Sulforaphane
| |
| |
| |
| Names | |
|---|---|
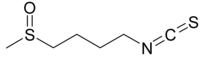
| Preferred IUPAC name
1-Isothiocyanato-4-(methanesulfinyl)butane | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H11NOS2 | |
| Molar mass | 177.29 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sulforaphane (sometimes sulphoraphane in
organosulfur compounds.[1] It is produced when the enzyme myrosinase transforms glucoraphanin, a glucosinolate
, into sulforaphane upon damage to the plant (such as from chewing or chopping during food preparation), which allows the two compounds to mix and react.
Sulforaphane is present in cruciferous vegetables, such as broccoli, Brussels sprouts, and cabbage.[1]
Sulforaphane has two possible
stereoisomers due to the presence of a stereogenic sulfur atom.[2]
The R-sulforaphane enantiomer occurs naturally, while the S-sulforaphane can be synthesized.[3]
 Glucoraphanin, the glucosinolate precursor to sulforaphane |
Occurrence and isolation
Sulforaphane occurs in
Research
Although there has been some basic research on how sulforaphane might have effects in vivo, there is no clinical evidence that consuming cruciferous vegetables and sulforaphane affects the risk of cancer or any other disease, as of 2017.[1][5][needs update]
