Staudinger synthesis
The Staudinger synthesis, also called the Staudinger ketene-imine cycloaddition, is a

Reviews on the mechanism, stereochemistry, and applications of the reaction have been published.[3][4][5]
History
The reaction was discovered in 1907 by the German chemist Hermann Staudinger.[6] The reaction did not attract interest until the 1940s, when the structure of penicillin was elucidated. The β-lactam moiety of the first synthetic penicillin was constructed using this cycloaddition,[7] and it remains a valuable tool in synthetic organic chemistry.
Mechanism
The first step is a

Stereochemistry
The

Variations
Reviews on asymmetric induction of the Staudinger synthesis, including the use of organic and organometallic catalysts, have been published.[1][5][13]
The imine can be replaced by adding

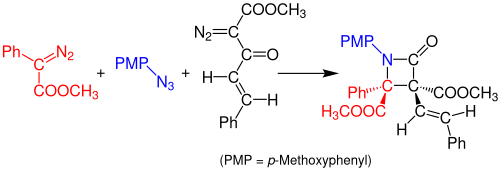
In 2014, Doyle and coworkers reported a one-pot, multicomponent Staudinger synthesis of β-lactams from azides and two diazo compounds. The reaction occurs by a rhodium acetate-catalyzed reaction between the aryldiazoacetate (red) and the organic azide (blue) to form an imine. A Wolff rearrangement of the diazoacetoacetate enone (black) forms a stable ketene, which reacts with the imine to form a stable β-lactam compound. The solvent used for this reaction is dichloromethane (DCM) and the solution needs to rest for 3 hours at room temperature. The yield of the reaction is about 99%.[14]
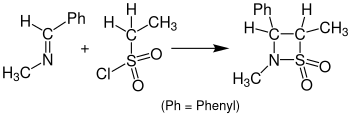
The reaction with sulfenes instead of ketenes leading to β-sultams is called Sulfa-Staudinger cycloaddition. The following illustration shows an example of the Sulfa-Staudinger cycloaddition. Benzylidenemethylamine reacts with ethanesulfonyl chloride to a β-sultam. For this reaction was tetrahydrofuran (THF) used as a solvent and the solution needed to rest for 24 hours.[15]
References
- ^ ISBN 9780470872208.)
{{cite book}}:|first=has generic name (help)CS1 maint: multiple names: authors list (link - PMID 18022986.
- ^ Fu, N.; Tidwell, T. T. "Preparation of β-lactams by [2+2] cycloaddition of ketenes and imines" Tetrahedron 2008, 64, 10465-10496. ([1])
- ISBN 978-0471187998.
- ^ PMID 18662024.
- .
- doi:10.1021/ja01164a534.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ISBN 9780470872208.)
{{cite book}}:|first=has generic name (help)CS1 maint: multiple names: authors list (link - S2CID 37085450.
- PMID 15624943.
- ^ PMID 16669675.
- ^ PMID 19132931.
- .
- PMID 24423056.
- PMID 25756543.
