Sulfur
 | ||||||||||||||||||||||||||||||||||||
| Sulfur | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alternative name | Sulphur (British spelling) | |||||||||||||||||||||||||||||||||||
| Allotropes | see Allotropes of sulfur | |||||||||||||||||||||||||||||||||||
| Appearance | Lemon yellow sintered microcrystals | |||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(S) | ||||||||||||||||||||||||||||||||||||
| Sulfur in the periodic table | ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
kJ/mol | ||||||||||||||||||||||||||||||||||||
| Heat of vaporization | beta (β-S8): 45 kJ/mol | |||||||||||||||||||||||||||||||||||
| Molar heat capacity | 22.75 J/(mol·K) | |||||||||||||||||||||||||||||||||||
Vapor pressure
| ||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||
Discovery | before 2000 BCE[6] | |||||||||||||||||||||||||||||||||||
| Recognized as an element by | Antoine Lavoisier (1777) | |||||||||||||||||||||||||||||||||||
| Isotopes of sulfur | ||||||||||||||||||||||||||||||||||||
34S abundances vary greatly (between 3.96 and 4.77 percent) in natural samples. | ||||||||||||||||||||||||||||||||||||
Sulfur (also spelled sulphur in
Sulfur is the tenth most abundant element by mass in the universe and the fifth most abundant on Earth. Though sometimes found in pure,
Sulfur is an
Characteristics
Physical properties

Sulfur forms several polyatomic molecules. The best-known allotrope is
Sulfur sublimes more or less between 20 °C (68 °F) and 50 °C (122 °F).[18]
Sulfur is insoluble in water but soluble in carbon disulfide and, to a lesser extent, in other nonpolar organic solvents, such as benzene and toluene.
Chemical properties
Under normal conditions, sulfur hydrolyzes very slowly to mainly form hydrogen sulfide and sulfuric acid:
8 + 4 H
2O → 3 H
2S + H
2SO
4
The reaction involves adsorption of protons onto S
8 clusters, followed by disproportionation into the reaction products.[19]
The second, fourth and sixth ionization energies of sulfur are 2252 kJ/mol, 4556 kJ/mol and 8495.8 kJ/mol, respectively. A composition of products of sulfur's reactions with oxidants (and its oxidation state) depends on that whether releasing out of a reaction energy overcomes these thresholds. Applying catalysts and / or supply of outer energy may vary sulfur's oxidation state and a composition of reaction products. While reaction between sulfur and oxygen at normal conditions gives sulfur dioxide (oxidation state +4), formation of sulfur trioxide (oxidation state +6) requires temperature 400–600 °C (750–1,100 °F) and presence of a catalyst.
In reactions with elements of lesser electronegativity, it reacts as an oxidant and forms sulfides, where it has oxidation state −2.
Sulfur reacts with nearly all other elements with the exception of the noble gases, even with the notoriously unreactive metal iridium (yielding iridium disulfide).[20] Some of those reactions need elevated temperatures.[21]
Allotropes

Sulfur forms over 30 solid allotropes, more than any other element.[22] Besides S8, several other rings are known.[23] Removing one atom from the crown gives S7, which is of a deeper yellow than S8. HPLC analysis of "elemental sulfur" reveals an equilibrium mixture of mainly S8, but with S7 and small amounts of S6.[24] Larger rings have been prepared, including S12 and S18.[25][26]
Isotopes
Sulfur has 23 known
The preponderance of 32S is explained by its production in the so-called alpha-process (one of the main classes of nuclear fusion reactions) in exploding stars. Other stable sulfur isotopes are produced in the bypass processes related with 34Ar, and their composition depends on a type of a stellar explosion. For example, proportionally more 33S comes from
On the planet Earth the sulfur isotopic composition was determined by the Sun. Though it is assumed that the distribution of different sulfur isotopes should be more or less equal, it has been found that proportions of two most abundant sulfur isotopes 32S and 34S varies in different samples. Assaying of these isotopes ratio (δ34S) in the samples allows to make suggestions about their chemical history, and with support of other methods, it allows to age-date the samples, estimate temperature of equilibrium between ore and water, determine pH and oxygen fugacity, identify the activity of sulfate-reducing bacteria in the time of formation of the sample, or suggest the main sources of sulfur in ecosystems.[30] However, there are ongoing discussions about what is the real reason of the δ34S shifts, biological activity or postdeposital alteration.[31]
For example, when
of the ore-bearing fluid during ore formation.Scientists measure the
In most
The radioactive 35S is formed in
Because of the weak beta activity of 35S, its compounds are relatively safe as long as they are not ingested or absorbed by the body.[36]
Natural occurrence



32S is created inside massive stars, at a depth where the temperature exceeds 2.5×109 K, by the
Sulfur, usually as sulfide, is present in many types of meteorites. Ordinary chondrites contain on average 2.1% sulfur, and carbonaceous chondrites may contain as much as 6.6%. It is normally present as troilite (FeS), but there are exceptions, with carbonaceous chondrites containing free sulfur, sulfates and other sulfur compounds.[38] The distinctive colors of Jupiter's volcanic moon Io are attributed to various forms of molten, solid, and gaseous sulfur.[39]
It is the fifth most common element by mass in the Earth. Elemental sulfur can be found near
Native sulfur is synthesised by
Common naturally occurring sulfur compounds include the
The main industrial source of sulfur is now petroleum and natural gas.[9]
Compounds
Common oxidation states of sulfur range from −2 to +6. Sulfur forms stable compounds with all elements except the noble gases.
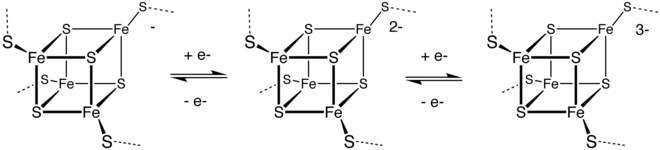
Electron transfer reactions

3)
Sulfur polycations, S82+, S42+ and S162+ are produced when sulfur is reacted with oxidising agents in a strongly acidic solution.[46] The colored solutions produced by dissolving sulfur in oleum were first reported as early as 1804 by C.F. Bucholz, but the cause of the color and the structure of the polycations involved was only determined in the late 1960s. S82+ is deep blue, S42+ is yellow and S162+ is red.[13]
Reduction of sulfur gives various polysulfides with the formula Sx2−, many of which have been obtained in crystalline form. Illustrative is the production of sodium tetrasulfide:
Some of these dianions dissociate to give radical anions, such as S3− gives the blue color of the rock lapis lazuli.

This reaction highlights a distinctive property of sulfur: its ability to catenate (bind to itself by formation of chains). Protonation of these polysulfide anions produces the polysulfanes, H2Sx, where x = 2, 3, and 4.[48] Ultimately, reduction of sulfur produces sulfide salts:
The interconversion of these species is exploited in the sodium–sulfur battery.
Hydrogenation
Treatment of sulfur with hydrogen gives hydrogen sulfide. When dissolved in water, hydrogen sulfide is mildly acidic:[7]
Hydrogen sulfide gas and the hydrosulfide anion are extremely toxic to mammals, due to their inhibition of the oxygen-carrying capacity of hemoglobin and certain cytochromes in a manner analogous to cyanide and azide (see below, under precautions).
Combustion
The two principal sulfur oxides are obtained by burning sulfur:
Many other sulfur oxides are observed including the sulfur-rich oxides include sulfur monoxide, disulfur monoxide, disulfur dioxides, and higher oxides containing peroxo groups.
Halogenation
Sulfur reacts with fluorine to give the highly reactive sulfur tetrafluoride and the highly inert sulfur hexafluoride.[49] Whereas fluorine gives S(IV) and S(VI) compounds, chlorine gives S(II) and S(I) derivatives. Thus, sulfur dichloride, disulfur dichloride, and higher chlorosulfanes arise from the chlorination of sulfur. Sulfuryl chloride and chlorosulfuric acid are derivatives of sulfuric acid; thionyl chloride (SOCl2) is a common reagent in organic synthesis.[50] Bromine also oxidizes sulfur to form sulfur dibromide and disulfur dibromide.[50]
Pseudohalides
Sulfur oxidizes cyanide and sulfite to give thiocyanate and thiosulfate, respectively.
Metal sulfides
Sulfur reacts with many metals. Electropositive metals give polysulfide salts. Copper, zinc, and silver are attacked by sulfur; see
Organic compounds
- Illustrative organosulfur compounds
-
(L)-cysteine, an amino acid containing a thiol group
-
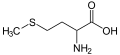
Methionine, an amino acid containing a thioether
-
Thiamine or vitamin B1
-
Biotin or vitamin B7
-
Penicillin, an antibiotic ("R" is the variable group)
-
Allicin, a chemical compound in garlic
-
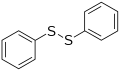
Diphenyl disulfide, a representative disulfide
-
Dibenzothiophene, a component of crude oil
-
Perfluorooctanesulfonic acid (PFOS), a surfactant
Some of the main classes of sulfur-containing organic compounds include the following:[53]
- thiolateions.
- Thioethers are the sulfur analogs of ethers.
- Sulfonium ions have three groups attached to a cationic sulfur center. Dimethylsulfoniopropionate (DMSP) is one such compound, important in the marine organic sulfur cycle.
- Sulfoxides and sulfones are thioethers with one and two oxygen atoms attached to the sulfur atom, respectively. The simplest sulfoxide, dimethyl sulfoxide, is a common solvent; a common sulfone is sulfolane.
- Sulfonic acids are used in many detergents.
Compounds with carbon–sulfur multiple bonds are uncommon, an exception being carbon disulfide, a volatile colorless liquid that is structurally similar to carbon dioxide. It is used as a reagent to make the polymer rayon and many organosulfur compounds. Unlike carbon monoxide, carbon monosulfide is stable only as an extremely dilute gas, found between solar systems.[54]
Organosulfur compounds are responsible for some of the unpleasant odors of decaying organic matter. They are widely known as the
Sulfur–sulfur bonds are a structural component used to stiffen rubber, similar to the disulfide bridges that rigidify proteins (see biological below). In the most common type of industrial "curing" or hardening and strengthening of natural
History
Antiquity

Being abundantly available in native form, sulfur was known in ancient times and is referred to in the
A natural form of sulfur known as shiliuhuang (石硫黄) was known in China since the 6th century BC and found in
Indian alchemists, practitioners of the "science of chemicals" (
Early
Modern times

Sulfur appears in a column of fixed (non-acidic) alkali in a chemical table of 1718.[68] Antoine Lavoisier used sulfur in combustion experiments, writing of some of these in 1777.[69]
Sulfur deposits in
In 1867, elemental sulfur was discovered in underground deposits in Louisiana and Texas. The highly successful Frasch process was developed to extract this resource.[72]
In the late 18th century, furniture makers used molten sulfur to produce decorative inlays.[73] Molten sulfur is sometimes still used for setting steel bolts into drilled concrete holes where high shock resistance is desired for floor-mounted equipment attachment points. Pure powdered sulfur was used as a medicinal tonic and laxative.[45]
With the advent of the contact process, the majority of sulfur today is used to make sulfuric acid for a wide range of uses, particularly fertilizer.[74]
In recent times, the main source of sulfur has become petroleum and natural gas. This is due to the requirement to remove sulfur from fuels in order to prevent acid rain, and has resulted in a surplus of sulfur.[9]
Spelling and etymology
Sulfur is derived from the Latin word sulpur, which was Hellenized to sulphur in the erroneous belief that the Latin word came from Greek. This spelling was later reinterpreted as representing an /f/ sound and resulted in the spelling sulfur, which appears in Latin toward the end of the Classical period. The true Ancient Greek word for sulfur, θεῖον, theîon (from earlier θέειον, théeion), is the source of the international chemical prefix thio-. The Modern Standard Greek word for sulfur is θείο, theío.
In 12th-century Anglo-French, it was sulfre. In the 14th century, the erroneously Hellenized Latin -ph- was restored in Middle English sulphre. By the 15th century, both full Latin spelling variants sulfur and sulphur became common in English. The parallel f~ph spellings continued in Britain until the 19th century, when the word was standardized as sulphur.[75] On the other hand, sulfur was the form chosen in the United States, whereas Canada uses both.
The IUPAC adopted the spelling sulfur in 1990 or 1971, depending on the source cited,[76] as did the Nomenclature Committee of the Royal Society of Chemistry in 1992, restoring the spelling sulfur to Britain.[77] Oxford Dictionaries note that "in chemistry and other technical uses ... the -f- spelling is now the standard form for this and related words in British as well as US contexts, and is increasingly used in general contexts as well."[78]
Production


Sulfur may be found by itself and historically was usually obtained in this form; pyrite has also been a source of sulfur.[79] In volcanic regions in Sicily, in ancient times, it was found on the surface of the Earth, and the "Sicilian process" was used: sulfur deposits were piled and stacked in brick kilns built on sloping hillsides, with airspaces between them. Then, some sulfur was pulverized, spread over the stacked ore and ignited, causing the free sulfur to melt down the hills. Eventually the surface-borne deposits played out, and miners excavated veins that ultimately dotted the Sicilian landscape with labyrinthine mines. Mining was unmechanized and labor-intensive, with pickmen freeing the ore from the rock, and mine-boys or carusi carrying baskets of ore to the surface, often through a mile or more of tunnels. Once the ore was at the surface, it was reduced and extracted in smelting ovens. The conditions in Sicilian sulfur mines were horrific, prompting Booker T. Washington to write "I am not prepared just now to say to what extent I believe in a physical hell in the next world, but a sulfur mine in Sicily is about the nearest thing to hell that I expect to see in this life."[80] Sulfur is still mined from surface deposits in poorer nations with volcanoes, such as Indonesia, and worker conditions have not improved much since Booker T. Washington's days.[81]
Elemental sulfur was extracted from salt domes (in which it sometimes occurs in nearly pure form) until the late 20th century. Sulfur is now produced as a side product of other industrial processes such as in oil refining, in which sulfur is undesired. As a mineral, native sulfur under salt domes is thought to be a fossil mineral resource, produced by the action of anaerobic bacteria on sulfate deposits. It was removed from such salt-dome mines mainly by the Frasch process.[45] In this method, superheated water was pumped into a native sulfur deposit to melt the sulfur, and then compressed air returned the 99.5% pure melted product to the surface. Throughout the 20th century this procedure produced elemental sulfur that required no further purification. Due to a limited number of such sulfur deposits and the high cost of working them, this process for mining sulfur has not been employed in a major way anywhere in the world since 2002.[82][83]

Today, sulfur is produced from petroleum,
The resulting hydrogen sulfide from this process, and also as it occurs in natural gas, is converted into elemental sulfur by the Claus process. This process entails oxidation of some hydrogen sulfide to sulfur dioxide and then the comproportionation of the two:[82][83]
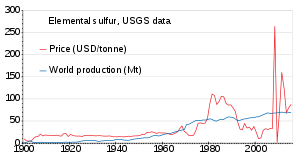
Owing to the high sulfur content of the
The world production of sulfur in 2011 amounted to 69 million tonnes (Mt), with more than 15 countries contributing more than 1 Mt each. Countries producing more than 5 Mt are China (9.6), the United States (8.8), Canada (7.1) and Russia (7.1).[86] Production has been slowly increasing from 1900 to 2010; the price was unstable in the 1980s and around 2010.[87]
Applications
Sulfuric acid
Elemental sulfur is used mainly as a precursor to other chemicals. Approximately 85% (1989) is converted to sulfuric acid (H2SO4):

In 2010, the United States produced more sulfuric acid than any other inorganic industrial chemical.[87] The principal use for the acid is the extraction of phosphate ores for the production of fertilizer manufacturing. Other applications of sulfuric acid include oil refining, wastewater processing, and mineral extraction.[45]
Other important sulfur chemistry
Sulfur reacts directly with methane to give
When silver-based photography was widespread, sodium and ammonium thiosulfate were widely used as "fixing agents". Sulfur is a component of gunpowder ("black powder").
Fertilizer
The plants requirement for sulfur equals or exceeds the requirement for phosphorus. It is an essential nutrient for plant growth, root nodule formation of legumes, and immunity and defense systems. Sulfur deficiency has become widespread in many countries in Europe.[89][90][91] Because atmospheric inputs of sulfur continue to decrease, the deficit in the sulfur input/output is likely to increase unless sulfur fertilizers are used. Atmospheric inputs of sulfur decrease because of actions taken to limit acid rains.[92][88]
Fungicide and pesticide

Elemental sulfur is one of the oldest fungicides and
Standard-formulation dusting sulfur is applied to crops with a sulfur duster or from a dusting plane. Wettable sulfur is the commercial name for dusting sulfur formulated with additional ingredients to make it water miscible.[85][93] It has similar applications and is used as a fungicide against mildew and other mold-related problems with plants and soil.
Elemental sulfur powder is used as an "organic" (i.e., "green") insecticide (actually an acaricide) against ticks and mites. A common method of application is dusting the clothing or limbs with sulfur powder.
A diluted solution of
Sulfur candles of almost pure sulfur were burned to
Pharmaceuticals
Sulfur (specifically
Many drugs contain sulfur.
Batteries
Due to their high energy density and the availability of sulfur, there is ongoing research in creating rechargeable lithium–sulfur batteries. Until now, carbonate electrolytes have caused failures in such batteries after a single cycle. In February 2022, researchers at Drexel University have not only created a prototypical battery that lasted 4000 recharge cycles, but also found the first monoclinic gamma sulfur that remained stable below 95 degrees Celsius.[97]
Biological role
Sulfur is an essential component of all living cells. It is the eighth most abundant element in the human body by weight,[98] about equal in abundance to potassium, and slightly greater than sodium and chlorine.[99] A 70 kg (150 lb) human body contains about 140 grams (4.9 oz) of sulfur.[100] The main dietary source of sulfur for humans is sulfur-containing amino-acids,[101] which can be found in plant and animal proteins.[102]
Transferring sulfur between inorganic and biomolecules
In the 1880s, while studying Beggiatoa (a bacterium living in a sulfur rich environment), Sergei Winogradsky found that it oxidized hydrogen sulfide (H2S) as an energy source, forming intracellular sulfur droplets. Winogradsky referred to this form of metabolism as inorgoxidation (oxidation of inorganic compounds).[103] Another contributor, who continued to study it was Selman Waksman.[104] Primitive bacteria that live around deep ocean volcanic vents oxidize hydrogen sulfide for their nutrition, as discovered by Robert Ballard.[10]
Sulfur oxidizers can use as energy sources reduced sulfur compounds, including hydrogen sulfide, elemental sulfur, sulfite, thiosulfate, and various polythionates (e.g., tetrathionate).[105] They depend on enzymes such as sulfur oxygenase and sulfite oxidase to oxidize sulfur to sulfate. Some lithotrophs can even use the energy contained in sulfur compounds to produce sugars, a process known as chemosynthesis. Some bacteria and archaea use hydrogen sulfide in place of water as the electron donor in chemosynthesis, a process similar to photosynthesis that produces sugars and uses oxygen as the electron acceptor. Sulfur-based chemosynthesis may be simplifiedly compared with photosynthesis:
There are bacteria combining these two ways of nutrition:
There are
There are studies pointing that many deposits of native sulfur in places that were the bottom of the ancient oceans have biological origin.[108][109][110] These studies indicate that this native sulfur have been obtained through biological activity, but what is responsible for that (sulfur-oxidizing bacteria or sulfate-reducing bacteria) is still unknown for sure.
Sulfur is absorbed by plants roots from soil as sulfate and transported as a phosphate ester. Sulfate is reduced to sulfide via sulfite before it is incorporated into cysteine and other organosulfur compounds.[111]
While the plants' role in transferring sulfur to animals by food chains is more or less understood, the role of sulfur bacteria is just getting investigated.[112][113]
Protein and organic metabolites
In all forms of life, most of the sulfur is contained in two proteinogenic amino acids (cysteine and methionine), thus the element is present in all proteins that contain these amino acids, as well as in respective peptides.[114] Some of the sulfur is comprised in certain metabolites—many of which are cofactors—and sulfated polysaccharides of connective tissue (chondroitin sulfates, heparin).
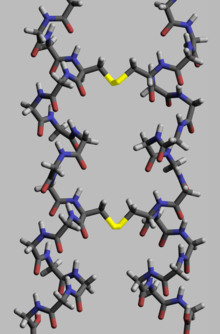
Proteins, to execute their biological function, need to have specific space geometry. Formation of this geometry is performed in a process called protein folding, and is provided by intra- and inter-molecular bonds. The process has several stages. While at premier stages a polypeptide chain folds due to hydrogen bonds, at later stages folding is provided (apart from hydrogen bonds) by covalent bonds between two sulfur atoms of two cysteine residues (so called disulfide bridges) at different places of a chain (tertiary protein structure) as well as between two cysteine residues in two separated protein subunits (quaternary protein structure). Both structures easily may be seen in insulin. As the bond energy of a covalent disulfide bridge is higher than the energy of a coordinate bond or hydrophobic interaction, higher disulfide bridges content leads to higher energy needed for protein denaturation. In general disulfide bonds are necessary in proteins functioning outside cellular space, and they do not change proteins' conformation (geometry), but serve as its stabilizers.[115] Within cytoplasm cysteine residues of proteins are saved in reduced state (i.e. in -SH form) by thioredoxins.[116]
This property manifests in following examples. Lysozyme is stable enough to be applied as a drug.[117] Feathers and hair have relative strength, and consisting in them keratin is considered indigestible by most organisms. However, there are fungi and bacteria containing keratinase, and are able to destruct keratin.
Many important cellular enzymes use prosthetic groups ending with -SH moieties to handle reactions involving acyl-containing biochemicals: two common examples from basic metabolism are
Methanogenesis, the route to most of the world's methane, is a multistep biochemical transformation of carbon dioxide. This conversion requires several organosulfur cofactors. These include coenzyme M, CH3SCH2CH2SO3−, the immediate precursor to methane.[123]
Metalloproteins and inorganic cofactors
Metalloproteins—in which the active site is a transition metal ion (or metal-sulfide cluster) often coordinated by sulfur atoms of cysteine residues
Sulfur is also present in molybdenum cofactor.[127]
Sulfate
Deficiency
In humans methionine is an essential amino acid; cysteine is conditionally essential and may be synthesized from non-essential serine (sulfur donor would be methionine in this case). Dietary deficiency rarely happens in common conditions. Artificial methionine deficiency is attempted to apply in cancer treatment,[128] but the method is still potentially dangerous.[129]
Isolated sulfite oxidase deficiency is a rare, fatal genetic disease preventing production of sulfite oxidase, needed to metabolize sulfites to sulfates.[130]
Precautions
| Hazards | |
|---|---|
| GHS labelling: | |
 
| |
| Warning | |
| H315[131] | |
| NFPA 704 (fire diamond) | |

Though elemental sulfur is only minimally absorbed through the skin and is of low toxicity to humans, inhalation of sulfur dust or contact with eyes or skin may cause irritation. Excessive ingestion of sulfur can cause a burning sensation or diarrhea,[133] and cases of life-threatening metabolic acidosis have been reported after patients deliberately consumed sulfur as a folk remedy.[134][135]
Toxicity of sulfur compounds
When sulfur burns in air, it produces
Sulfur trioxide (made by catalysis from sulfur dioxide) and sulfuric acid are similarly highly acidic and corrosive in the presence of water. Concentrated sulfuric acid is a strong dehydrating agent that can strip available water molecules and water components from sugar and organic tissue.[138]
The burning of
Notes
- organosulfurcompounds.
- ^ A figure of sulfur's melting point in 115.21°C had been determined by two laboratories of the US Department of Energy (Jefferson Lab and Los Alamos National Lab).[12] Greenwood and Earnshaw say that at fast heating for microcrystalline α-S8 the melting point in 115.1 °C (239.2 °F) is specified.[7]
- ^ Historically, it was a rather difficult task to find the exact melting point of sulfur.[15] When heated sluggishly, the factual melting point may lie within the range from 114.6 °C (238.3 °F) or even lower, to 120.4 °C (248.7 °F)[7] (factors, that interfere a definite melting point, is a polymerlike nature of sulfur[16] and a large number of allotropes.[17]) Melting point may be presented as a temperature interval, depending on the allotrope composition of a sample at a moment of melting.
See also
- Blue lava
- Stratospheric sulfur aerosols
- Sulfur assimilation
- Sulfur isotope biogeochemistry
- Ultra-low sulfur diesel
References
- ^ "Standard Atomic Weights: Sulfur". CIAAW. 2009.
- ISSN 1365-3075.
- ^ ISBN 978-1-62708-155-9.
- ISBN 0-8493-0486-5.
- ISBN 0-8493-0464-4.
- ^ "Sulfur History". Georgiagulfsulfur.com. Retrieved 12 February 2022.
- ^ ISBN 0-7506-3365-4.
- ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 4 (11th ed.). Cambridge University Press. p. 571.
- ^ a b c d Laurence Knight (19 July 2014). "Sulphur surplus: Up to our necks in a diabolical element". BBC.
- ^ a b "Sulfur". Elements. BBC. 11 October 2014.. Download here.
- .
- ^ "Sulfur | S (Element) - PubChem". pubchem.ncbi.nlm.nih.gov. Retrieved 15 April 2024.
- ^ ISBN 978-0-08-037941-8.
- ISBN 978-0-07-149999-6.
- ^ "7.5: Changes of State". Chemistry LibreTexts. 3 October 2013. Retrieved 15 April 2024.
- PMID 15473808.
- ISSN 2296-6463.
- ISSN 0019-7866.
- ISSN 0007-0599. Retrieved 23 June 2022.
- doi:10.1021/ic50060a047. Archived from the original(PDF) on 12 April 2019. Retrieved 19 January 2019.
- ISBN 978-0-12-352651-9.
- ISBN 978-3-540-40191-9.
- ISBN 978-3-540-11345-4.
- .
- .
- .
- ^ Sulfur. Commission on Isotopic Abundances and Atomic Weights
- ISBN 1-4398-5511-0.
- ^ "Searching for the Origins of Presolar Grains". Energy.gov. Retrieved 4 February 2023.
- ISBN 9780128243602.
- ^ "NASA Astrobiology". astrobiology.nasa.gov. Retrieved 4 February 2023.
- .
- S2CID 1248182.
- S2CID 258600480.
- ^ Kim, Ik Soo; Kwak, Seung Im; Park, Ul Jae; Bang, Hong Sik; Han, Hyun Soo (1 July 2005). Production of Sulfur-35 by the Cation Exchange Process. 2005 autumn meeting of the KNS, Busan (Korea, Republic of), 27–28 Oct 2005.
- ^ "Sulfur-35 (35 S) safety information and specific handling precautions" (PDF). Yale Environmental Health & Safety.
- ^ Cameron, A. G. W. (1957). "Stellar Evolution, Nuclear Astrophysics, and Nucleogenesis" (PDF). CRL-41.
- ISBN 978-0-908678-84-6.
- S2CID 44208045.
- ^ Rickwood, P. C. (1981). "The largest crystals" (PDF). American Mineralogist. 66: 885–907.
- OCLC 79256100.
- S2CID 199492543.
- ISBN 0-471-80580-7.
- ^ "Sulphur: Mineral information, data and localities". www.mindat.org.
- ^ ISBN 978-3-527-30673-2.
- ^ Shriver, Atkins. Inorganic Chemistry, Fifth Edition. W. H. Freeman and Company, New York, 2010; pp 416
- PMID 23851903.
- ^ Brauer, G., ed. (1963). Handbook of Preparative Inorganic Chemistry. Vol. 1 (2nd ed.). New York: Academic Press. p. 421.
- .
- ^ ISBN 978-3527306732.
- ISBN 0-521-21489-0.
- ISSN 2296-6846.
- ^ ISBN 0-471-95512-4.
- doi:10.1086/182072.
- OCLC 697506461.
- ^ "Sulfur in the Bible (14 instances)". bible.knowing-jesus.com. Retrieved 19 May 2022.
- ISBN 978-3-540-78593-4.
- ^ Odyssey, book 22, lines 480–495. www.perseus.tufts.edu. Retrieved on 16 August 2012.
- ISBN 0-19-814687-6, pp. 247–249.
- ^ S2CID 144187385.
- OCLC 489677531.
- ISBN 0-486-20162-7.
- ISBN 978-0-226-89499-7.
- PMID 2450900.
- PMID 2365870.
- PMID 15303787.
- PMID 15303787.
- ISBN 978-0-521-56672-8.
- ISBN 978-0-8122-1649-3.
- ISBN 9780191542619. Retrieved 7 February 2013.
- S2CID 145807900.
- .
- S2CID 250882259.
- OCLC 62805047.
- ^ "sulphur". Oxford English Dictionary (Online ed.). Oxford University Press. (Subscription or participating institution membership required.)
- PMID 21378874.
- .
- ^ "sulphur – definition of sulphur in English". Oxford Dictionaries. Archived from the original on 20 November 2016. Retrieved 19 November 2016.
- OCLC 74650396.
- ^ Washington, Booker T. (1912). The Man Farthest Down: A Record of Observation and Study in Europe. Doubleday, Page. p. 214.
- ^ McElvaney, Kevin (25 February 2015). "The Men Who Mine Volcanos". The Atlantic. Retrieved 26 February 2015.
- ^ .
- ^ .
- ISBN 978-0-8412-0713-4.
- ^ OCLC 531718953.
- ^ Apodaca, Lori E. (2012) "Sulfur". Mineral Commodity Summaries. United States Geological Survey.
- ^ a b Apodaca, Lori E. "Mineral Yearbook 2010: Sulfur" (PDF). United States Geological Survey.
- ^ a b "FAQ – The Sulphur Institute". sulphurinstitute.org. The Sulphur Institute. 2020. Retrieved 27 February 2020.
- .
- S2CID 44208638.
- S2CID 42207099.
- ^ Glossary, United States: NASA Earth Observatory, acid rain, archived from the original on 13 December 2011, retrieved 15 February 2013
- ^ Every, Richard L.; et al. (20 August 1968). "Method for Preparation of Wettable Sulfur" (PDF). Retrieved 20 May 2010.
- ISBN 978-3-540-07738-1.
- ISBN 978-3-8047-2575-1.
- ISBN 978-3-030-25598-5, retrieved 8 March 2023
- S2CID 246704531.
- ^ "Sulphur and the Human Body". The Sulfur Institute. Retrieved 3 April 2021.
- ^ "What is the body made of?". New Scientist. Archived from the original on 3 November 2021. Retrieved 9 November 2021.
- ThoughtCo. Archivedfrom the original on 13 April 2021. Retrieved 21 November 2021.
- PMID 11896744.
- PMID 23815141.
- PMID 22092289.
- PMID 19871997.
- .
- PMID 18929068, retrieved 17 May 2022
- .
- PMID 17793426.
- S2CID 128827551.
- ^ "Petrography and mineralogy of the crystalline limestone of Fatha Formation from Mishraq area, Iraq". ResearchGate. Retrieved 15 April 2022.
- ISBN 978-3-8274-0103-8.
- PMID 6127737.
- PMID 28419734.
- ^ Gutiérrez-Preciado, A.; Romero, H.; Peimbert, M. (2010). "An Evolutionary Perspective on Amino Acids". Nature Education. 3 (9): 29.
- ISBN 978-0-8153-3218-3.
- PMID 11012661.
- ^ "Lysozyme". Drugs.com. Retrieved 19 May 2022.
- ISBN 978-1-57259-153-0.
- PMID 10448523.
- PMID 1731369.
- ^ "The Function of Biotin". www.chem.uwec.edu. Retrieved 10 June 2022.
- ^ Edwards, Katie A. "Thiamine Biochemistry". thiamine.dnr.cornell.edu. Retrieved 10 June 2022.
- PMID 9782487.
- PMID 24970223.
- PMID 12954327.
- ISBN 978-0-935702-73-6.
- PMID 16669776.
- S2CID 73470156.
- PMID 33673497.
- PMID 16234925.
- ^ "Sulfur 84683". S.
- ^ "Chemical Datasheet. SULFUR, MOLTEN".
- ^ "Sulfur General Fact Sheet". npic.orst.edu. Retrieved 2 September 2022.
- PMID 3718141.
- PMID 904661. Retrieved 2 September 2022.
- ^ "Sulfur Dioxide | Toxicological Profile | ATSDR". Centers for Disease Control and Prevention. 26 March 2014. Retrieved 24 October 2023.
- ^ "Sulfur General Fact Sheet". npic.orst.edu. Retrieved 20 January 2024.
- ^ Baker, Colin (1 March 2007). "The dehydration of sucrose". Education in Chemistry. Royal Society of Chemistry. Retrieved 14 June 2018.
- ^ "4.1: Fossil Fuels and Products of Combustion". Engineering LibreTexts. 28 February 2021. Retrieved 24 October 2023.
- ^ "Hydrogen Sulfide Toxicity: Practice Essentials, Pathophysiology, Etiology". Medscape. 30 March 2017 – via eMedicine.
- ^ Summers, Vincent (8 April 2017). "Hydrogen Sulfide or Hydrogen Cyanide: Which is More Dangerous?". Quirky Science. Retrieved 23 August 2022.
- ^ "Hydrogen Sulfide – Hazards | Occupational Safety and Health Administration". www.osha.gov. Retrieved 23 August 2022.
Further reading
Sigel, Astrid; Freisinger, Eva; Sigel, Roland K.O., eds. (2020). Transition Metals and Sulfur: A Strong Relationship for Life. Guest Editors Martha E Sosa Torres and Peter M.H.Kroneck. Berlin/Boston: de Gruyter. pp. xlv+455.
External links
- Sulfur at The Periodic Table of Videos(University of Nottingham)
- Atomic Data for Sulfur, NISTPhysical Measurement Laboratory
- Sulfur phase diagram Archived 23 February 2010 at the Wayback Machine, Introduction to Chemistry for Ages 13–17
- Crystalline, liquid and polymerization of sulfur on Vulcano Island, Italy
- Sulfur and its use as a pesticide
- The Sulphur Institute
- Nutrient Stewardship and The Sulphur Institute