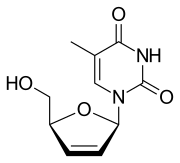
Stavudine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Zerit |
| Other names | 2′,3′-didehydro-2′,3′-dideoxythymidine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a694033 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | >80% |
| Protein binding | Negligible |
| Metabolism | Kidney elimination (~40%) |
| Elimination half-life | 0.8–1.5 hours (in adults) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Stavudine (d4T), sold under the brand name Zerit among others, is an
Common side effects include
Stavudine was first described in 1966 and approved for use in the United States in 1994.
Medical uses
Stavudine is used in the treatment of
The World Health Organization (WHO) recommends stavudine to be phased out to due to its high toxicity levels. If the drug must be used, it is recommended to use low dosages to reduce the occurrence of side effects; however, a 2015 Cochrane review found no clear advantage between high and low dosage regimens.[9]
Pregnancy and breastfeeding
Stavudine has been demonstrated to affect the fetus in animal studies but no data are available from human studies.[2] Pregnant women should therefore be given stavudine only if the potential benefits outweigh the potential harm to the fetus. Additionally, there have been case reports of fatal lactic acidosis in pregnant women receiving combination therapy of stavudine and didanosine with other antiviral agents.[2]
The Centers for Disease Control and Prevention recommend that HIV-infected mothers not breastfeed their infants, in order to avoid the risk of HIV transmission through breast milk.[10] There is also evidence that stavudine gets into animal breast milk, although no data are available for human breast milk.[2]
Children
Stavudine is safe for use in children infected with HIV from birth through adolescence. Adverse effects and safety profile are the same as adults.[2]
Elderly
There is no data available for stavudine use in HIV-infected adults aged 65 years or older. However, among 12,000 people over the age of 65, 30% developed peripheral neuropathy.[2] Additionally, since the elderly are more likely to have decreased renal function, they are more likely to develop toxic side effects.[11]
Adverse events
Common side effects[2]
Severe side effects[2]
- Peripheral neuropathy
- Lactic acidosis
- Pancreatitis
- Hepatotoxicity
- Hepatomegaly with steatosis
- Lipoatrophy/lipodystrophy (fat redistribution/accumulation)
Individuals are monitored for the development of these serious adverse effects. The development of peripheral neuropathy is shown to be dose related, and may be resolved if the drug is discontinued. Individuals with advanced HIV-1 disease, a history of peripheral neuropathy, or individuals on other drugs that have association with neuropathy develop this side effect more often.[2]
Stavudine has been shown in laboratory test to be
HLA-B*4001 may be used as a genetic marker to predict which patients will develop stavudine-associated lipodystrophy, to avoid or shorten the duration of stavudine according to a study in Thailand.[12]
It is still used as first choice in first line therapy in resource poor settings such as in India. Only in case of development of peripheral neuropathy or pregnancy is it changed to the next choice, zidovudine. Safety and effectiveness of dosage titration was not reported in treatment naive patients. It was only reported in those patients with sustained virologic suppression. These findings are not generalized to stavudine used in ART naive patients who have high viral loads.
In November 2009, the
Mechanism of action
Stavudine is a nucleoside
Pharmacokinetics
Absorption: Stavudine has rapid absorption and good oral bioavailability (F = 0.86).[8]
Distribution: Stavudine does not bind to proteins in the blood.[8]
Metabolism: The clearance of stavudine is affected minimally by hepatic metabolism.
Elimination: Stavudine is mostly eliminated in the urine and mostly in its unchanged form.[8]
Drug interactions
Simultaneous use of zidovudine is not recommended, as it can inhibit the intracellular phosphorylation of stavudine. Other anti-HIV drugs do not possess this property.
Stavudine is not protein-bound nor does it inhibit the major cytochrome P450 isoforms. Thus, significant drug interactions with drugs metabolized through these pathways or drugs that are protein-bound are unlikely.[8]
History
Stavudine was first created by Jerome Horwitz in the 1960s and was originally named D4T.[14] When the AIDS epidemic occurred in the 1980s, William Prusoff and others at Yale University discovered the anti-HIV properties of stavudine.[15]
In 1990, Yale patented the use of the drug stavudine (d4T) to treat HIV, and granted an exclusive license to
Stavudine was the first drug to be granted parallel track status in 1992, by the US
In 2018, Mylan Pharmaceuticals discontinued manufacturing stavudine 20 mg, 30 mg, and 40 mg capsules.[18]
References
- FDA. Retrieved 22 October 2023.
- ^ a b c d e f g h i "Stavudine capsule". DailyMed. 21 September 2019. Retrieved 13 October 2020.
- ^ "Zerit EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 13 October 2020.
- ^ a b c d e f g h i j "Stavudine Monograph for Professionals - Drugs.com". www.drugs.com. Archived from the original on 10 November 2016. Retrieved 9 November 2016.
- ISBN 9783527607495. Archivedfrom the original on 8 September 2017.
- ^ "Updated Guidelines for Antiretroviral Postexposure Prophylaxis After Sexual, Injection Drug Use, or Other Nonoccupational Exposure to HIV—United States, 2016" (PDF). Center for Disease Control and Prevention. Annals of Emergency Medicine. Archived (PDF) from the original on 20 November 2016. Retrieved 11 August 2016.
- ^ "Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents" (PDF). Panel on Antiretroviral Guidelines for Adults and Adolescents. U.S. Department of Health and Human Services (HHS). 14 July 2016. Archived from the original (PDF) on 1 November 2016. Retrieved 11 August 2016.
- ^ a b c d e f "Zerit (stavudine) capsules and powder for oral solution prescribing information" (PDF). Princeton, NJ: Bristol-Myers Squibb. December 2012. Archived (PDF) from the original on 31 January 2017.
- PMID 25627012.
- ^ "Pregnant Women, Infants, and Children | Gender | HIV by Group | HIV/AIDS | CDC". www.CDC.gov. Archived from the original on 15 November 2016. Retrieved 15 November 2016.
- ^ "FDA Guideline for Industry: Geriatric Population" (PDF). U.S. Food and Drug Administration. August 1994. Archived (PDF) from the original on 14 September 2016.
- PMID 20073992.
- ^ "New HIV recommendations to improve health, reduce infections and save lives". World Health Organization. 30 November 2009. Archived from the original on 18 January 2010.
- ^ "Jerome Horwitz Obituary". Telegraph.co.uk. Archived from the original on 7 November 2016. Retrieved 6 November 2016.
- ^ from the original on 7 November 2016. Retrieved 6 November 2016.
- ^ Ouellette LL (September 2010). "How Many Patents Does It Take To Make a Drug? Follow-On Pharmaceutical Patents and University Licensing". Michigan Telecommunications and Technology Law Review. 17 (1): 299–336.
- ^ "FDA Approval of Stavudine (d4T) | News | AIDSinfo". AIDSinfo. Archived from the original on 7 November 2016. Retrieved 6 November 2016.
- ^ "Stavudine". Discontinuations Reported to FDA. U.S. Food and Drug Administration (FDA). 30 April 2018.
