Sulfur dioxide

| |
| |

| |
| Names | |
|---|---|
| IUPAC name
Sulfur dioxide
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 3535237 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.028.359 |
| EC Number |
|
| E number | E220 (preservatives) |
| 1443 | |
| KEGG | |
| MeSH | Sulfur+dioxide |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1079, 2037 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| SO 2 | |
| Molar mass | 64.066 g/mol |
| Appearance | Colorless and pungent gas |
| Odor | Pungent; similar to a just-struck match[1] |
| Density | 2.619 kg m−3[2] |
| Melting point | −72 °C; −98 °F; 201 K |
| Boiling point | −10 °C (14 °F; 263 K) |
| 94 g/L[3] forms sulfurous acid | |
| Vapor pressure | 230 kPa at 10 °C; 330 kPa at 20 °C; 462 kPa at 30 °C; 630 kPa at 40 °C[4] |
| Acidity (pKa) | ~1.81 |
| Basicity (pKb) | ~12.19 |
| −18.2·10−6 cm3/mol | |
| Viscosity | 12.82 μPa·s[5] |
| Structure | |
| C2v | |
| Digonal | |
| Dihedral | |
| 1.62 D | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
248.223 J K−1 mol−1 |
Std enthalpy of (ΔfH⦵298)formation |
−296.81 kJ mol−1 |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H314, H331[6] | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LC50 (median concentration)
|
3000 ppm (mouse, 30 min) 2520 ppm (rat, 1 hr)[8] |
LCLo (lowest published)
|
993 ppm (rat, 20 min) 611 ppm (rat, 5 hr) 764 ppm (mouse, 20 min) 1000 ppm (human, 10 min) 3000 ppm (human, 5 min)[8] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 5 ppm (13 mg/m3)[7] |
REL (Recommended)
|
TWA 2 ppm (5 mg/m3) ST 5 ppm (13 mg/m3)[7] |
IDLH (Immediate danger) |
100 ppm[7] |
| Related compounds | |
| Sulfur monoxide Sulfur trioxide Disulfur monoxide | |
Related compounds
|
Ozone Selenium dioxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sulfur dioxide (
Structure and bonding
SO2 is a bent molecule with C2v symmetry point group. A valence bond theory approach considering just s and p orbitals would describe the bonding in terms of resonance between two resonance structures.

The sulfur–oxygen bond has a bond order of 1.5. There is support for this simple approach that does not invoke d orbital participation.[11] In terms of electron-counting formalism, the sulfur atom has an oxidation state of +4 and a formal charge of +1.
Occurrence

Sulfur dioxide is found on Earth and exists in very small concentrations in the atmosphere at about 15 ppb.[12]
On other planets, sulfur dioxide can be found in various concentrations, the most significant being the
As an ice, it is thought to exist in abundance on the Galilean moons—as subliming ice or frost on the trailing hemisphere of Io,[17] and in the crust and mantle of Europa, Ganymede, and Callisto, possibly also in liquid form and readily reacting with water.[18]
Production
Sulfur dioxide is primarily produced for sulfuric acid manufacture (see contact process, but other processes predated that at least since 16th century[10]). In the United States in 1979, 23.6 million metric tons (26 million U.S. short tons) of sulfur dioxide were used in this way, compared with 150,000 metric tons (165,347 U.S. short tons) used for other purposes. Most sulfur dioxide is produced by the combustion of elemental sulfur. Some sulfur dioxide is also produced by roasting pyrite and other sulfide ores in air.[19]
Combustion routes
Sulfur dioxide is the product of the burning of sulfur or of burning materials that contain sulfur:
- 1⁄8 S8 + O2 → SO2, ΔH = −297 kJ/mol
To aid combustion, liquified sulfur (140–150 °C, 284-302 °F) is sprayed through an atomizing nozzle to generate fine drops of sulfur with a large surface area. The reaction is
The combustion of hydrogen sulfide and organosulfur compounds proceeds similarly. For example:
- H2S + 3⁄2 O2 → SO2 + H2O
The roasting of sulfide ores such as pyrite, sphalerite, and cinnabar (mercury sulfide) also releases SO2:[20]
- 2 FeS2 + 11⁄2 O2 → Fe2O3 + 4 SO2
- ZnS + 3⁄2 O2 → ZnO + SO2
- HgS + O2 → Hg + SO2
- 2 FeS + 7⁄2 O2 → Fe2O3 + 2 SO2
A combination of these reactions is responsible for the largest source of sulfur dioxide, volcanic eruptions. These events can release millions of tons of SO2.
Reduction of higher oxides
Sulfur dioxide can also be a byproduct in the manufacture of calcium silicate cement; CaSO4 is heated with coke and sand in this process:
- 2 CaSO4 + 2 SiO2 + C → 2 CaSiO3 + 2 SO2 + CO2
Until the 1970s, commercial quantities of sulfuric acid and cement were produced by this process in Whitehaven, England. Upon being mixed with shale or marl, and roasted, the sulfate liberated sulfur dioxide gas, used in sulfuric acid production, the reaction also produced calcium silicate, a precursor in cement production.[21]
On a laboratory scale, the action of hot concentrated sulfuric acid on copper turnings produces sulfur dioxide.
- Cu + 2 H2SO4 → CuSO4 + SO2 + 2 H2O
Tin also reacts with concentrated sulfuric acid but it produces tin(II) sulfate which can later be pyrolyzed at 360°C into tin dioxide and dry sulfur dioxide.
- Sn + H2SO4 → SnSO4 + H2
- SnSO4 → SnO2 + SO2
From sulfites
The reverse reaction occurs upon acidification:
- H+ + HSO−3 → SO2 + H2O
Reactions
Sulfites results by the action of aqueous base on sulfur dioxide:
- SO2 + 2 NaOH → Na2SO3 + H2O
Sulfur dioxide is a mild but useful reducing agent. It is oxidized by halogens to give the sulfuryl halides, such as sulfuryl chloride:
- SO2 + Cl2 → SO2Cl2
Sulfur dioxide is the
- SO2 + 2 H2S → 3 S + 2 H2O
The sequential oxidation of sulfur dioxide followed by its hydration is used in the production of sulfuric acid.
- SO2 + H2O + 1⁄2 O2 → H2SO4
Sulfur dioxide dissolves in water to give "sulfurous acid", which cannot be isolated and is instead an acidic solution of bisulfite, and possibly sulfite, ions.
- SO2 + H2O ⇌ HSO−3 + H+ Ka = 1.54×10−2; pKa = 1.81
Laboratory reactions
Sulfur dioxide is one of the few common acidic yet reducing gases. It turns moist litmus pink (being acidic), then white (due to its bleaching effect). It may be identified by bubbling it through a
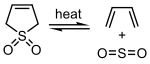
Sulfur dioxide can react with certain 1,3-dienes in a cheletropic reaction to form cyclic sulfones. This reaction is exploited on an industrial scale for the synthesis of sulfolane, which is an important solvent in the petrochemical industry.
Sulfur dioxide can bind to metal ions as a
Uses
The overarching, dominant use of sulfur dioxide is in the production of sulfuric acid.[19]
Precursor to sulfuric acid
Sulfur dioxide is an intermediate in the production of sulfuric acid, being converted to sulfur trioxide, and then to oleum, which is made into sulfuric acid. Sulfur dioxide for this purpose is made when sulfur combines with oxygen. The method of converting sulfur dioxide to sulfuric acid is called the contact process. Several million tons are produced annually for this purpose.
Food preservative
Sulfur dioxide is sometimes used as a
Winemaking
Sulfur dioxide was first used in winemaking by the Romans, when they discovered that burning sulfur candles inside empty wine vessels keeps them fresh and free from vinegar smell.[26]
It is still an important compound in winemaking, and is measured in
Sulfur dioxide exists in wine in free and bound forms, and the combinations are referred to as total SO2. Binding, for instance to the carbonyl group of acetaldehyde, varies with the wine in question. The free form exists in equilibrium between molecular SO2 (as a dissolved gas) and bisulfite ion, which is in turn in equilibrium with sulfite ion. These equilibria depend on the pH of the wine. Lower pH shifts the equilibrium towards molecular (gaseous) SO2, which is the active form, while at higher pH more SO2 is found in the inactive sulfite and bisulfite forms. The molecular SO2 is active as an antimicrobial and antioxidant, and this is also the form which may be perceived as a pungent odor at high levels. Wines with total SO2 concentrations below 10 ppm do not require "contains sulfites" on the label by US and EU laws. The upper limit of total SO2 allowed in wine in the US is 350 ppm; in the EU it is 160 ppm for red wines and 210 ppm for white and rosé wines. In low concentrations, SO2 is mostly undetectable in wine, but at free SO2 concentrations over 50 ppm, SO2 becomes evident in the smell and taste of wine.[citation needed]
SO2 is also a very important compound in winery sanitation. Wineries and equipment must be kept clean, and because bleach cannot be used in a winery due to the risk of cork taint,[30] a mixture of SO2, water, and citric acid is commonly used to clean and sanitize equipment. Ozone (O3) is now used extensively for sanitizing in wineries due to its efficacy, and because it does not affect the wine or most equipment.[31]
As a reducing agent
Sulfur dioxide is also a good reductant. In the presence of water, sulfur dioxide is able to decolorize substances. Specifically, it is a useful reducing bleach for papers and delicate materials such as clothes. This bleaching effect normally does not last very long. Oxygen in the atmosphere reoxidizes the reduced dyes, restoring the color. In municipal wastewater treatment, sulfur dioxide is used to treat chlorinated wastewater prior to release. Sulfur dioxide reduces free and combined chlorine to chloride.[32]
Sulfur dioxide is fairly soluble in water, and by both IR and Raman spectroscopy; the hypothetical sulfurous acid, H2SO3, is not present to any extent. However, such solutions do show spectra of the hydrogen sulfite ion, HSO3−, by reaction with water, and it is in fact the actual reducing agent present:
- SO2 + H2O ⇌ HSO3− + H+
As a fumigant
In the beginning of the 20th century, sulfur dioxide was used in Buenos Aires as a fumigant to kill rats that carried the Yersinia pestis bacterium, which causes bubonic plague. The application was successful, and the application of this method was extended to other areas in South America. In Buenos Aires, where these apparatuses were known as Sulfurozador, but later also in Rio de Janeiro, New Orleans and San Francisco, the sulfur dioxide treatment machines were brought into the streets to enable extensive disinfection campaigns, with effective results.[33]
Biochemical and biomedical roles
Sulfur dioxide or its conjugate base bisulfite is produced biologically as an intermediate in both sulfate-reducing organisms and in sulfur-oxidizing bacteria, as well. The role of sulfur dioxide in mammalian biology is not yet well understood.[34] Sulfur dioxide blocks nerve signals from the pulmonary stretch receptors and abolishes the Hering–Breuer inflation reflex.
It is considered that endogenous sulfur dioxide plays a significant physiological role in regulating
It was shown that in children with pulmonary arterial hypertension due to congenital heart diseases the level of homocysteine is higher and the level of endogenous sulfur dioxide is lower than in normal control children. Moreover, these biochemical parameters strongly correlated to the severity of pulmonary arterial hypertension. Authors considered homocysteine to be one of useful biochemical markers of disease severity and sulfur dioxide metabolism to be one of potential therapeutic targets in those patients.[36]
Endogenous sulfur dioxide also has been shown to lower the
Endogenous sulfur dioxide in low concentrations causes endothelium-dependent vasodilation. In higher concentrations it causes endothelium-independent vasodilation and has a negative inotropic effect on cardiac output function, thus effectively lowering blood pressure and myocardial oxygen consumption. The vasodilating and bronchodilating effects of sulfur dioxide are mediated via ATP-dependent calcium channels and L-type ("dihydropyridine") calcium channels. Endogenous sulfur dioxide is also a potent antiinflammatory, antioxidant and cytoprotective agent. It lowers blood pressure and slows hypertensive remodeling of blood vessels, especially thickening of their intima. It also regulates lipid metabolism.[38]
Endogenous sulfur dioxide also diminishes myocardial damage, caused by
As a reagent and solvent in the laboratory
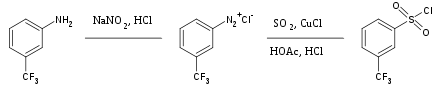
Sulfur dioxide is a versatile inert solvent widely used for dissolving highly oxidizing salts. It is also used occasionally as a source of the sulfonyl group in
As a result of its very low
As a refrigerant
Being easily condensed and possessing a high
Safety

Ingestion
In the United States, the Center for Science in the Public Interest lists the two food preservatives, sulfur dioxide and sodium bisulfite, as being safe for human consumption except for certain asthmatic individuals who may be sensitive to them, especially in large amounts.[42] Symptoms of sensitivity to sulfiting agents, including sulfur dioxide, manifest as potentially life-threatening trouble breathing within minutes of ingestion.[43] Sulphites may also cause symptoms in non-asthmatic individuals, namely dermatitis, urticaria, flushing, hypotension, abdominal pain and diarrhea, and even life-threatening anaphylaxis.[44]
Inhalation
Incidental exposure to sulfur dioxide is routine, e.g. the smoke from
In 2008, the
Environmental role
Air pollution

Major

Such an increase in sulfate aerosol emissions had a variety of effects. At the time, the most visible one was acid rain, caused by precipitation from clouds carrying high concentrations of sulfate aerosols in the troposphere.[54] At its peak, acid rain has eliminated brook trout and some other fish species and insect life from lakes and streams in geographically sensitive areas, such as Adirondack Mountains in the United States.[55] Acid rain worsens soil function as some of its microbiota is lost and heavy metals like aluminium are mobilized (spread more easily) while essential nutrients and minerals such as magnesium can leach away because of the same. Ultimately, plants unable to tolerate lowered pH are killed, with montane forests being some of the worst-affected ecosystems due to their regular exposure to sulfate-carrying fog at high altitudes.[56][57][58][59][60] While acid rain was too dilute to affect human health directly, breathing smog or even any air with elevated sulfate concentrations is known to contribute to heart and lung conditions, including asthma and bronchitis.[51] Further, this form of pollution is linked to preterm birth and low birth weight, with a study of 74,671 pregnant women in Beijing finding that every additional 100 µg/m3 of SO2 in the air reduced infants' weight by 7.3 g, making it and other forms of air pollution the largest attributable risk factor for low birth weight ever observed.[61]
Control measures

Due largely to the US EPA's
.| Year | SO2 |
|---|---|
| 1970 | 31,161,000 short tons (28.3 Mt) |
| 1980 | 25,905,000 short tons (23.5 Mt) |
| 1990 | 23,678,000 short tons (21.5 Mt) |
| 1996 | 18,859,000 short tons (17.1 Mt) |
| 1997 | 19,363,000 short tons (17.6 Mt) |
| 1998 | 19,491,000 short tons (17.7 Mt) |
| 1999 | 18,867,000 short tons (17.1 Mt) |
In particular, calcium oxide (lime) reacts with sulfur dioxide to form calcium sulfite:
- CaO + SO2 → CaSO3
Aerobic oxidation of the CaSO3 gives CaSO4, anhydrite. Most gypsum sold in Europe comes from flue-gas desulfurization.
To control sulfur emissions, dozens of methods with relatively high efficiencies have been developed for fitting of coal-fired power plants.[63] Sulfur can be removed from coal during burning by using limestone as a bed material in fluidized bed combustion.[64]
Sulfur can also be removed from fuels before burning, preventing formation of SO2 when the fuel is burnt. The Claus process is used in refineries to produce sulfur as a byproduct. The Stretford process has also been used to remove sulfur from fuel. Redox processes using iron oxides can also be used, for example, Lo-Cat[65] or Sulferox.[66]
Fuel additives such as calcium additives and magnesium carboxylate may be used in marine engines to lower the emission of sulfur dioxide gases into the atmosphere.[67]
Impact on climate change

Projected impacts

Since changes in aerosol concentrations already have an impact on the global climate, they would necessarily influence future projections as well. In fact, it is impossible to fully estimate the warming impact of all greenhouse gases without accounting for the counteracting cooling from aerosols.[77][78]
Regardless of the current strength of aerosol cooling, all futureSolar geoengineering
Properties
Table of thermal and physical properties of saturated liquid sulfur dioxide:[98][99]
| Temperature (°C) | Density (kg/m^3) | Specific heat (kJ/kg K) | Kinematic viscosity (m^2/s) | Conductivity (W/m K) | Thermal diffusivity (m^2/s) | Prandtl Number | Bulk modulus (K^-1) |
| -50 | 1560.84 | 1.3595 | 4.84E-07 | 0.242 | 1.14E-07 | 4.24 | - |
| -40 | 1536.81 | 1.3607 | 4.24E-07 | 0.235 | 1.13E-07 | 3.74 | - |
| -30 | 1520.64 | 1.3616 | 3.71E-07 | 0.23 | 1.12E-07 | 3.31 | - |
| -20 | 1488.6 | 1.3624 | 3.24E-07 | 0.225 | 1.11E-07 | 2.93 | - |
| -10 | 1463.61 | 1.3628 | 2.88E-07 | 0.218 | 1.10E-07 | 2.62 | - |
| 0 | 1438.46 | 1.3636 | 2.57E-07 | 0.211 | 1.08E-07 | 2.38 | - |
| 10 | 1412.51 | 1.3645 | 2.32E-07 | 0.204 | 1.07E-07 | 2.18 | - |
| 20 | 1386.4 | 1.3653 | 2.10E-07 | 0.199 | 1.05E-07 | 2 | 1.94E-03 |
| 30 | 1359.33 | 1.3662 | 1.90E-07 | 0.192 | 1.04E-07 | 1.83 | - |
| 40 | 1329.22 | 1.3674 | 1.73E-07 | 0.185 | 1.02E-07 | 1.7 | - |
| 50 | 1299.1 | 1.3683 | 1.62E-07 | 0.177 | 9.99E-08 | 1.61 | - |
See also
References
- ^ Sulfur dioxide Archived 2019-12-30 at the Wayback Machine, U.S. National Library of Medicine
- ^ "Sulfur Dioxide". Archived from the original on 2023-09-24. Retrieved 2024-03-22., U.S. National Library of Medicine
- ISBN 0-8493-0487-3.
- ^ "Hazardous Substances Data Bank".
- ISSN 0009-2460.
- ^ "C&L Inventory".
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0575". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Sulfur dioxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ ISBN 978-0-08-037941-8.
- ^ ISBN 978-0-19-965272-3.
- doi:10.1039/A700708F.
- ^ US EPA O (2016-05-04). "Sulfur Dioxide Trends". www.epa.gov. Retrieved 2023-02-16.
- S2CID 59323909.
- S2CID 7246517.
- ISBN 978-3-540-34681-4.
- ^ "James Webb Space Telescope reveals an exoplanet atmosphere as never seen before".
- ISBN 978-94-010-8891-6.
- ^ Europa's Hidden Ice Chemistry – NASA Jet Propulsion Laboratory. Jpl.nasa.gov (2010-10-04). Retrieved on 2013-09-24.
- ^ ISBN 978-3527306732.
- ^ Shriver, Atkins. Inorganic Chemistry, Fifth Edition. W. H. Freeman and Company; New York, 2010; p. 414.
- ^ WHITEHAVEN COAST ARCHAEOLOGICAL SURVEY. lakestay.co.uk (2007)
- ^ "Information archivée dans le Web" (PDF).
- ISBN 978-85-99141-03-8. Archived from the original(PDF) on 2020-06-04. Retrieved 2020-06-04.
- ^ Current EU approved additives and their E Numbers, The Food Standards Agency website.
- ^ a b Preserving foods: Drying fruits and Vegetable (PDF), University of Georgia cooperative extension service, archived from the original (PDF) on 2022-09-27, retrieved 2022-06-06
- ^ "Practical Winery & vineyard Journal Jan/Feb 2009". www.practicalwinery.com. 1 Feb 2009. Archived from the original on 2013-09-28.
- ^ Sulphites in wine, MoreThanOrganic.com.
- ^ Jackson, R.S. (2008) Wine science: principles and applications, Amsterdam; Boston: Elsevier/Academic Press
- .
- ^ Chlorine Use in the Winery. Purdue University
- ^ Use of ozone for winery and environmental sanitation Archived 2017-09-12 at the Wayback Machine, Practical Winery & Vineyard Journal.
- ISBN 0-07-041677-X.
- PMID 29886876.
- PMID 20540708.
- S2CID 11924999.
- PMID 25224243.
- PMID 24853429.
- PMID 21925165.
- PMID 20733562.
- ^ Hoffman, R. V. (1990). "m-Trifluoromethylbenzenesulfonyl Chloride". Organic Syntheses; Collected Volumes, vol. 7, p. 508.
- ISSN 0002-7863.
- ^ "Center for Science in the Public Interest – Chemical Cuisine". Retrieved March 17, 2010.
- ^ "California Department of Public Health: Food and Drug Branch: Sulfites" (PDF). Archived from the original (PDF) on July 23, 2012. Retrieved September 27, 2013.
- PMID 24834193.
- ^ Sulfur Dioxide Basics U.S. Environmental Protection Agency
- ^ a b Sulfur Dioxide (SO2) Pollution. United States Environmental Protection Agency
- ^ "NIOSH Pocket Guide to Chemical Hazards".
- ^ "Volcanic Sulfur Aerosols Affect Climate and the Earth's Ozone Layer". United States Geological Survey. Archived from the original on 14 November 2015. Retrieved 17 February 2009.
- .
- ^ IPCC, 1990: Chapter 1: Greenhouse Gases and Aerosols [R.T. Watson, H. Rodhe, H. Oeschger and U. Siegenthaler]. In: Climate Change: The IPCC Scientific Assessment [J.T.Houghton, G.J.Jenkins and J.J.Ephraums (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 31–34,
- ^ a b Effects of Acid Rain – Human Health Archived January 18, 2008, at the Wayback Machine. Epa.gov (June 2, 2006). Retrieved on 2013-02-09.
- ^ Henneman L, Choirat C, Dedoussi I, Dominici F, Roberts J, Zigler C (24 November 2023). "Mortality risk from United States coal electricity generation". Science. 382 (6673): 941.
- S2CID 55497518.
- .
- ^ "Effects of Acid Rain - Surface Waters and Aquatic Animals". US EPA. Archived from the original on 14 May 2009.
- PMID 12387412.
- ^ US EPA: Effects of Acid Rain – Forests Archived July 26, 2008, at the Wayback Machine
- (PDF) from the original on December 24, 2012. Retrieved February 9, 2013.
- from the original on March 30, 2015. Retrieved April 22, 2020.
- ISSN 1944-7973.
- S2CID 2707126.
- PMID 30518902.
- PMID 29422539.
- ISBN 978-1-59126-049-3.
- ^ FAQ's About Sulfur Removal and Recovery using the LO-CAT® Hydrogen Sulfide Removal System. gtp-merichem.com
- ^ Process screening analysis of alternative gas treating and sulfur removal for gasification. (December 2002) Report by SFA Pacific, Inc. prepared for U.S. Department of Energy (PDF) Retrieved on 2011-10-31.
- ^ May, Walter R. Marine Emissions Abatement Archived 2015-04-02 at the Wayback Machine. SFA International, Inc., p. 6.
- ^ .
- S2CID 154006620.
- .
- .
- .
- S2CID 4320853.
Spacecraft measurements have established that the total radiative output of the Sun varies at the 0.1−0.3% level
- ^ a b "Aerosol pollution has caused decades of global dimming". American Geophysical Union. 18 February 2021. Archived from the original on 27 March 2023. Retrieved 18 December 2023.
- ^ Adam D (18 December 2003). "Goodbye sunshine". The Guardian. Retrieved 26 August 2009.
- S2CID 233645438.
- ^ PMID 30518902.
- PMID 32734279.
- .
- ^ Hausfather Z (29 April 2021). "Explainer: Will global warming 'stop' as soon as net-zero emissions are reached?". Carbon Brief. Retrieved 3 March 2023.
- S2CID 250077615.
- NOAA. Retrieved 14 October 2022.
- Nature. Retrieved 14 October 2022.
- PMID 34219915.
- PMID 35297408.
- S2CID 252446168.
- S2CID 229506768.
- ^ Palumbo, P., A. Rotundi, V. Della Corte, A. Ciucci, L. Colangeli, F. Esposito, E. Mazzotta Epifani, V. Mennella , J.R. Brucato, F.J.M. Rietmeijer, G. J. Flynn, J.-B. Renard, J.R. Stephens, and E. Zona. "The DUSTER experiment: collection and analysis of aerosol in the high stratosphere". Retrieved 19 February 2009.
{{cite web}}: CS1 maint: multiple names: authors list (link)[permanent dead link] - S2CID 55623817.
- .
- ^ "Aerosols and Incoming Sunlight (Direct Effects)". NASA. 2 November 2010.
- ^ "Stratospheric Injections Could Help Cool Earth, Computer Model Shows". ScienceDaily. 15 September 2006. Retrieved 19 February 2009.
- PMID 18757275.
- .
- ISSN 1748-9326.
- ^ Andrew Charlton-Perez, Eleanor Highwood. "Costs and benefits of geo-engineering in the Stratosphere" (PDF). Archived from the original (PDF) on 14 January 2017. Retrieved 17 February 2009.
- .
- ISBN 9780072406559.
- ISBN 9780471457282.
External links
- Global map of sulfur dioxide distribution
- United States Environmental Protection Agency Sulfur Dioxide page
- International Chemical Safety Card 0074
- IARC Monographs. "Sulfur Dioxide and some Sulfites, Bisulfites and Metabisulfites". vol. 54. 1992. p. 131.
- NIOSH Pocket Guide to Chemical Hazards
- CDC – Sulfure Dioxide – NIOSH Workplace Safety and Health Topic
- Sulfur Dioxide, Molecule of the Month