Tannin
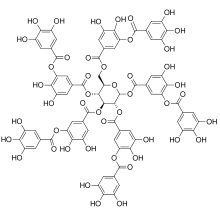


Tannins (or tannoids) are a class of astringent, polyphenolic biomolecules that bind to and precipitate proteins and various other organic compounds including amino acids and alkaloids.
The term tannin (from
The tannin compounds are widely distributed in many species of plants, where they play a role in protection from
Tannins have
Structure and classes of tannins
There are three major classes of tannins: Shown below are the base unit or monomer of the tannin. Particularly in the flavone-derived tannins, the base shown must be (additionally) heavily hydroxylated and polymerized in order to give the high molecular weight polyphenol motif that characterizes tannins. Typically, tannin molecules require at least 12 hydroxyl groups and at least five phenyl groups to function as protein binders.[4]
| Base unit / scaffold |  Gallic acid |
 Phloroglucinol |
 Flavan-3-ol | |
|---|---|---|---|---|
| Polymer class | Hydrolyzable tannins
|
Phlorotannins | Condensed tannins[5] | Phlobatannins (C-ring isomerized condensed tannins)[5]
|
| Sources | Plants | Brown algae | Plants | Tree heartwood |
Oligostilbenoids (oligo- or polystilbenes) are oligomeric forms of stilbenoids and constitute a minor class of tannins.[6]
Pseudo-tannins
Pseudo-tannins are low molecular weight compounds associated with other compounds. They do not change color during the Goldbeater's skin test, unlike hydrolysable and condensed tannins, and cannot be used as tanning compounds.[4] Some examples of pseudo tannins and their sources are:[7]
| Pseudo tannin | Source(s) |
|---|---|
| Gallic acid | Rhubarb |
| Flavan-3-ols (Catechins) | Tea, acacia, catechu, cocoa, guarana |
| Chlorogenic acid | mate
|
| Ipecacuanhic acid | Carapichea ipecacuanha |
History
At these times, molecule formulas were determined through
In 1966, Edwin Haslam proposed a first comprehensive definition of plant polyphenols based on the earlier proposals of Bate-Smith, Swain and Theodore White, which includes specific structural characteristics common to all phenolics having a tanning property. It is referred to as the White–Bate-Smith–Swain–Haslam (WBSSH) definition.[17][self-published source?]
Occurrence
Tannins are distributed in species throughout the
The most abundant polyphenols are the condensed tannins, found in virtually all families of plants, and comprising up to 50% of the dry weight of leaves.[19][20]
Cellular localization
This section needs additional citations for verification. (September 2021) |
In all vascular plants studied, tannins are manufactured by a chloroplast-derived organelle, the tannosome.[21] Tannins are mainly physically located in the vacuoles or surface wax of plants. These storage sites keep tannins active against plant predators, but also keep some tannins from affecting plant metabolism while the plant tissue is alive.
Tannins are classified as ergastic substances, i.e., non-protoplasm materials found in cells. Tannins, by definition, precipitate proteins. In this condition, they must be stored in organelles able to withstand the protein precipitation process. Idioblasts are isolated plant cells which differ from neighboring tissues and contain non-living substances. They have various functions such as storage of reserves, excretory materials, pigments, and minerals. They could contain oil, latex, gum, resin or pigments etc. They also can contain tannins. In Japanese persimmon (Diospyros kaki) fruits, tannin is accumulated in the vacuole of tannin cells, which are idioblasts of parenchyma cells in the flesh.[22]
Presence in soils
The convergent evolution of tannin-rich plant communities has occurred on nutrient-poor acidic soils throughout the world. Tannins were once believed to function as anti-herbivore defenses, but more and more ecologists now recognize them as important controllers of decomposition and nitrogen cycling processes. As concern grows about global warming, there is great interest to better understand the role of polyphenols as regulators of carbon cycling, in particular in northern boreal forests.[23]
Presence in water and wood
The
Tannins leaching from an unprepared driftwood decoration in an aquarium can cause pH lowering and coloring of the water to a tea-like tinge. A way to avoid this is to boil the
Softwoods, while in general much lower in tannins than hardwoods,[27] are usually not recommended for use in an aquarium[28] so using a hardwood with a very light color, indicating a low tannin content, can be an easy way to avoid tannins. Tannic acid is brown in color, so in general white woods have a low tannin content. Woods with a lot of yellow, red, or brown coloration to them (like cedar, redwood, red oak, etc.) tend to contain a lot of tannin.[29]
-
Tannin-rich fresh water draining into Cox Bight from Freney Lagoon, Southwest Conservation Area, Tasmania, Australia
-
Upper Tahquamenon falls Panoramic view
-
The tannin-richOparara Riverin the West Coast region of New Zealand
Extraction
There is no single protocol for extracting tannins from all plant material. The procedures used for tannins are widely variable.[30] It may be that acetone in the extraction solvent increases the total yield by inhibiting interactions between tannins and proteins during extraction[30] or even by breaking hydrogen bonds between tannin-protein complexes.[31]
Tests for tannins
There are three groups of methods for the analysis of tannins: precipitation of proteins or alkaloids, reaction with phenolic rings, and depolymerization.[32]
Alkaloid precipitation
Alkaloids such as caffeine, cinchonine, quinine or strychnine, precipitates polyphenols and tannins. This property can be used in a quantitation method.[33]
Goldbeater's skin test
When goldbeater's skin or ox skin is dipped in HCl, rinsed in water, soaked in the tannin solution for 5 minutes, washed in water, and then treated with 1% FeSO4 solution, it gives a blue black color if tannin was present.[34]
Ferric chloride test
The following describes the use of
Other methods
The hide-powder method is used in tannin analysis for
- Hide-powder method
400 mg of sample tannins are dissolved in 100 ml of distilled water. 3 g of slightly chromated hide-powder previously dried in vacuum for 24h over CaCl2 are added and the mixture stirred for 1 h at ambient temperature. The suspension is filtered without vacuum through a sintered glass filter. The weight gain of the hide-powder expressed as a percentage of the weight of the starting material is equated to the percentage of tannin in the sample.
- Stiasny's method
100 mg of sample tannins are dissolved in 10 ml distilled water. 1 ml of 10M HCl and 2 ml of 37% formaldehyde are added and the mixture heated under reflux for 30 min. The reaction mixture is filtered while hot through a sintered glass filter. The precipitate is washed with hot water (5× 10 ml) and dried over CaCl2. The yield of tannin is expressed as a percentage of the weight of the starting material.
Reaction with phenolic rings
The bark tannins of
Colorimetric methods have existed such as the Neubauer-Löwenthal method which uses potassium permanganate as an oxidizing agent and indigo sulfate as an indicator, originally proposed by Löwenthal in 1877.[43] The difficulty is that the establishing of a titer for tannin is not always convenient since it is extremely difficult to obtain the pure tannin. Neubauer proposed to remove this difficulty by establishing the titer not with regard to the tannin but with regard to crystallised oxalic acid, whereby he found that 83 g oxalic acid correspond to 41.20 g tannin. Löwenthal's method has been criticized. For instance, the amount of indigo used is not sufficient to retard noticeably the oxidation of the non-tannins substances. The results obtained by this method are therefore only comparative.[44][45] A modified method, proposed in 1903 for the quantification of tannins in wine, Feldmann's method, is making use of calcium hypochlorite, instead of potassium permanganate, and indigo sulfate.[46]
Food items with tannins
Pomegranates
Accessory fruits
Berries

Most berries, such as cranberries,[48] and blueberries,[49] contain both hydrolyzable and condensed tannins.
Nuts
Herbs and spices
Cloves, tarragon, cumin, thyme, vanilla, and cinnamon all contain tannins.[citation needed]
Legumes
Most legumes contain tannins. Red-colored beans contain the most tannins, and white-colored beans have the least. Peanuts without shells have a very low tannin content. Chickpeas (garbanzo beans) have a smaller amount of tannins.[53]
Chocolate
Chocolate liquor contains about 6% tannins.[54]
Drinks with tannins
Principal human dietary sources of tannins are tea and coffee.
Coffee pulp has been found to contain low to trace amounts of tannins.[59]
Fruit juices
Although citrus fruits do not contain tannins, orange-colored juices often contain tannins from food colouring. Apple, grape and berry juices all contain high amounts of tannins. Sometimes tannins are even added to juices and ciders to create a more astringent feel to the taste.[60]
Beer
In addition to the alpha acids extracted from hops to provide bitterness in beer, condensed tannins are also present. These originate both from malt and hops. Trained brewmasters, particularly those in Germany, consider the presence of tannins to be a flaw[citation needed]. However, in some styles, the presence of this astringency is acceptable or even desired, as, for example, in a Flanders red ale.[61]
In lager type beers, the tannins can form a precipitate with specific haze-forming proteins in the beer resulting in
Properties for animal nutrition
Tannins have traditionally been considered antinutritional, depending upon their chemical structure and dosage.[63]
Many studies suggest that chestnut tannins have positive effects on silage quality in the round bale silages, in particular reducing NPNs (non-protein nitrogen) in the lowest wilting level.[64]
Improved fermentability of
Tannin uses and market

Tannins have been used since antiquity in the processes of tanning hides for leather, and in helping preserve iron artifacts (as with Japanese iron teapots).
Industrial tannin production began at the beginning of the 19th century with the industrial revolution, to produce tanning material for the need for more leather. Before that time, processes used plant material and were long (up to six months).[68]
There was a collapse in the vegetable tannin market in the 1950s–1960s, due to the appearance of
The cost of the final product depends on the method used to extract the tannins, in particular the use of solvents, alkali and other chemicals used (for instance glycerin). For large quantities, the most cost-effective method is hot water extraction.
Tannic acid is used worldwide as clarifying agent in alcoholic drinks and as aroma ingredient in both alcoholic and soft drinks or juices. Tannins from different botanical origins also find extensive uses in the wine industry.[citation needed]
Uses
Tannins are an important ingredient in the process of tanning leather.
Tannins produce different colors with
Tannins can also be used as a mordant, and is especially useful in natural dyeing of cellulose fibers such as cotton.[72] The type of tannin used may or may not have an impact on the final color of the fiber.
Tannin is a component in a type of industrial
Tannins can be used for production of anti-corrosive primers for treating rusted steel surfaces prior to painting, converting rust to iron tannate and consolidating and sealing the surface.
The use of resins made of tannins has been investigated to remove mercury and methylmercury from solution.[75] Immobilized tannins have been tested to recover uranium from seawater.[76]
References
- ISBN 978-0-8018-8402-3.
- ISBN 978-0-684-80001-1.
- ^ Bate-Smith and Swain (1962). "Flavonoid compounds". In Florkin M.; Mason H. S (eds.). Comparative biochemistry. Vol. III. New York: Academic Press. pp. 75–809.
- ^ a b "Notes on Tannins from PharmaXChange.info". Archived from the original on 4 January 2015.
- ^ ISBN 978-1-4684-7511-1
- .
- ISBN 978-81-224-1501-8. Archivedfrom the original on 2 June 2013. Retrieved 31 January 2011.
- ^ ISBN 978-1-4067-7301-9.
- ^ Löwe, Zeitschrift für Chemie, 1868, 4, 603
- PMID 16742048.
- .
- PMID 16742368.
- PMID 16744910.
- .
- PMID 16747927.
- PMID 18037145.
- ^ Quideau, Stéphane (22 September 2009). "Why bother with Polyphenols". Groupe Polyphenols. Archived from the original on 10 March 2012. Retrieved 21 August 2012.[self-published source]
- .
- ^ "Tannin in Tropical Woods". Doat J, Bois. For Tmp., 1978, volume 182, pp. 34–37
- ISBN 978-0-8493-6811-0.
- PMID 24026439.
- ^ "Identification of Molecular Markers Linked to the Trait of Natural Astringency Loss of Japanese Persimmon (Diospyros kaki) Fruit". Shinya Kanzaki, Keizo Yonemori and Akira Sugiura, J. Am. Soc. Hort. Sci., 2001, 126(1), pp. 51–55 (article Archived 4 September 2015 at the Wayback Machine)
- PMID 10802549.
- S2CID 23420808.
- ^ "Tannins, lignins and humic acids in well water on www.gov.ns.ca" (PDF). Archived from the original (PDF) on 17 May 2013.
- ^ Preparing Driftwood for Your Freshwater Aquarium Archived 7 July 2011 at the Wayback Machine
- S2CID 21250123.
- ^ "Driftwood Do's & Don'ts – Pet Fish". Archived from the original on 24 July 2011.
- ^ "Tannin and hardwood flooring". Archived from the original on 17 April 2011.
- ^ a b The Tannin Handbook, Ann E. Hagerman, 1998 (book Archived 28 January 2014 at the Wayback Machine)
- ^ "Condensed tannins". Porter L. J., 1989, in Natural Products of Woody Plants I, Rowe J. W. (ed), Springer-Verlag: Berlin, Germany, pages 651–690
- ISBN 978-1-4613-6540-2.
- ^ Plant Polyphenols: Synthesis, Properties, Significance. Richard W. Hemingway, Peter E. Laks, Susan J. Branham (page 263)
- ISBN 978-81-963961-5-2.
- ^ "Antibacterial activity of leave extracts of Nymphaea lotus (Nymphaeaceae) on Methicillin resistant Staphylococcus aureus (MRSA) and Vancomycin resistant Staphylococcus aureus (VRSA) isolated from clinical samples". Akinjogunla O. J., Yah C. S., Eghafona N. O. and Ogbemudia F. O., Annals of Biological Research, 2010, 1 (2), pages 174–184
- ^ "Phytochemical Analysis and Antimicrobial Activity Of Scoparia dulcis and Nymphaea lotus". Jonathan Yisa, Australian Journal of Basic and Applied Sciences, 2009, 3(4): pp. 3975–3979
- ^ "Tannin analysis of Acacia mearnsii bark – a comparison of the hide-powder and Stiasny methods". Zheng G.C., Lin Y.L. and Yazaki Y., ACIAR Proceedings Series, 1991, No. 35, pp. 128–131 (abstract Archived 9 July 2014 at the Wayback Machine)
- ^ Study on Fast Determination Content of Condensed Tannin Using Stiasny Method. Chen Xiangming, Chen Heru and Li Weibin, Guangdong Chemical Industry, 2006-07 (abstract Archived 2 April 2015 at the Wayback Machine)
- .
- ^ Leather Chemists' Pocket-Book: A Short Compendium of Analytical Methods. Henry Richardson Procter, Edmund Stiasny and Harold Brumwel, E. & F.N. Spon, Limited, 1912–223 pages (book at Internet Archive Archived 16 December 2016 at the Wayback Machine)
- ^ Chemical study of bark from Commiphora angolensis Engl. Cardoso Do Vale, J., Bol Escola Farm Univ Coimbra Edicao Cient, 1962, volume 3, page 128 (abstract Archived 7 June 2014 at the Wayback Machine)
- .
- S2CID 95511307.
- S2CID 85362459.
- .
- ^ "Nouvelle methode de dosage du tannin" (PDF). Schweizerische Wochenschrift für Chemie und Pharmacie (in French). Archived from the original (PDF) on 8 August 2014.
- S2CID 6548208.
- PMID 15927928. Archived from the original(PDF) on 28 December 2010.
- S2CID 6548208.
- . Retrieved 15 September 2020.
- ^ Howes, F. N. (1948). Nuts: Their production and everyday uses. Faber.
- ^ Amarowicz, R.; Pegg, R.B. (2008). Assessment of the antioxidant and pro-oxidant activities of tree nut extracts with a pork model system (PDF). International Congress of Meat Science and Technology. Archived from the original (PDF) on 27 April 2021. Retrieved 9 September 2019.
- PMID 7665384.
- ISBN 978-0-393-05869-7. Archivedfrom the original on 16 December 2016.
- PMID 15643541.
- S2CID 42400092.
- ISBN 978-0-15-100714-1
- PMID 21399572.
- .
- ^ "tannin2". www.cider.org.uk. Retrieved 21 March 2019.
- PMID 3536314.
- ^ "Brewtan range – Natural solutions for beer stabilisation – Application fact-sheet" (PDF). natural-specialities.com. Ajinomoto OmniChem. Archived from the original (PDF) on 14 July 2011. Retrieved 10 March 2010.
- ^ Muller-Harvey I.; McAllan A. B. (1992). "Tannins: Their biochemistry and nutritional properties". Adv. Plant Cell Biochem. Biotechnol. 1: 151–217.
- PMID 17106105.
- .
- S2CID 21617545.
- PMID 35225785.

- ^ "1854 - 1906: The foundation". Silvateam. 15 May 2015. Retrieved 9 August 2022.
- ^ ""The Status of Mangrove Ecosystems: Trends in the Utilisation and Management of Mangrove Resources". D. Macintosh and S. Zisman".
- ISBN 978-0-7506-4881-3. Archivedfrom the original on 16 December 2016.
- ^ Lemay, Marie-France (21 March 2013). "Iron Gall Ink". Traveling Scriptorium: A Teaching Kit. Yale University. Archived from the original on 15 February 2017. Retrieved 18 January 2017.
- ISSN 1319-6103.
- .
- ^ Li, Jingge; Maplesden, Frances (1998). "Commercial production of tannins from radiata pine bark for wood adhesives" (PDF). IPENZ Transactions. 25 (1/EMCh). Archived from the original (PDF) on 22 January 2003.
- S2CID 24811963.
- .
External links
- Tannins: fascinating but sometimes dangerous molecules
- "Tannin Chemistry" (PDF). (1.41 MB)
- Haslam, Edwin (1989). Plant polyphenols: vegetable tannins revisited. CUP Archive. ISBN 978-0-521-32189-1.




