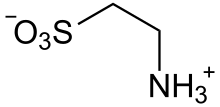
Taurine
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-Aminoethanesulfonic acid | |
| Other names
Tauric acid
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.003.168 |
IUPHAR/BPS |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H7NO3S | |
| Molar mass | 125.14 g/mol |
| Appearance | colorless or white solid |
| Density | 1.734 g/cm3 (at −173.15 °C) |
| Melting point | 305.11 °C (581.20 °F; 578.26 K) Decomposes into simple molecules |
| Acidity (pKa) | <0, 9.06 |
| Related compounds | |
Related compounds
|
Sulfamic acid Aminomethanesulfonic acid Homotaurine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Taurine (/ˈtɔːriːn/), or 2-aminoethanesulfonic acid, is a non-proteinogenic naturally occurred amino sulfonic acid that is widely distributed in animal tissues.[1] It is a major constituent of bile and can be found in the large intestine, and accounts for up to 0.1% of total human body weight.
Taurine is named after Latin taurus (cognate to Ancient Greek ταῦρος, taûros) meaning bull or ox, as it was first isolated from ox bile in 1827 by German scientists Friedrich Tiedemann and Leopold Gmelin.[2] It was discovered in human bile in 1846 by Edmund Ronalds.[3]
Although taurine is abundant in human organs with diverse putative roles, it is not an essential human dietary nutrient and is not included among nutrients with a recommended intake level.[4] Taurine is commonly sold as a dietary supplement, but there is no good clinical evidence that taurine supplements provide any benefit to human health.[5] Taurine is used as a food additive for cats (who require it as an essential nutrient), dogs, and poultry.[6]
Taurine concentrations in
Chemical and biochemical features
Taurine exists as a
found in the intestinal tract.Synthesis
Synthetic taurine is obtained by the ammonolysis of isethionic acid (2-hydroxyethanesulfonic acid), which in turn is obtained from the reaction of ethylene oxide with aqueous sodium bisulfite. A direct approach involves the reaction of aziridine with sulfurous acid.[11]
In 1993, about 5000–6000 tonnes of taurine were produced for commercial purposes: 50% for pet food and 50% in pharmaceutical applications.[12] As of 2010, China alone has more than 40 manufacturers of taurine. Most of these enterprises employ the ethanolamine method to produce a total annual production of about 3000 tonnes.[13]
In the laboratory, taurine can be produced by alkylation of ammonia with bromoethanesulfonate salts.[14]
Biosynthesis
Taurine is naturally derived from cysteine. Mammalian taurine synthesis occurs in the pancreas via the cysteine sulfinic acid pathway. In this pathway, cysteine is first oxidized to its sulfinic acid, catalyzed by the enzyme cysteine dioxygenase. Cysteine sulfinic acid, in turn, is decarboxylated by sulfinoalanine decarboxylase to form hypotaurine. Hypotaurine is enzymatically oxidized to yield taurine by hypotaurine dehydrogenase.[15]
Taurine is also produced by the transsulfuration pathway, which converts homocysteine into cystathionine. The cystathionine is then converted to hypotaurine by the sequential action of three enzymes: cystathionine gamma-lyase, cysteine dioxygenase, and cysteine sulfinic acid decarboxylase. Hypotaurine is then oxidized to taurine as described above.[16]
A pathway for taurine biosynthesis from

In food
Taurine occurs naturally in fish and meat.
Taurine is partially destroyed by heat in processes such as baking and boiling. This is a concern for cat food, as cats have a dietary requirement for taurine and can easily become deficient. Either raw feeding or addition of extra taurine can satisfy this requirement.[22][23]
Lysine and taurine can both mask the metallic flavor of potassium chloride, a salt substitute.[24]
Breast milk
Prematurely born infants are believed to lack the enzymes needed to convert cystathionine to cysteine, and may, therefore, become deficient in taurine. Taurine is present in breast milk, and has been added to many infant formulas, as a measure of prudence, since the early 1980s. However, this practice has never been rigorously studied, and as such it has yet to be proven to be necessary, or even beneficial.[25]
Energy drinks and dietary supplements
Taurine is an ingredient in some energy drinks in amounts of 1–3 g per serving.[5][26][27][28] A 1999 assessment of European consumption of energy drinks found that taurine intake was 40–400 mg per day.[21]
Research
Taurine is not regarded as an
Safety and toxicity
According to the European Food Safety Authority (EFSA), taurine is "considered to be a skin and eye irritant and skin sensitiser, and to be hazardous if inhaled;" it may be safe to consume up to 6 grams of taurine per day.[6] Other sources indicate that taurine is safe for supplemental intake in normal healthy adults at up to 3 grams per day.[5][29]
A 2008 review found no documented reports of negative or positive health effects associated with the amount of taurine used in energy drinks, concluding, "The amounts of guarana, taurine, and ginseng found in popular energy drinks are far below the amounts expected to deliver either therapeutic benefits or adverse events".[30]
Animal dietary requirement
Cats
Decreased plasma taurine concentration has been demonstrated to be associated with feline dilated cardiomyopathy. Unlike CRD, the condition is reversible with supplementation.[35]
Taurine is now a requirement of the Association of American Feed Control Officials (AAFCO) and any dry or wet food product labeled approved by the AAFCO should have a minimum of 0.1% taurine in dry food and 0.2% in wet food.[36] Studies suggest the amino acid should be supplied at 10 mg per kilogram of bodyweight per day for domestic cats.[37]
Other mammals
A number of other mammals also have a requirement for taurine. While the majority of dogs can synthesize taurine, case reports have described a singular
Birds
Taurine appears to be essential for the development of
Taurine can be synthesized by chickens. Supplementation has no effect on chickens raised under adequate lab conditions, but seems to help with growth under stresses such as heat and dense housing.[40]
Fish
Species of fish, mostly carnivorous ones, show reduced growth and survival when the fish-based feed in their food is replaced with soy meal or feather meal. Taurine has been identified as the factor responsible for this phenomenon; supplementation of taurine to plant-based fish feed reverses these effects. Future aquaculture is expected to use more of these more environmentally-friendly protein sources, so supplementation would become more important.[41]
The need of taurine in fish is conditional, differing by species and growth stage. The Olive flounder, for example, has lower capacity to synthesize taurine compared to the rainbow trout. Juvenile fish are less efficient at taurine biosyntheis due to reduced cysteine sulfinate decarboxylase levels.[42]
Derivatives
- Taurine is used in the preparation of the anthelmintic drug netobimin (Totabin).
- Taurolidine
- tauroselcholic acid
- Tauromustine
- 5-Taurinomethyluridine and 5-taurinomethyl-2-thiouridine are modified uridines in (human) mitochondrial tRNA.[43]
- Tauryl is the functional group attaching at the sulfur, 2-aminoethylsulfonyl.[44]
- Taurino is the functional group attaching at the nitrogen, 2-sulfoethylamino.
- Thiotaurine
- Peroxytaurine which is a degradation product by both superoxide and heat degradation.
See also
- Homotaurine (tramiprosate), precursor to acamprosate
- Taurates, a group of surfactants
References
- PMID 14553911.
- .
- ^ Ronalds BF (2019). "Bringing Together Academic and Industrial Chemistry: Edmund Ronald' Contribution". Substantia. 3 (1): 139–152.
- ^ a b "Daily Value on the New Nutrition and Supplement Facts Labels". US Food and Drug Administration. 25 February 2022. Retrieved 26 August 2023.
- ^ a b c d e f g h "Taurine". Drugs.com. 15 May 2023. Retrieved 26 August 2023.
- ^ .
- .
- ^ PMID 26101608.
- .
- PMID 7431022.
- ISBN 978-3527306732.
- ISBN 978-0-471-23896-6.
- ^ Amanda Xia (2010-01-03). "China Taurine Market Is Expected To Recover". Press release and article directory: technology. Archived from the original on 2018-09-20. Retrieved 2010-05-24.
- .
- PMID 13979247.
- PMID 23170060.
- PMID 13233249.
- S2CID 77903.
- PMID 16702333.
- S2CID 27844955.
- ^ a b "Opinion on Caffeine, Taurine and D-Glucurono – g -Lactone as constituents of so-called 'energy' drinks". Directorate-General Health and Consumers, European Commission, European Union. 1999-01-21. Archived from the original on 2006-06-23.
- PMID 3428381.
- PMID 12864905. Retrieved January 27, 2024.
- PMID 24008059.
- PMID 15499132.
- ^ "Original Rockstar Ingredients". rockstar69.com. Archived from the original on 2007-11-03. Retrieved 2023-06-23.
- ^ Chang PL (2008-05-03). "Nos Energy Drink – Review". energyfanatics.com. Archived from the original on 2008-06-17. Retrieved 2010-05-21.
- PMID 34039357.
- PMID 18325648.
the newer method described as the Observed Safe Level (OSL) or Highest Observed Intake (HOI) was utilized. The OSL risk assessments indicate that based on the available published human clinical trial data, the evidence for the absence of adverse effects is strong for Tau at supplemental intakes up to 3 g/d, Gln at intakes up to 14 g/d and Arg at intakes up to 20 g/d, and these levels are identified as the respective OSLs for normal healthy adults.
- S2CID 207262028.
- PMID 641594.
- PMID 1138364.
- ISBN 978-0-309-07483-4.
- PMID 1138364.
- PMID 3616607.
- ^ "AAFCO Cat Food Nutrient Profiles". Archived from the original on 2015-05-29. Retrieved 30 May 2015.
- .
- PMID 24337931. (abstracts of animal citations used to provide list of species)
- PMID 17698490.
- S2CID 209599794.
- .
- .
- PMID 12456664.
- ISBN 978-1-4200-4615-1.
