Stevens rearrangement
The Stevens rearrangement in
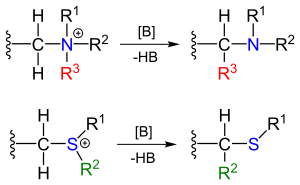
The reactants can be obtained by alkylation of the corresponding amines and sulfides. The substituent R next the amine methylene bridge is an electron-withdrawing group.
The original 1928 publication by Thomas S. Stevens[2] concerned the reaction of 1-phenyl-2-(N,N-dimethylamino)ethanone with benzyl bromide to the ammonium salt followed by the rearrangement reaction with sodium hydroxide in water to the rearranged amine.
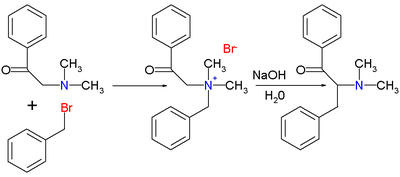
A 1932 publication[3] described the corresponding sulfur reaction.
Reaction mechanism
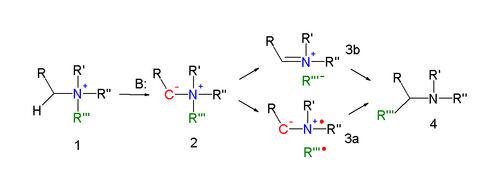
The reaction mechanism of the Stevens rearrangement is one of the most controversial reaction mechanisms in organic chemistry.[4] Key in the reaction mechanism[5][6] for the Stevens rearrangement (explained for the nitrogen reaction) is the formation of an ylide after deprotonation of the ammonium salt by a strong base. Deprotonation is aided by electron-withdrawing properties of substituent R. Several reaction modes exist for the actual rearrangement reaction.
A
In an alternative reaction mechanism the N–C bond of the leaving group is
Scope
Competing reactions are the
In one application a double-Stevens rearrangement expands a cyclophane ring.[7] The ylide is prepared in situ by reaction of the diazo compound ethyl diazomalonate with a sulfide catalyzed by dirhodium tetraacetate in refluxing xylene.
Enzymatic reaction
Recently, γ-butyrobetaine hydroxylase,[8][9] an enzyme that is involved in the human carnitine biosynthesis pathway, was found to catalyze a C-C bond formation reaction in a fashion analogous to a Stevens type rearrangement.[8][10] The substrate for the reaction is meldonium.[11]
See also
References
- )
- .
- .
- ^ Bhakat, S (2011). "The controversial reaction mechanism of Stevens rearrangement: A review". J. Chem. Pharm. Res. 3 (1): 115–121.
- ISBN 0-471-58589-0)
- ISBN 0-12-429785-4
- ^ PMID 21168767.
- PMID 20599753.
- PMID 22765904.
- PMID 3342076.

