Steel
| Steels |
|---|
 |
| Phases |
| Microstructures |
| Classes |
| Other iron-based materials |
|
Steel is an alloy of iron and carbon with improved strength and fracture resistance compared to other forms of iron. Because of its high tensile strength and low cost, steel is one of the most commonly manufactured materials in the world. Steel is used in buildings, as concrete reinforcing rods, in bridges, infrastructure, tools, ships, trains, cars, bicycles, machines, electrical appliances, furniture, and weapons.
Iron is always the main element in steel, but many other elements may be present or added.
Iron is the base metal of steel. Depending on the temperature, it can take two crystalline forms (allotropic forms):
The carbon in typical steel alloys may contribute up to 2.14% of its weight. Varying the amount of carbon and many other alloying elements, as well as controlling their chemical and physical makeup in the final steel (either as solute elements, or as precipitated phases), impedes the movement of the dislocations that make pure iron ductile, and thus controls and enhances its qualities. These qualities include the
of the resulting steel. The increase in steel's strength compared to pure iron is possible only by reducing iron's ductility.Steel was produced in
Further refinements in the process, such as

The noun steel originates from the Proto-Germanic adjective stahliją or stakhlijan 'made of steel', which is related to stahlaz or stahliją 'standing firm'.[4]
The carbon content of steel is between 0.02% and 2.14% by weight for plain carbon steel (iron-carbon alloys). Too little carbon content leaves (pure) iron quite soft, ductile, and weak. Carbon contents higher than those of steel make a brittle alloy commonly called pig iron. Alloy steel is steel to which other alloying elements have been intentionally added to modify the characteristics of steel. Common alloying elements include: manganese, nickel, chromium, molybdenum, boron, titanium, vanadium, tungsten, cobalt, and niobium.[5] Additional elements, most frequently considered undesirable, are also important in steel: phosphorus, sulfur, silicon, and traces of oxygen, nitrogen, and copper.
Plain carbon-iron alloys with a higher than 2.1% carbon content are known as cast iron. With modern steelmaking techniques such as powder metal forming, it is possible to make very high-carbon (and other alloy material) steels, but such are not common. Cast iron is not malleable even when hot, but it can be formed by casting as it has a lower melting point than steel and good castability properties.[5] Certain compositions of cast iron, while retaining the economies of melting and casting, can be heat treated after casting to make malleable iron or ductile iron objects. Steel is distinguishable from wrought iron (now largely obsolete), which may contain a small amount of carbon but large amounts of slag.
Material properties
Origins and production


Iron is commonly found in the Earth's crust in the form of an ore, usually an iron oxide, such as magnetite or hematite. Iron is extracted from iron ore by removing the oxygen through its combination with a preferred chemical partner such as carbon which is then lost to the atmosphere as carbon dioxide. This process, known as smelting, was first applied to metals with lower melting points, such as tin, which melts at about 250 °C (482 °F), and copper, which melts at about 1,100 °C (2,010 °F), and the combination, bronze, which has a melting point lower than 1,083 °C (1,981 °F). In comparison, cast iron melts at about 1,375 °C (2,507 °F).[6] Small quantities of iron were smelted in ancient times, in the solid-state, by heating the ore in a charcoal fire and then welding the clumps together with a hammer and in the process squeezing out the impurities. With care, the carbon content could be controlled by moving it around in the fire. Unlike copper and tin, liquid or solid iron dissolves carbon quite readily.[citation needed]
All of these temperatures could be reached with ancient methods used since the Bronze Age. Since the oxidation rate of iron increases rapidly beyond 800 °C (1,470 °F), it is important that smelting take place in a low-oxygen environment. Smelting, using carbon to reduce iron oxides, results in an alloy (pig iron) that retains too much carbon to be called steel.[6] The excess carbon and other impurities are removed in a subsequent step.[citation needed]
Other materials are often added to the iron/carbon mixture to produce steel with the desired properties.
To inhibit corrosion, at least 11% chromium can be added to steel so that a hard oxide forms on the metal surface; this is known as stainless steel. Tungsten slows the formation of cementite, keeping carbon in the iron matrix and allowing martensite to preferentially form at slower quench rates, resulting in high-speed steel. The addition of lead and sulfur decrease grain size, thereby making the steel easier to turn, but also more brittle and prone to corrosion. Such alloys are nevertheless frequently used for components such as nuts, bolts, and washers in applications where toughness and corrosion resistance are not paramount. For the most part, however, p-block elements such as sulfur, nitrogen, phosphorus, and lead are considered contaminants that make steel more brittle and are therefore removed from steel during the melting processing.[7]
Properties

The density of steel varies based on the alloying constituents but usually ranges between 7,750 and 8,050 kg/m3 (484 and 503 lb/cu ft), or 7.75 and 8.05 g/cm3 (4.48 and 4.65 oz/cu in).[8]
Even in a narrow range of concentrations of mixtures of carbon and iron that make steel, several different metallurgical structures, with very different properties can form. Understanding such properties is essential to making quality steel. At
When steels with exactly 0.8% carbon (known as a eutectoid steel), are cooled, the
As the rate of cooling is increased the carbon will have less time to migrate to form carbide at the grain boundaries but will have increasingly large amounts of pearlite of a finer and finer structure within the grains; hence the carbide is more widely dispersed and acts to prevent slip of defects within those grains, resulting in hardening of the steel. At the very high cooling rates produced by quenching, the carbon has no time to migrate but is locked within the face-centred austenite and forms
Martensite has a lower density (it expands during the cooling) than does austenite, so that the transformation between them results in a change of volume. In this case, expansion occurs. Internal stresses from this expansion generally take the form of
Heat treatment
There are many types of
.Annealing is the process of heating the steel to a sufficiently high temperature to relieve local internal stresses. It does not create a general softening of the product but only locally relieves strains and stresses locked up within the material. Annealing goes through three phases: recovery, recrystallization, and grain growth. The temperature required to anneal a particular steel depends on the type of annealing to be achieved and the alloying constituents.[14]
Quenching involves heating the steel to create the austenite phase then quenching it in water or
Production

When iron is
The ingots are then heated in a soaking pit and
History
Ancient

Steel was known in antiquity and was produced in bloomeries and crucibles.[19][20]
The earliest known production of steel is seen in pieces of ironware excavated from an archaeological site in Anatolia (Kaman-Kalehöyük) which are nearly 4,000 years old, dating from 1800 BC.[21][22]
Steel was produced in Celtic Europe from around 800 BC,[23] high-carbon steel was produced in Britain from 490-375 BC,[24][25] and ultrahigh-carbon steel was produced in the Netherlands from the 2nd-4th centuries AD.[26] The Roman author Horace identifies steel weapons such as the falcata in the Iberian Peninsula, while Noric steel was used by the Roman military.[27]
The reputation of Seric iron of South Asia (wootz steel) grew considerably in the rest of the world.
The
There is evidence that carbon steel was made in Western Tanzania by the ancestors of the Haya people as early as 2,000 years ago by a complex process of "pre-heating" allowing temperatures inside a furnace to reach 1300 to 1400 °C.[31][32][33][34][35][36]
Wootz and Damascus
Evidence of the earliest production of high carbon steel in
The manufacture of
Crucible steel, formed by slowly heating and cooling pure iron and carbon (typically in the form of charcoal) in a crucible, was produced in Merv by the 9th to 10th century AD.[39] In the 11th century, there is evidence of the production of steel in Song China using two techniques: a "berganesque" method that produced inferior, inhomogeneous steel, and a precursor to the modern Bessemer process that used partial decarburization via repeated forging under a cold blast.[52]
Modern

Since the 17th century, the first step in European steel production has been the smelting of iron ore into pig iron in a blast furnace.[53] Originally employing charcoal, modern methods use coke, which has proven more economical.[54][55][56]
Processes starting from bar iron
In these processes,
The production of steel by the
The raw material for this process were bars of iron. During the 17th century, it was realized that the best steel came from oregrounds iron of a region north of Stockholm, Sweden. This was still the usual raw material source in the 19th century, almost as long as the process was used.[58][59]
Crucible steel is steel that has been melted in a crucible rather than having been forged, with the result that it is more homogeneous. Most previous furnaces could not reach high enough temperatures to melt the steel. The early modern crucible steel industry resulted from the invention of Benjamin Huntsman in the 1740s. Blister steel (made as above) was melted in a crucible or in a furnace, and cast (usually) into ingots.[59][60]
Processes starting from pig iron


The modern era in
Another 19th-century steelmaking process was the
These methods of steel production were rendered obsolete by the Linz-Donawitz process of basic oxygen steelmaking (BOS), developed in 1952,[63] and other oxygen steel making methods. Basic oxygen steelmaking is superior to previous steelmaking methods because the oxygen pumped into the furnace limited impurities, primarily nitrogen, that previously had entered from the air used,[64] and because, with respect to the open hearth process, the same quantity of steel from a BOS process is manufactured in one-twelfth the time.[63] Today, electric arc furnaces (EAF) are a common method of reprocessing scrap metal to create new steel. They can also be used for converting pig iron to steel, but they use a lot of electrical energy (about 440 kWh per metric ton), and are thus generally only economical when there is a plentiful supply of cheap electricity.[65]
Industry
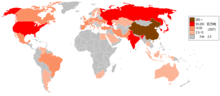
The steel industry is often considered an indicator of economic progress, because of the critical role played by steel in infrastructural and overall economic development.[66] In 1980, there were more than 500,000 U.S. steelworkers. By 2000, the number of steelworkers had fallen to 224,000.[67]
The
In 2005, the British Geological Survey stated China was the top steel producer with about one-third of the world share; Japan, Russia, and the United States were second, third, and fourth, respectively, according to the survey.[71] The large production capacity of steel results also in a significant amount of carbon dioxide emissions inherent related to the main production route.
At the end of 2008, the steel industry faced a sharp downturn that led to many cut-backs.[72]
In 2021, it was estimated that around 7% of the global greenhouse gas emissions resulted from the steel industry.[73][74] Reduction of these emissions are expected to come from a shift in the main production route using cokes, more recycling of steel and the application of carbon capture and storage or carbon capture and utilization technology.
Recycling
Steel is one of the world's most-recycled materials, with a recycling rate of over 60% globally;[3] in the United States alone, over 82,000,000 metric tons (81,000,000 long tons; 90,000,000 short tons) were recycled in the year 2008, for an overall recycling rate of 83%.[75]
As more steel is produced than is scrapped, the amount of recycled raw materials is about 40% of the total of steel produced - in 2016, 1,628,000,000 tonnes (1.602×109 long tons; 1.795×109 short tons) of crude steel was produced globally, with 630,000,000 tonnes (620,000,000 long tons; 690,000,000 short tons) recycled.[76]
Contemporary

Carbon
Modern steels are made with varying combinations of alloy metals to fulfill many purposes.
Recent
Carbon Steels are often
Alloy


Alloy steels are plain-carbon steels in which small amounts of alloying elements like chromium and vanadium have been added. Some more modern steels include
Standards
Most of the more commonly used steel alloys are categorized into various grades by standards organizations. For example, the
Uses

Iron and steel are used widely in the construction of roads, railways, other infrastructure, appliances, and buildings. Most large modern structures, such as
Other common applications include
Historical

Before the introduction of the
With the advent of faster and cheaper production methods, steel has become easier to obtain and much cheaper. It has replaced wrought iron for a multitude of purposes. However, the availability of plastics in the latter part of the 20th century allowed these materials to replace steel in some applications due to their lower fabrication cost and weight.
Long


- As reinforcing bars and mesh in reinforced concrete
- Railroad tracks
- Structural steel in modern buildings and bridges
- Wires
- Input to reforging applications
Flat carbon
- Major appliances
- Magnetic cores
- The inside and outside body of automobiles, trains, and ships.
Weathering (COR-TEN)
- Intermodal containers
- Outdoor sculptures
- Architecture
- train cars
Stainless

- Cutlery
- Rulers
- Surgical instruments
- Watches
- Guns
- Rail passenger vehicles
- Tablets
- Trash Cans
- Body piercing jewellery
- Inexpensive rings
- Components of spacecraft and space stations
Low-background
Steel manufactured after
See also
- Bulat steel
- Direct reduction
- Carbon steel
- Damascus steel
- Galvanising
- History of the steel industry (1970–present)
- Iron in folklore
- List of blade materials
- Machinability
- Noric steel
- Pelletizing
- Rolling
- Rolling mill
- Rust Belt
- Second Industrial Revolution
- Silicon steel
- Steel abrasive
- Steel mill
- Tamahagane, used in Japanese swords
- Tinplate
- Toledo steel
- Wootz steel
References
- JSTOR 2120336. Retrieved November 13, 2020.
- ^ "Decarbonization in steel | McKinsey". www.mckinsey.com. Retrieved 2022-05-20.
- ^ a b Hartman, Roy A. (2009). "Recycling". Encarta. Archived from the original on 2008-04-14.
- ^ Harper, Douglas. "steel". Online Etymology Dictionary.
- ^ ISBN 0-08-032532-7.
- ^ a b Smelting. Encyclopædia Britannica. 2007.
- ^ a b c "Alloying of Steels". Metallurgical Consultants. 2006-06-28. Archived from the original on 2007-02-21. Retrieved 2007-02-28.
- ^ Elert, Glenn. "Density of Steel". Retrieved 2009-04-23.
- ^ Sources differ on this value so it has been rounded to 2.1%, however the exact value is rather academic because plain-carbon steel is very rarely made with this level of carbon. See:
- Smith & Hashemi 2006, p. 363—2.08%.
- Degarmo, Black & Kohser 2003, p. 75—2.11%.
- Ashby & Jones 1992—2.14%.
- ^ Smith & Hashemi 2006, p. 363.
- ^ Smith & Hashemi 2006, pp. 365–372.
- ^ a b Smith & Hashemi 2006, pp. 373–378.
- ^ "Quench hardening of steel". keytometals.com. Archived from the original on 2009-02-17. Retrieved 2009-07-19.
- ^ Smith & Hashemi 2006, p. 249.
- ^ Smith & Hashemi 2006, p. 388.
- ^ Smith & Hashemi 2006, p. 361
- ^ Smith & Hashemi 2006, pp. 361–362.
- ^ Bugayev et al. 2001, p. 225
- ^ a b Davidson 1994, p. 20.
- ^ ISBN 0-85115-355-0. Archived from the originalon 2018-11-19.
- ^ Akanuma, H. (2005). "The significance of the composition of excavated iron fragments taken from Stratum III at the site of Kaman-Kalehöyük, Turkey". Anatolian Archaeological Studies. 14. Tokyo: Japanese Institute of Anatolian Archaeology: 147–158.
- ^ "Ironware piece unearthed from Turkey found to be oldest steel". The Hindu. Chennai, India. 2009-03-26. Archived from the original on 2009-03-29. Retrieved 2022-08-13.
- ISBN 9781135632434.
Plainer (1968) has shown that already at the start of the Iron Age, around 800 BC, some (Celtic) smiths had mastered the techniques for producing fine, hard and sharp-cutting edges by carburising blades to make the iron-carbon alloy, steel.
- ^ "East Lothian's Broxmouth fort reveals edge of steel". BBC News. 15 January 2014.
- ISBN 978-1-908332-05-9.
- .
- ^ "Noricus ensis", Horace, Odes, i. 16.9
- ISBN 90-04-09632-9.
- ^ Needham, Joseph (1986). Science and Civilization in China: Volume 4, Part 3, Civil Engineering and Nautics. Taipei: Caves Books, Ltd. p. 563.
- ISBN 0-521-49781-7.
- S2CID 37926350.
- .
- ^ Schmidt, Peter (1978). Historical Archaeology: A Structural Approach in an African Culture. Westport, CT: Greenwood Press.
- ^ Avery, Donald; Schmidt, Peter (1996). "Preheating: Practice or illusion". The Culture and Technology of African Iron Production. Gainesville: University of Florida Press. pp. 267–276.
- ^ Schmidt, Peter (2019). "Science in Africa: A history of ingenuity and invention in African iron technology". In Worger, W; Ambler, C; Achebe, N (eds.). A Companion to African History. Hoboken, NJ: Wiley Blackwell. pp. 267–288.
- ^ Childs, S. Terry (1996). "Technological history and culture in western Tanzania". In Schmidt, P. (ed.). The Culture and Technology of African Iron Production. Gainesville, FL: University of Florida Press.
- ^ Wilford, John Noble (1996-02-06). "Ancient Smelter Used Wind To Make High-Grade Steel". The New York Times.
- ^ OCLC 82439861. Archived from the originalon 2019-02-11. Retrieved 2014-12-05.
- ^ a b Feuerbach, Ann (2005). "An investigation of the varied technology found in swords, sabres and blades from the Russian Northern Caucasus" (PDF). IAMS. 25: 27–43 (p. 29). Archived from the original (PDF) on 2011-04-30.
- doi:10.5334/pia.60.
- ^ Hobbies – Volume 68, Issue 5 – p. 45. Lightner Publishing Company (1963)
- ^ Mahathevan, Iravatham (24 June 2010). "An epigraphic perspective on the antiquity of Tamil". The Hindu. Archived from the original on 1 July 2010. Retrieved 31 October 2010.
- ^ Ragupathy, P (28 June 2010). "Tissamaharama potsherd evidences ordinary early Tamils among population". Tamilnet. Retrieved 31 October 2010.
- ISBN 0-521-05802-3. Archived from the original(PDF) on 2017-07-03. Retrieved 2017-08-04.
- ISBN 978-0-543-92943-3.
- ^ S2CID 205026185.
- ^ a b Coghlan, Herbert Henery. (1977). Notes on prehistoric and early iron in the Old World. Oxprint. pp. 99–100
- ISBN 978-0-543-92943-3.
- ISBN 0-671-54800-X.
- S2CID 136774602.
- ^ Wayman, M.L. & Juleff, G. (1999). "Crucible Steelmaking in Sri Lanka". Historical Metallurgy. 33 (1): 26.
- S2CID 154556274.
- ^ ISBN 0-901462-88-8.
- ^ Raistrick, A. (1953) A Dynasty of Ironfounders.
- ^ Hyde, C.K. (1977) Technological Change and the British iron industry. Princeton
- ^ Trinder, B. (2000) The Industrial Revolution in Shropshire. Chichester.
- ^ Barraclough 1984, pp. 48–52.
- ^ King, P.W. (2003). "The Cartel in Oregrounds Iron: trading in the raw material for steel during the eighteenth century". Journal of Industrial History. 6 (1): 25–49.
- ^ a b c d "Iron and steel industry". Encyclopædia Britannica. 2007.
- ^ Barraclough, K.C. (1984) Steel before Bessemer: II Crucible Steel: the growth of technology. The Metals Society, London.
- ISBN 0-8337-3463-6.
- ^ Bessemer process. Vol. 2. Encyclopædia Britannica. 2005. p. 168.
- ^ a b Sherman, Zander (4 September 2019). "How my great-grandfather's Dofasco steel empire rose and fell, and his descendants with it". The Globe and Mail Inc.
- ^ Basic oxygen process. Encyclopædia Britannica. 2007.
- ^ Fruehan & Wakelin 1998, pp. 48–52.
- ^ "Steel Industry". Archived from the original on 2009-06-18. Retrieved 2009-07-12.
- United States Government Printing Office.
- ^ Chopra, Anuj (February 12, 2007). "India's steel industry steps onto world stage". Cristian Science Monitor. Retrieved 2009-07-12.
- ^ "Worldsteel | World crude steel output decreases by -2.8% in 2015". Archived from the original on 2017-02-02. Retrieved 2016-12-26.
- ^ "Top Steelmakers in 2017" (PDF). World Steel Association. Archived from the original (PDF) on August 23, 2018. Retrieved August 22, 2018.
- ^ "Long-term planning needed to meet steel demand". The News. 2008-03-01. Archived from the original on 2010-11-02. Retrieved 2010-11-02.
- ^ Uchitelle, Louis (2009-01-01). "Steel Industry, in Slump, Looks to Federal Stimulus". The New York Times. Retrieved 2009-07-19.
- ^ Rossi, Marcello (2022-08-04). "The Race to Remake the $2.5 Trillion Steel Industry With Green Steel". Singularity Hub. Retrieved 2022-08-06.
- ^ "Global Steel Industry's GHG Emissions". Global Efficiency Intelligence. 6 January 2021. Retrieved 2022-08-06.
- ISBN 978-1-4113-3015-3.
- ^ The World Steel Association (2018-03-01). "Steel and raw materials" (PDF).
- ^ "High strength low alloy steels". Schoolscience.co.uk. Archived from the original on 2020-09-21. Retrieved 2007-08-14.
- ^ "Dual-phase steel". Intota Expert Knowledge Services. Archived from the original on 2011-05-25. Retrieved 2007-03-01.
- ^ Werner, Ewald. "Transformation Induced Plasticity in low alloyed TRIP-steels and microstructure response to a complex stress history". Archived from the original on December 23, 2007. Retrieved 2007-03-01.
- ^ Mirko, Centi; Saliceti Stefano. "Transformation Induced Plasticity (TRIP), Twinning Induced Plasticity (TWIP) and Dual-Phase (DP) Steels". Tampere University of Technology. Archived from the original on 2008-03-07. Retrieved 2007-03-01.
- ^ Galvanic protection. Encyclopædia Britannica. 2007.
- ^ "Steel Glossary". American Iron and Steel Institute (AISI). Retrieved 2006-07-30.
- ^ "Steel Interchange". American Institute of Steel Construction Inc. (AISC). Archived from the original on 2007-12-22. Retrieved 2007-02-28.
- ^ "Properties of Maraging Steels". Archived from the original on 2009-02-25. Retrieved 2009-07-19.
- ^ Tweedale, Geoffrey, ed. (1987). Sheffield Steel and America: A Century of Commercial and Technological Independence. Cambridge University Press. pp. 57–62.
- ISBN 0-8031-3362-6. Archived from the original(PDF) on 2007-01-27.
- ^ Steel Construction Manual, 8th Edition, second revised edition, American Institute of Steel Construction, 1986, ch. 1 pp. 1–5
- ^ Ochshorn, Jonathan (2002-06-11). "Steel in 20th Century Architecture". Encyclopedia of Twentieth Century Architecture. Retrieved 2010-04-26.
- ^ Venables, John D.; Girifalco, Louis A.; Patel, C. Kumar N.; McCullough, R.L.; Marchant, Roger Eric; Kukich, Diane S. (2007). Materials science. Encyclopædia Britannica.
Bibliography
- Ashby, Michael F.; Jones, David Rayner Hunkin (1992). An introduction to microstructures, processing and design. Butterworth-Heinemann.
- Barraclough, K. C. (1984). Steel before Bessemer: I Blister Steel: the birth of an industry. London: The Metals Society.
- Bugayev, K.; Konovalov, Y.; Bychkov, Y.; Tretyakov, E.; Savin, Ivan V. (2001). Iron and Steel Production. The Minerva Group, Inc. ISBN 978-0-89499-109-7.
- Davidson, H. R. Ellis (1994). The Sword in Anglo-Saxon England: Its Archaeology and Literature. Woodbridge, Suffolk, UK: Boydell Press. ISBN 0-85115-355-0.
- Degarmo, E. Paul; Black, J T.; Kohser, Ronald A. (2003). Materials and Processes in Manufacturing (9th ed.). Wiley. ISBN 0-471-65653-4.
- Fruehan, R. J.; Wakelin, David H. (1998). The Making, Shaping, and Treating of Steel (11th ed.). Pittsburgh, PA: AISE Steel Foundation. ISBN 0-930767-03-9.
- Verein Deutscher Eisenhüttenleute (Ed.). Steel – A Handbook for Materials Research and Engineering, Volume 1: Fundamentals. Springer-Verlag Berlin, Heidelberg and Verlag Stahleisen, Düsseldorf 1992, 737 p. ISBN 3-540-52968-3, 3-514-00377-7.
- Verein Deutscher Eisenhüttenleute (Ed.). Steel – A Handbook for Materials Research and Engineering, Volume 2: Applications. Springer-Verlag Berlin, Heidelberg and Verlag Stahleisen, Düsseldorf 1993, 839 pages, ISBN 3-540-54075-X, 3-514-00378-5.
- Smith, William F.; Hashemi, Javad (2006). Foundations of Materials Science and Engineering (4th ed.). McGraw-Hill. ISBN 0-07-295358-6.
Further reading
- Mark Reutter, Making Steel: Sparrows Point and the Rise and Ruin of American Industrial Might. University of Illinois Press, 2005.
- Duncan Burn, The Economic History of Steelmaking, 1867–1939: A Study in Competition Archived 2012-07-26 at the Wayback Machine. Cambridge University Press, 1961.
- Harukiyu Hasegawa, The Steel Industry in Japan: A Comparison with Britain Archived 2012-04-18 at the Wayback Machine. Routledge, 1996.
- J.C. Carr and W. Taplin, History of the British Steel Industry Archived 2012-07-29 at the Wayback Machine. Harvard University Press, 1962.
- H. Lee Scamehorn, Mill & Mine: The Cf&I in the Twentieth Century Archived 2012-07-26 at the Wayback Machine. University of Nebraska Press, 1992.
- Warren, Kenneth, Big Steel: The First Century of the United States Steel Corporation, 1901–2001 Archived 2010-05-01 at the Wayback Machine. University of Pittsburgh Press, 2001.
External links
- Official website of the World Steel Association (worldsteel)
- steeluniversity.org: Online steel education resources, an initiative of World Steel Association
- Metallurgy for the Non-Metallurgist from the American Society for Metals
- MATDAT Database of Properties of Unalloyed, Low-Alloy and High-Alloy Steels – obtained from published results of material testing

