Telomere
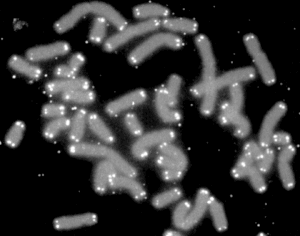
A telomere (
Discovery
The existence of a special structure at the ends of chromosomes was independently proposed in 1938 by Hermann Joseph Muller, studying the fruit fly Drosophila melanogaster, and in 1939 by Barbara McClintock, working with maize.[1] Muller observed that the ends of irradiated fruit fly chromosomes did not present alterations such as deletions or inversions. He hypothesized the presence of a protective cap, which he coined "telomeres", from the Greek telos (end) and meros (part).[2]
In the early 1970s, Soviet theorist
In 1975–1977,
Structure and function
End replication problem

During DNA replication,
If coding sequences are degraded in this process, potentially vital genetic code would be lost. Telomeres are non-coding, repetitive sequences located at the termini of linear chromosomes to act as buffers for those coding sequences further behind. They "cap" the end-sequences and are progressively degraded in the process of DNA replication.
The "end replication problem" is exclusive to linear chromosomes as circular chromosomes do not have ends lying without reach of DNA-polymerases. Most
Telomere ends and shelterin

At the very 3'-end of the telomere there is a 300 base pair overhang which can invade the double-stranded portion of the telomere forming a structure known as a T-loop. This loop is analogous to a knot, which stabilizes the telomere, and prevents the telomere ends from being recognized as breakpoints by the DNA repair machinery. Should non-homologous end joining occur at the telomeric ends, chromosomal fusion would result. The T-loop is maintained by several proteins, collectively referred to as the shelterin complex. In humans, the shelterin complex consists of six proteins identified as
Telomerase

Many organisms have a ribonucleoprotein enzyme called telomerase, which carries out the task of adding repetitive nucleotide sequences to the ends of the DNA. Telomerase "replenishes" the telomere "cap" and requires no ATP
Length
Telomere length varies greatly between species, from approximately 300
Shortening
Oxidative damage
Apart from the end replication problem, in vitro studies have shown that telomeres accumulate damage due to oxidative stress and that oxidative stress-mediated DNA damage has a major influence on telomere shortening in vivo. There is a multitude of ways in which oxidative stress, mediated by reactive oxygen species (ROS), can lead to DNA damage; however, it is yet unclear whether the elevated rate in telomeres is brought about by their inherent susceptibility or a diminished activity of DNA repair systems in these regions.[26] Despite widespread agreement of the findings, widespread flaws regarding measurement and sampling have been pointed out; for example, a suspected species and tissue dependency of oxidative damage to telomeres is said to be insufficiently accounted for.[27] Population-based studies have indicated an interaction between anti-oxidant intake and telomere length. In the Long Island Breast Cancer Study Project (LIBCSP), authors found a moderate increase in breast cancer risk among women with the shortest telomeres and lower dietary intake of beta carotene, vitamin C or E.[28] These results [29] suggest that cancer risk due to telomere shortening may interact with other mechanisms of DNA damage, specifically oxidative stress.
Association with aging
Although telomeres shorten during the lifetime of an individual, it is telomere shortening-rate rather than telomere length that is associated with the lifespan of a species.
Telomere shortening is associated with aging, mortality, and aging-related diseases in experimental animals.[6][32] Although many factors can affect human lifespan, such as smoking, diet, and exercise, as persons approach the upper limit of human life expectancy, longer telomeres may be associated with lifespan.[33]
Potential effect of psychological stress
Meta-analyses found that increased perceived psychological stress was associated with a small decrease in telomere length—but that these associations attenuate to no significant association when accounting for publication bias. The literature concerning telomeres as integrative biomarkers of exposure to stress and adversity is dominated by cross-sectional and correlational studies, which makes causal interpretation problematic.[29][34] A 2020 review argued that the relationship between psychosocial stress and telomere length appears strongest for stress experienced in utero or early life.[35]
Lengthening

The phenomenon of limited cellular division was first observed by Leonard Hayflick, and is now referred to as the Hayflick limit.[36][37] Significant discoveries were subsequently made by a group of scientists organized at Geron Corporation by Geron's founder Michael D. West, that tied telomere shortening with the Hayflick limit.[38] The cloning of the catalytic component of telomerase enabled experiments to test whether the expression of telomerase at levels sufficient to prevent telomere shortening was capable of immortalizing human cells. Telomerase was demonstrated in a 1998 publication in Science to be capable of extending cell lifespan, and now is well-recognized as capable of immortalizing human somatic cells.[39]
Two studies on long-lived
A study reported that telomere length of different mammalian species correlates inversely rather than directly with lifespan, and concluded that the contribution of telomere length to lifespan remains controversial.[41] There is little evidence that, in humans, telomere length is a significant biomarker of normal aging with respect to important cognitive and physical abilities.[42]
Sequences
Experimentally verified and predicted telomere sequence motifs from more than 9000 species are collected in research community curated database TeloBase.[43] Some of the experimentally verified telomere nucleotide sequences are also listed in Telomerase Database website (see nucleic acid notation for letter representations).
| Group | Organism | Telomeric repeat (5' to 3' toward the end) |
|---|---|---|
| Vertebrates | Xenopus
|
TTAGGG |
| Filamentous fungi | Neurospora crassa | TTAGGG |
Slime moulds
|
Physarum, Didymium | TTAGGG |
| Dictyostelium | AG(1-8) | |
Kinetoplastid protozoa
|
Trypanosoma, Crithidia | TTAGGG |
| Ciliate protozoa | Tetrahymena, Glaucoma | TTGGGG |
| Paramecium | TTGGG(T/G) | |
Oxytricha, Stylonychia, Euplotes
|
TTTTGGGG | |
| Apicomplexan protozoa | Plasmodium | TTAGGG(T/C) |
| Higher plants | Arabidopsis thaliana | TTTAGGG |
| Cestrum elegans | TTTTTTAGGG[44] | |
| Allium | CTCGGTTATGGG[45] | |
Green algae Chlamydomonas
|
TTTTAGGG | |
| Insects | Bombyx mori | TTAGG |
| Bombus terrestris | TTAGGTTGGGG[46] | |
| Vespula vulgaris | TTGCGTCTGGG[46] | |
Roundworms
|
Ascaris lumbricoides | TTAGGC |
| Fission yeasts | Schizosaccharomyces pombe | TTAC(A)(C)G(1-8) |
| Budding yeasts | Saccharomyces cerevisiae | TGTGGGTGTGGTG (from RNA template) or G(2-3)(TG)(1-6)T (consensus) |
| Saccharomyces castellii | TCTGGGTG | |
Candida glabrata
|
GGGGTCTGGGTGCTG | |
| Candida albicans | GGTGTACGGATGTCTAACTTCTT | |
| Candida tropicalis | GGTGTA[C/A]GGATGTCACGATCATT | |
| Candida maltosa | GGTGTACGGATGCAGACTCGCTT | |
| Candida guillermondii | GGTGTAC | |
Candida pseudotropicalis
|
GGTGTACGGATTTGATTAGTTATGT | |
| Kluyveromyces lactis | GGTGTACGGATTTGATTAGGTATGT |
Research on disease risk
This section needs more primary sources. (March 2018) |  |
Preliminary research indicates that disease risk in aging may be associated with telomere shortening,
Measurement
Several techniques are currently employed to assess average telomere length in eukaryotic cells. One method is the Terminal Restriction Fragment (TRF) southern blot.[47][48] There is a Web-based Analyser of the Length of Telomeres (WALTER), software processing the TRF pictures.[49] A Real-Time PCR assay for telomere length involves determining the Telomere-to-Single Copy Gene (T/S) ratio, which is demonstrated to be proportional to the average telomere length in a cell.[50]
Tools have also been developed to estimate the length of telomere from whole genome sequencing (WGS) experiments. Amongst these are TelSeq,[51] Telomerecat[52] and telomereHunter.[53] Length estimation from WGS typically works by differentiating telomere sequencing reads and then inferring the length of telomere that produced that number of reads. These methods have been shown to correlate with preexisting methods of estimation such as PCR and TRF. Flow-FISH is used to quantify the length of telomeres in human white blood cells. A semi-automated method for measuring the average length of telomeres with Flow FISH was published in Nature Protocols in 2006.[54]
While multiple companies offer telomere length measurement services, the utility of these measurements for widespread clinical or personal use has been questioned.[55][56] Nobel Prize winner Elizabeth Blackburn, who was co-founder of one company, promoted the clinical utility of telomere length measures.[57]
In wildlife
During the last two decades, eco-evolutionary studies have investigated the relevance of life-history traits and environmental conditions on telomeres of wildlife. Most of these studies have been conducted in endotherms, i.e. birds and mammals. They have provided evidence for the inheritance of telomere length; however, heritability estimates vary greatly within and among species.[58] Age and telomere length often negatively correlate in vertebrates, but this decline is variable among taxa and linked to the method used for estimating telomere length.[59] In contrast, the available information shows no sex differences in telomere length across vertebrates.[60] Phylogeny and life history traits such as body size or the pace of life can also affect telomere dynamics. For example, it has been described across species of birds and mammals.[61] In 2019, a meta-analysis confirmed that the exposure to stressors (e.g. pathogen infection, competition, reproductive effort and high activity level) was associated with shorter telomeres across different animal taxa.[62]
Studies on
See also
- Epigenetic clock
- Centromere
- DNA damage theory of aging
- Immortality
- Maximum life span
- Rejuvenation (aging)
- Senescence, biological aging
- Tankyrase
- Telomere-binding protein
- G-quartet
- Immortal DNA strand hypothesis
Notes
- ^ During replication, multiple DNA-polymerases are involved.
References
- S2CID 11726588.
- ^ Muller, H.J. (1938). The Remaking of Chromosomes. Woods Hole. pp. 181–198.
- PMID 5158754.
- PMID 4754905.
- S2CID 26381790.
- ^ PMID 642006.
- ^ "Elizabeth H. Blackburn, Carol W. Greider, Jack W. Szostak: The Nobel Prize in Physiology or Medicine 2009". Nobel Foundation. 2009-10-05. Retrieved 2012-06-12.
- PMID 4754905.
- PMID 22661228.
- OCLC 191854286.
- ^ Maloy S (July 12, 2002). "Bacterial Chromosome Structure". Retrieved 2008-06-22.
- PMID 20569239.
- PMID 2780561.
- PMID 19589679.
- PMID 27182535.
- S2CID 37387314.
- PMID 31285335.
- PMID 15471900.
- PMID 15520642.
- S2CID 11920124.
- ^ Barnes, R.P., de Rosa, M., Thosar, S.A., et al., Telomeric 8-oxo-guanine drives rapid premature senescence in the absence of telomere shortening, Nature, June 30, 2022; Nat Struct Mol Biol 29, 639–652 (2022). https://doi.org/10.1038/s41594-022-00790-y
- S2CID 4360698.
- PMID 20826803.
- S2CID 721901.
- PMID 17012276.
- PMID 29604323.
- PMID 29212750.
- PMID 19089916.
- ^ PMID 26853993.
- ^ PMID 35165420.
- PMID 33504179.
- PMID 22782639.
- PMID 28394764.
- PMID 30225068.
- S2CID 209748557.
- PMID 13905658.
- PMID 14315085.
- S2CID 9440710.
- S2CID 35667874.
- S2CID 13841086.
- PMID 21518243.
- S2CID 10309423.
- PMID 37602392.
- PMID 25828846.
- S2CID 206331112.
- ^ PMID 36546771.
- PMID 2664709.
- S2CID 23833545.
- PMID 33752601.
- PMID 12000852.
- PMID 24609383.
- PMID 29358629.
- PMID 31138115.
- S2CID 20463557.
- ^ Pollack, Andrew (May 18, 2011). "A Blood Test Offers Clues to Longevity". The New York Times.
- S2CID 44594597.
- .
- PMID 29335377.
- S2CID 237328316.
- S2CID 226291119.
- PMID 33662151.
- S2CID 208319503.
- PMID 29335373.
External links
- Telomeres and Telomerase: The Means to the End Nobel Lecture by Elizabeth Blackburn, which includes a reference to the impact of stress, and pessimism on telomere length
- Telomerase and the Consequences of Telomere Dysfunction Nobel Lecture by Carol Greider
- DNA Ends: Just the Beginning Nobel Lecture by Jack Szostak

