895.
- ^ PMID 16861688.
- ^ PMID 16009720.
- PMID 18276850.
- ^ Shields P, Tsang AY (9 October 2006). "Mannitol Salt Agar Plates Protocols". www.asmscience.org. Retrieved 31 December 2020.
- ^ "Mannitol Salt Agar (MSA) | Culture Media". Microbe Notes. 14 January 2020. Retrieved 31 December 2020.
- PMID 14879282.
- ISBN 978-1-904455-29-5.
- ISBN 978-1-904455-18-9.
- ^ PMID 18718557.
- PMID 16207977.
- ^ .
- PMID 6751182.
- PMID 9860802.
- PMID 19207079.
- PMID 29249276.
- PMID 11294701.
- S2CID 31594289.
- PMID 35065702.
- PMID 28870171.
- PMID 22496652.
- S2CID 4408938.
- PMID 8218273.
- PMID 8215264.
- PMID 14438510.
- PMID 11418528.
- S2CID 21659477.
- PMID 12390067.
- PMID 9249217.
- PMID 12672861.
- PMID 15811021.
- PMID 22571705.
- PMID 35303568.
- ^ "General Information: Community acquired MRSA". CDC. 25 March 2016.
- .
- .
- S2CID 29362695.
- PMID 17472720.
- PMID 16310147.
- PMID 18337614.
- PMID 32847132.
- ^ "CDC Media Relations: Press Release". www.cdc.gov. Retrieved 22 March 2024.
- PMID 10655374.
- PMID 17205449.
- ^ Shafiei Y, Razavilar V, Javadi A (2011). "Thermal Death Time of Staphylococcus Aureus (PTCC=29213) and Staphylococcus Epidermidis (PTCC=1435) in Distilled Water" (PDF). Australian Journal of Basic and Applied Sciences. 5 (11): 1551–4. Archived (PDF) from the original on 2 July 2015.
- PMID 25039601.
- S2CID 84137649.
- S2CID 86676949.
- PMID 18843708.
- PMID 28516472.
- ^ "Food standard agency".
- ^ "A Shot Against MRSA?" (PDF). Resources for the Future. 20 April 2009. Archived (PDF) from the original on 8 March 2016. Retrieved 7 October 2015.
- ^ "Strengthening the immune system as an antimicrobial strategy against Staphylococcus aureus infections" (PDF). FORMATEX RESEARCH CENTER. 11 December 2013. Archived (PDF) from the original on 22 November 2015. Retrieved 7 October 2015.
- ^ "Intercell, Merck terminate V710 Phase II/III trial against S. aureus infection". Merck & Co. 8 June 2011. Retrieved 7 October 2015.
- ^ "Nabi Biopharmaceuticals Completes Final PentaStaph(TM) Milestone" (Press release). GLOBE NEWSWIRE. 27 April 2011. Retrieved 7 October 2015.
- ^ "Vaccines to prevent antibiotic-resistant Staphylococcus aureus (MRSA)infections" (PDF). University of Chicago. Archived (PDF) from the original on 10 September 2016. Retrieved 11 May 2017.
- ^ Clinical trial number NCT01160172 for "A Study to Evaluate the Safety, Reactogenicity and Immunogenicity of GSK Biologicals' Staphylococcal Investigational Vaccine in Healthy Adults" at ClinicalTrials.gov
- ^ "Status of vaccine research and development of vaccines for Staphylococcus aureus" (PDF). ELSEVIER. 19 April 2016. Archived (PDF) from the original on 11 October 2016. Retrieved 10 October 2016.
- ^ "Pfizer Begins Phase 2b Study of Its Investigational Multi-antigen Staphylococcus aureus Vaccine in Adults Undergoing Elective Spinal Fusion Surgery". Pfizer Inc. 7 July 2015. Retrieved 24 February 2016.
- ^ a b Clinical trial number NCT02388165 for "Safety and Efficacy of SA4Ag Vaccine in Adults Having Elective Posterior Instrumented Lumbar Spinal Fusion Procedure (STRIVE)" at ClinicalTrials.gov
- PMID 28143674.
- PMID 26015481.
- PMID 33584628.
- PMID 38590439.
- PMID 13252206.
- PMID 16735421.
- ^ EUCAST (1 January 2020). Routine and extended internal quality control for MIC determination and disk diffusion as recommended by EUCAST: version 10.0. Växjö, Sweden: European Society of Clinical Microbiology and Infectious Diseases. p. 9.
Further reading
- Loskill P, Pereira PM, Jung P, Bischoff M, Herrmann M, Pinho MG, et al. (September 2014). "Reduction of the peptidoglycan crosslinking causes a decrease in stiffness of the Staphylococcus aureus cell envelope". Biophysical Journal. 107 (5): 1082–1089. PMID 25185544.
- Benson MA, Ohneck EA, Ryan C, Alonzo F, Smith H, Narechania A, et al. (August 2014). "Evolution of hypervirulence by a MRSA clone through acquisition of a transposable element". Molecular Microbiology. 93 (4): 664–681. PMID 24962815.
External links
- StopMRSANow.org — Discusses how to prevent the spread of MRSA
- TheMRSA.com — Understand what the MRSA infection is all about.
- "Staphylococcus aureus". NCBI Taxonomy Browser. 1280.
- Packham C (16 March 2015). "Successful in vivo test of breakthrough Staphylococcus aureus vaccine". Medical Press. Archived from the original on 19 September 2012. Retrieved 18 March 2015.
- Type strain of Staphylococcus aureus at BacDive – the Bacterial Diversity Metadatabase
