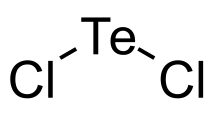
Tellurium dichloride
| |

| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cl2Te | |
| Molar mass | 198.50 g·mol−1 |
| Appearance | black solid[1] |
| Density | 6.9 g·cm−3[1] |
| Melting point | 208 °C[1] |
| Boiling point | 328 °C[1] |
| reacts[1] | |
| Solubility | reacts with tetrachloromethane[1]
|
| Related compounds | |
Other anions
|
Ditellurium bromide, Te2Br |
Other cations
|
Dichlorine monoxide, OCl2 Sulfur dichloride, SCl2 Selenium dichloride, SeCl2 Polonium dichloride, PoCl2 |
Related compounds
|
Tritellurium dichloride, Te3Cl2 Tellurium tetrachloride, TeCl4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tellurium dichloride is a chloride of tellurium with the chemical formula TeCl2.
Preparation
Tellurium dichloride can be produced by reacting tellurium with
It can also be produced by the comproportionation of tellurium and tellurium tetrachloride.[4]
Properties
Tellurium dichloride is a black solid that reacts with water. It will melt into a black liquid and vapourize into a purple gas.bond angle of 97.0°.[5]
Reactions
Tellurium dichloride reacts with barium chloride in water to form barium tellurite.
