Aromatic sulfonation
In

Stoichiometry and mechanism

Typical conditions involve heating the aromatic compound with sulfuric acid:[2]
- C6H6 + H2SO4 → C6H5SO3H + H2O
Sulfur trioxide or its protonated derivative is the actual electrophile in this electrophilic aromatic substitution.
To drive the equilibrium, dehydrating agents such as thionyl chloride can be added.
- C6H6 + H2SO4 + SOCl2 → C6H5SO3H + SO2 + 2 HCl
Chlorosulfuric acid is also an effective agent:
- C6H6 + HSO3Cl → C6H5SO3H + HCl
In contrast to
Specialized sulfonation methods
Many method have been developed for introducing sulfonate groups aside from direction sulfonation.
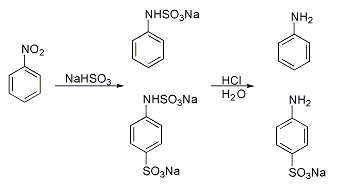
Piria reaction
A classic named reaction is the Piria reaction (
Tyrer sulfonation process
In the Tyrer sulfonation process (1917),[6] at some time of technological importance, benzene vapor is led through a vessel containing 90% sulfuric acid the temperature of which is increased from 100 to 180°C. Water and benzene are continuously removed in a condenser and the benzene layer fed back to the vessel. In this way an 80% yield is obtained.

Applications
Aromatic sulfonic acids are intermediates in the preparation of dyes and many pharmaceuticals. Sulfonation of anilines lead to a large group of sulfa drugs.

Sulfonation of
Reactions of aryl sulfonic acids
As a functional group, aryl sulfonic acids undergo desulfonation when heated in water:
- R−C6H4SO3H + H2O → R−C6H5 + H2SO4
When treated with strong base, benzenesulfonic acid derivatives convert to phenols.[8]
- C6H5SO3H + 2 NaOH → C6H5OH + Na2SO+3 H2O
See also
References
- OCLC 642506595.
- ISBN 978-1-118-13357-6.
- ISSN 0075-4617.
- .
- ^ U.S. patent 1,210,725
- ISBN 3-342-00280-8.
- .
