Teicoplanin
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌtaɪkoʊˈpleɪnɪn/ TY-koh-PLAY-nin |
| Trade names | Targocid |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Intravenous, intramuscular |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 90% (given IM) |
| Protein binding | 90% to 95% |
| Metabolism | Nil |
| Elimination half-life | 70 to 100 hours |
| Excretion | Kidney (97% unchanged) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | Variable |
| Molar mass | 1564.3 to 1907.7 g/mol |
| Melting point | 260 °C (500 °F) (dec.) |
| |
| | |
Teicoplanin is an semisynthetic
Teicoplanin is marketed by
Oral teicoplanin has been demonstrated to be effective in the treatment of
Its strength is considered to be due to the length of the hydrocarbon chain.[6]
Teicoplanin is produced by so-called "rare" actinobacterium Actinoplanes teichomyceticus ATCC 31121,[7] belonging to the Micromonosporaceae family. Biosynthetic pathway leading to teicoplanin, as well as the regulatory circuit governing the biosynthesis, were studied intensively in recent years allowing to construct an integrated model of the biosynthesis.[8]
Indications
Teicoplanin is applicable in treatment of a wide range of infections with Gram-positive bacteria, including endocarditis, septicaemia, soft tissue and skin infections, and venous catheter-associated infections.[9]
Susceptible organisms
Teicoplanin has demonstrated in vitro efficacy against Gram-positive bacteria including staphylococci (including MRSA), streptococci, enterococci, and against anaerobic Gram-positive bacteria including Clostridium spp. Teicoplanin is not effective against Gram-negative bacteria as the large, polar molecules of the compound are unable to pass through the external membrane of these organisms.[9] The following represents MIC susceptibility data for a few medically significant pathogens:[4]
- Clostridium difficile: 0.06 μg/ml - 0.5 μg/ml
- Staphylococcus aureus: ≤0.06 μg/ml - ≥128 μg/ml
- Staphylococcus epidermidis: ≤0.06 μg/ml - 32 μg/ml
Pharmacology
Teicoplanin exhibits a very long biological half-life of about 45-70h (sufficient plasma levels can thus be sustained with once-daily administration). Elimination is almost exclusively renal. It is mostly excreted unchanged.[9]
Adverse effects
Adverse effects of teicoplanin are usually limited to local effects or hypersensitivity reactions. While there is potenial for nephrotoxicity and ototoxicity, incidence of such organ toxicity is rare if recommended serum concentrations are successfully maintained.[9]
Considerations
Reduced renal function slows teicoplanin clearance, consequently increasing its elimination half-life. Elimination half-life is longer in the elderly due to the reduced renal function in this population.[9]
Chemistry
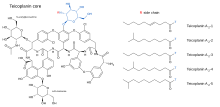
Teicoplanin (TARGOCID, marketed by Sanofi Aventis Ltd) is actually a mixture of several compounds, five major (named teicoplanin A2-1 through A2-5) and four minor (named teicoplanin RS-1 through RS-4).[10][11] All teicoplanins share a same glycopeptide core, termed teicoplanin A3-1 — a fused ring structure to which two carbohydrates (mannose and N-acetylglucosamine) are attached. The major and minor components also contain a third carbohydrate moiety — β-D-glucosamine — and differ only by the length and conformation of a side-chain attached to it. Teicoplanin A2-4 and RS-3 have chiral side chains while all other side chains are achiral. Teicoplanin A3 lacks both the side chains as well as the β-D-glucosamine moiety.
The structures of the teicoplanin core and the side-chains that characterize the five major as well as four minor teicoplanin compounds are shown below.

Teicoplanin refers to a complex of related natural products isolated from the fermentation broth of a strain of
Gene cluster
The study of the genetic cluster encoding the biosynthesis of teicoplanin identified 49 putative open reading frames (ORFs) involved in the compound's biosynthesis, export, resistance, and regulation. Thirty-five of these ORFs are similar to those found in other glycopeptide gene clusters. The function of each of these genes is described by Li and co-workers.[14] A summary of the gene layout and purpose is shown below.
Gene layout. The genes are numbered. The letters L and R designate transcriptional direction. The presence of the * symbol means a gene is found after NRPs, which are represented by A, B, C, and D. Based on the figure from: Li, T-L.; Huang, F.; Haydock, S. F.; Mironenko, T.; Leadlay, P. F.; Spencer, J. B. Chemistry & Biology. 2004, 11, p. 109.
[11-L] [10-L] [9-R] [8-R] [7-R] [6-R] [5-R] [4-L][3-L] [2-L] [1-R] [A-R] [B-R] [C-R] [D-R] [1*-R] [2*-R] [3*-R] [4*-R] [5*-R] [6*-R] [7*-R] [8*-R] [9*-R] [10*-R] [11*-R] [12*-R] [13*-R] [14*-R] [15*-R] [16*-R] [17*-R] [18*-R] [19*-R] [20*-R] [21*-R] [22*-R] [23*-R] [24*-R] [25*-L] [26*-L] [27*-R] [28*-R] [29*-R] [30*-R][31*-R] [32*-L] [33*-L] [34*-R]
| Enzyme produced by gene sequence | Regulatory proteins | Other enzymes | Resistant enzymes | Β-hydroxy-tyrosine and 4-hydroxy-phenylglycin biosynthetic enzymes | Glycosyl transferases | Peptide synthetases | P450 oxygenases | Halogenase | 3,5-dihydroxy phenylglycin biosynthetic enzymes |
| Genes | 11, 10, 3, 2, 15*, 16*, 31* | 9, 8, 1*, 2*, 4*, 11*, 13*, 21*, 26*, 27*, 30*, 32*, 33*, 34* | 7, 6, 5 | 4, 12*, 14*, 22*, 23*, 24*, 25*, 28*, 29* | 1, 3*, 10* | A, B, C, D | 5*, 6*, 7*, 9* | 8* | 17*, 18*, 19*, 20*, 23* |
Heptapeptide backbone synthesis
The heptapeptide backbone of teicoplanin is assembled by the nonribosomal peptide synthetases (NRPSs) TeiA, TeiB, TeiC and TeiD. Together these comprise seven modules, each containing a number of domains, with each module responsible for the incorporation of a single amino acid. Modules 1, 4, and 5 activate L-4-Hpg as the aminoacyl-AMP, modules 2 and 6 activate L-Tyr, and modules 3 and 7 activate L-3,5-Dpg. The activated amino acids are covalently bound to the NRPS as thioesters by a phosphopantetheine cofactor, which is attached to the peptidyl carrier protein (PCP) domain. The enzyme bound amino acids are then joined by amide bonds by the action of the condensation (C) domain.
The heptapetide of teicoplanin contains 4 D-amino acids, formed by epimerization of the activated L-amino acids. Modules 2, 4 and 5 each contain an epimerization (E) domain which catalyzes this change. Module 1 does not contain an E domain, and epimerization is proposed to be catalysed by the C domain.[15] In all, six of the seven total amino acids of the teicoplanin backbone are composed of nonproteinogenic or modified amino acids. Eleven enzymes are coordinatively induced to produce these six required residues.[16] Teicoplanin contains two chlorinated positions, 2 (3-Cl-Tyr) and 6 (3-Cl-β-Hty). The halogenase Tei8* has been acts to catalyze the halogenation of both tyrosine residues. Chlorination occurs at the amino acyl-PCP level during the biosynthesis, prior to phenolic oxidative coupling, with the possibility of tyrosine or β-hydroxytyrosine being the substrate of chlorination.[17] Hydroxylation of the tyrosine residue of module 6 also occurs in trans during the assembly of the heptapeptide backbone.
Modification after heptapeptide backbone formation
Once the heptapeptide backbone has been formed, the linear enzyme-bound intermediate is cyclized.[16] Gene disruption studies indicate cytochrome P450 oxygenases as the enzymes that performs the coupling reactions. The X-domain in the final NRPS module is required to recruit the oxygenase enzymes.[18] OxyB forms the first ring by coupling residues 4 and 6, and OxyE then couples residues 1 and 3. OxyA couples residues 2 and 4, followed by the formation of a C-C bond between residues 5 and 7 by OxyC.[19] The regioselectivity and atropisomer selectivity of these probable one-electron coupling reactions has been suggested to be due to the folding and orientation requirements of the partially crossed-linked substrates in the enzyme active site.[16] The coupling reactions are shown below.

Specific glycosylation has been shown to occur after the formation of the heptpeptide aglycone.[20] Three separate glycosyl transferases are required for the glycosylation of the teicoplanin aglycone. Tei10* catalyses the addition of GlcNAc to residue 4, followed by deacetylation by Tei2*. The acyl chain (produced by the action of Tei30* and Tei13*) is then added by Tei11*. Tei1 then adds a second GlcNAc to the β-hydroxyl group of residue 6, followed by mannosylation of residue 7 catalysed by Tei3*.[21]
References
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ Human Medicines Evaluation Division (10 December 2020). "Active substance: teicoplanin" (PDF). List of nationally authorised medicinal products. European Medicines Agency.
- ^ S2CID 21551939.
- ^ a b "Teicoplanin Susceptibility and Minimum Inhibitory Concentration (MIC) Data" (PDF). TOKU-E.
- PMID 1444298.
- ^ Gilpin M, Milner P (1997). "Resisting changes -- Over the past 40 years the glycopeptide antibiotics have played a crucial role in treating bacterial infections. But how long can it continue ?". Royal Society of Chemistry. Archived from the original on 2002-12-21. Retrieved 2006-10-15. - includes picture of Teicoplanin's structure.
- ^ "Actinoplanes teichomyceticus Parenti et al". ATCC (American Type Culture Collection).
- S2CID 211170874.
- ^ S2CID 195699141.
- PMID 1416858.
- S2CID 253529204.
- S2CID 45038487.
- PMID 10692413.
- PMID 15113000.
- S2CID 218481859.
- ^ PMID 15700951.
- PMID 28989629.
- S2CID 4466657.
- PMID 27752135.
- PMID 11356118.
- PMID 27285718.
