Sulfonic acid

In
Preparation

Aryl sulfonic acids are produced by the process of
In this reaction, sulfur trioxide is an
Alkylsulfonic acids can be prepared by many methods. In sulfoxidation, alkanes are irradiated with a mixture of sulfur dioxide and oxygen. This reaction is employed industrially to produce alkyl sulfonic acids, which are used as surfactants.[2]
Direct reaction of alkanes with sulfur trioxide is not generally useful, except for the conversion methanesulfonic acid to methanedisulfonic acid.
Many alkane sulfonic acids can be obtained by the addition of
Sulfonic acids can be prepared by oxidation of thiols:
This pathway is the basis of the biosynthesis of taurine.
Hydrolysis routes
Many sulfonic acids are prepared by hydrolysis of
Vinylsulfonic acid is derived by hydrolysis of carbyl sulfate, (C2H4(SO3)2), which in turn is obtained by the addition of sulfur trioxide to ethylene.
Properties
Sulfonic acids are strong acids. They are commonly cited as being around a million times stronger than the corresponding
Because of their polarity, sulfonic acids tend to be crystalline solids or viscous, high-boiling liquids. They are also usually colourless and nonoxidizing,[6] which makes them suitable for use as acid catalysts in organic reactions. Their polarity, in conjunction with their high acidity, renders short-chain sulfonic acids water-soluble, while longer-chain ones exhibit detergent-like properties.
The structure of sulfonic acids is illustrated by the prototype, methanesulfonic acid. The sulfonic acid group, RSO2OH features a tetrahedral sulfur centre, meaning that sulfur is at the center of four atoms: three oxygens and one carbon. The overall geometry of the sulfur centre is reminiscent of the shape of sulfuric acid.
- Representative sulfonic acids and sulfonates
-
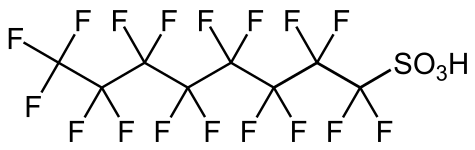
PFOS, a surfactant and a controversial pollutant.
-
p-Toluenesulfonic acid, a widely used reagent in organic synthesis.
-
Nafion, a polymeric sulfonic acid useful in fuel cells.
-
Sodium dodecylbenzenesulfonate, an alkylbenzenesulfonate surfactant used in laundry detergents.
-
Coenzyme-M, is a cofactor required for the biosynthesis of methane, found in natural gas.
Applications
Both alkyl and aryl sulfonic acids are known, most large-scale applications are associated with the aromatic derivatives.
Detergents and surfactants
Dyes
Many if not most of the
Acid catalysts
Being strong acids, sulfonic acids are also used as
Drugs
Sulfa drugs, a class of antibacterials, are produced from sulfonic acids.
Lignosulfonates
In the

Reactions
Hydrolysis
Arylsulfonic acids are susceptible to hydrolysis, the reverse of the sulfonation reaction. Whereas benzenesulfonic acid hydrolyzes above 200 °C, most related derivatives are easier to hydrolyze. Thus, heating aryl sulfonic acids in aqueous acid produces the parent arene. This reaction is employed in several scenarios. In some cases the sulfonic acid serves as a water-solubilizing protecting group, as illustrated by the purification of para-xylene via its sulfonic acid derivative. In the synthesis of 2,6-dichlorophenol, phenol is converted to its 4-sulfonic acid derivative, which then selectively chlorinates at the positions flanking the phenol. Hydrolysis releases the sulfonic acid group.[10]
Esterification
Sulfonic acids can be converted to
- RSO2Cl + R′OH → RSO2OR′ + HCl
Halogenation
Sulfonyl halide groups occur when a sulfonyl functional group is singly bonded to a halogen atom. They have the general formula R−SO2−X where X is a halide, usually chloride or fluoride. Chlorides are produced by chlorination of sulfonic acids using thionyl chloride. Sulfonyl fluorides can be produced by treating sulfonic acids with sulfur tetrafluoride:[11]
- SF4 + RSO3H → SOF2 + RSO2F + HF
Displacement by hydroxide
Although strong, the (aryl)C−SO3− bond can be broken by nucleophilic reagents. Of historic and continuing significance is the α-sulfonation of anthroquinone followed by displacement of the sulfonate group by other nucleophiles, which cannot be installed directly.
- C6H5SO3Na + NaOH → C6H5OH + Na2SO3
The conditions for this reaction are harsh, however, requiring 'fused alkali' or molten sodium hydroxide at 350 °C for benzenesulfonic acid itself.
Notes
- ^ Neither the parent sulfonic acid nor the parent sulfurous acid have been isolated or even observed, although the monoanion of these hypothetical species exists in solution as an equilibrium mixture of tautomers: HS(=O)2(O−) ⇌ S(=O)(OH)(O−).
References
- ^ ISBN 0-471-60180-2
- ^ ISBN 978-3527306732.
- ISSN 0001-4842.
- OCLC 708034394.
- )
- ISSN 1463-9262.
- .
- ^ ISBN 978-3527306732.
- PMID 17973436.
- ISBN 978-3527306732.
- ISBN 978-0-471-26418-7.
- .
- ISSN 0009-2665.
- .