Tempering (metallurgy)

Tempering is a process of heat treating, which is used to increase the
Introduction
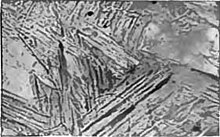
Tempering is a
Precise control of time and temperature during the tempering process is crucial to achieve the desired balance of physical properties. Low tempering temperatures may only relieve the internal stresses, decreasing brittleness while maintaining a majority of the hardness. Higher tempering temperatures tend to produce a greater reduction in the hardness, sacrificing some
In
History
Tempering is an ancient heat-treating technique. The oldest known example of tempered martensite is a
Terminology
In metallurgy, one may encounter many terms that have very specific meanings within the field, but may seem rather vague when viewed from the outside. Terms such as "hardness," "impact resistance," "toughness," and "strength" can carry many different connotations, making it sometimes difficult to discern the specific meaning. Some of the terms encountered, and their specific definitions are:
- tensile strength (the ultimate tearing strength), shear strength (resistance to transverse, or cutting forces), and compressive strength(resistance to elastic shortening under a load).
- Charpy test. Toughness often increases as strength decreases, because a material that bends is less likely to break.
- Hardness – A surface's resistance to scratching, abrasion, or indentation. In conventional metal alloys, there is a linear relation between indentation hardness and tensile strength, which eases the measurement of the latter.[7]
- Brittleness – Brittleness describes a material's tendency to break before bending or deforming either elastically or plastically. Brittleness increases with decreased toughness, but is greatly affected by internal stresses as well.
- malleabilityof the substance.
- Elasticity – Also called flexibility, this is the ability to deform, bend, compress, or stretch and return to the original shape once the external stress is removed. Elasticity is inversely related to the Young's modulus of the material.
- Impact resistance– Usually synonymous with high-strength toughness, it is the ability to resist shock-loading with minimal deformation.
- spalling, or galling.
- Structural integrity – The ability to withstand a maximum-rated load while resisting fracture, resisting fatigue, and producing a minimal amount of flexing or deflection, to provide a maximum service life.
Carbon steel
Very few metals react to heat treatment in the same manner, or to the same extent, that
Except in rare cases where maximum hardness or wear resistance is needed, such as the untempered steel used for
Quenched steel
Tempering is most often performed on steel that has been heated above its upper critical (A3) temperature and then quickly cooled, in a process called quenching, using methods such as immersing the hot steel in water, oil, or forced-air. The quenched steel, being placed in or very near its hardest possible state, is then tempered to incrementally decrease the hardness to a point more suitable for the desired application. The hardness of the quenched steel depends on both cooling speed and on the composition of the alloy. Steel with a high carbon content will reach a much harder state than steel with a low carbon content. Likewise, tempering high-carbon steel to a certain temperature will produce steel that is considerably harder than low-carbon steel that is tempered at the same temperature. The amount of time held at the tempering temperature also has an effect. Tempering at a slightly elevated temperature for a shorter time may produce the same effect as tempering at a lower temperature for a longer time. Tempering times vary, depending on the carbon content, size, and desired application of the steel, but typically range from a few minutes to a few hours.
Tempering quenched steel at very low temperatures, between 66 and 148 °C (151 and 298 °F), will usually not have much effect other than a slight relief of some of the internal stresses and a decrease in brittleness. Tempering at higher temperatures, from 148 to 205 °C (298 to 401 °F), will produce a slight reduction in hardness, but will primarily relieve much of the internal stresses. In some steels with low alloy content, tempering in the range of 260 and 340 °C (500 and 644 °F) causes a decrease in ductility and an increase in brittleness, and is referred to as the "tempered martensite embrittlement" (TME) range. Except in the case of blacksmithing, this range is usually avoided. Steel requiring more strength than toughness, such as tools, are usually not tempered above 205 °C (401 °F). Instead, a variation in hardness is usually produced by varying only the tempering time. When increased toughness is desired at the expense of strength, higher tempering temperatures, from 370 to 540 °C (698 to 1,004 °F), are used. Tempering at even higher temperatures, between 540 and 600 °C (1,004 and 1,112 °F), will produce excellent toughness, but at a serious reduction in strength and hardness. At 600 °C (1,112 °F), the steel may experience another stage of embrittlement, called "temper embrittlement" (TE), which occurs if the steel is held within the temperature range of temper embrittlement for too long. When heating above this temperature, the steel will usually not be held for any amount of time, and quickly cooled to avoid temper embrittlement.[3]
Normalized steel
Steel that has been heated above its upper critical temperature and then cooled in standing air is called normalized steel. Normalized steel consists of pearlite, martensite, and sometimes bainite grains, mixed together within the microstructure. This produces steel that is much stronger than full-annealed steel, and much tougher than tempered quenched steel. However, added toughness is sometimes needed at a reduction in strength. Tempering provides a way to carefully decrease the hardness of the steel, thereby increasing the toughness to a more desirable point. Cast steel is often normalized rather than annealed, to decrease the amount of distortion that can occur. Tempering can further decrease the hardness, increasing the ductility to a point more like annealed steel.[8] Tempering is often used on carbon steels, producing much the same results. The process, called "normalize and temper", is used frequently on steels such as 1045 carbon steel, or most other steels containing 0.35 to 0.55% carbon. These steels are usually tempered after normalizing, to increase the toughness and relieve internal stresses. This can make the metal more suitable for its intended use and easier to machine.[9]
Welded steel
Steel that has been
Quench and self-temper
Modern
Blacksmithing
Tempering was originally a process used and developed by blacksmiths (forgers of iron). The process was most likely developed by the Hittites of Anatolia (modern-day Turkey), in the twelfth or eleventh century BC. Without knowledge of metallurgy, tempering was originally devised through a trial-and-error method.
Because few methods of precisely measuring temperature existed until modern times, the temperature was usually judged by watching the tempering colors of the metal. Tempering often consisted of heating above a charcoal or coal
Tempering colors

If steel has been freshly ground, sanded, or polished, it will form an oxide layer on its surface when heated. As the temperature of the steel is increased, the thickness of the iron oxide will also increase. Although iron oxide is not normally transparent, such thin layers do allow light to pass through, reflecting off both the upper and lower surfaces of the layer. This causes a phenomenon called thin-film interference, which produces colors on the surface. As the thickness of this layer increases with temperature, it causes the colors to change from a very light yellow, to brown, to purple, and then to blue. These colors appear at very precise temperatures and provide the blacksmith with a very accurate gauge for measuring the temperature. The various colors, their corresponding temperatures, and some of their uses are:
- Faint-yellow – 176 °C (349 °F) – gravers, razors, scrapers
- Light-straw – 205 °C (401 °F) – rock drills, reamers, metal-cutting saws
- Dark-straw – 226 °C (439 °F) – scribers, planer blades
- Brown – 260 °C (500 °F) – taps, dies, drill bits, hammers, cold chisels
- Purple – 282 °C (540 °F) – surgical tools, punches, stone carving tools
- Dark blue – 310 °C (590 °F) – screwdrivers, wrenches
- Light blue – 337 °C (639 °F) – springs, wood-cutting saws
- Grey-blue – 371 °C (700 °F) and higher – structural steel
Beyond the grey-blue color, the iron oxide loses its transparency, and the temperature can no longer be judged in this way. The layer will also increase in thickness as time passes, which is another reason overheating and immediate cooling is used. Steel in a tempering oven, held at 205 °C (401 °F) for a long time, will begin to turn brown, purple, or blue, even though the temperature did not exceed that needed to produce a light-straw color.
Differential tempering

Differential tempering is a method of providing different amounts of temper to different parts of the steel. The method is often used in
Differential tempering consists of applying heat to only a portion of the blade, usually the spine, or the center of double-edged blades. For single-edged blades, the heat, often in the form of a flame or a red-hot bar, is applied to the spine of the blade only. The blade is then carefully watched as the tempering colors form and slowly creep toward the edge. The heat is then removed before the light-straw color reaches the edge. The colors will continue to move toward the edge for a short time after the heat is removed, so the smith typically removes the heat a little early, so that the pale yellow just reaches the edge, and travels no farther. A similar method is used for double-edged blades, but the heat source is applied to the center of the blade, allowing the colors to creep out toward each edge.[13]
Interrupted quenching
Interrupted quenching methods are often referred to as tempering, although the processes are very different from traditional tempering. These methods consist of quenching to a specific temperature that is above the martensite start (Ms) temperature, and then holding at that temperature for extended amounts of time. Depending on the temperature and the amount of time, this allows either pure bainite to form, or holds off forming the martensite until much of the internal stresses relax. These methods are known as austempering and martempering.[14]
Austempering

Austempering is a technique used to form pure bainite, a transitional microstructure found between pearlite and martensite. In normalizing, both upper and lower bainite are usually found mixed with pearlite. To avoid the formation of pearlite or martensite, the steel is quenched in a bath of molten metals or salts. This quickly cools the steel past the point where pearlite can form and into the bainite-forming range. The steel is then held at the bainite-forming temperature, beyond the point where the temperature reaches an equilibrium, until the bainite fully forms. The steel is then removed from the bath and allowed to air-cool, without the formation of either pearlite or martensite.
Depending on the holding temperature, austempering can produce either upper or lower bainite. Upper bainite is a laminate structure formed at temperatures typically above 350 °C (662 °F) and is a much tougher microstructure. Lower bainite is a needle-like structure, produced at temperatures below 350 °C, and is stronger but much more brittle.[15] In either case, austempering produces greater strength and toughness for a given hardness, which is determined mostly by composition rather than cooling speed, and reduced internal stresses which could lead to breakage. This produces steel with superior impact resistance. Modern punches and chisels are often austempered. Because austempering does not produce martensite, the steel does not require further tempering.[14]
Martempering
Martempering is similar to austempering, in that the steel is quenched in a bath of molten metal or salts to quickly cool it past the pearlite-forming range. However, in martempering, the goal is to create martensite rather than bainite. The steel is quenched to a much lower temperature than is used for austempering; to just above the martensite start temperature. The metal is then held at this temperature until the temperature of the steel reaches an equilibrium. The steel is then removed from the bath before any bainite can form, and then is allowed to air-cool, turning it into martensite. The interruption in cooling allows much of the internal stresses to relax before the martensite forms, decreasing the brittleness of the steel. However, the martempered steel will usually need to undergo further tempering to adjust the hardness and toughness, except in rare cases where maximum hardness is needed but the accompanying brittleness is not. Modern files are often martempered.[14]
Physical processes
Tempering involves a three-step process in which unstable martensite decomposes into ferrite and unstable carbides, and finally into stable cementite, forming various stages of a microstructure called tempered martensite. The martensite typically consists of laths (strips) or plates, sometimes appearing acicular (needle-like) or lenticular (lens-shaped). Depending on the carbon content, it also contains a certain amount of "retained austenite." Retained austenite are crystals that are unable to transform into martensite, even after quenching below the martensite finish (Mf) temperature. An increase in alloying agents or carbon content causes an increase in retained austenite. Austenite has much higher stacking-fault energy than martensite or pearlite, lowering the wear resistance and increasing the chances of galling, although some or most of the retained austenite can be transformed into martensite by cold and cryogenic treatments prior to tempering.
The martensite forms during a diffusionless transformation, in which the transformation occurs due to shear stresses created in the crystal lattices rather than by chemical changes that occur during precipitation. The shear stresses create many defects, or "dislocations," between the crystals, providing less-stressful areas for the carbon atoms to relocate. Upon heating, the carbon atoms first migrate to these defects and then begin forming unstable carbides. This reduces the amount of total martensite by changing some of it to ferrite. Further heating reduces the martensite even more, transforming the unstable carbides into stable cementite.
The first stage of tempering occurs between room temperature and 200 °C (392 °F). In the first stage, carbon precipitates into ε-carbon (Fe2,4C). In the second stage, occurring between 150 °C (302 °F) and 300 °C (572 °F), the retained austenite transforms into a form of lower-bainite containing ε-carbon rather than cementite (archaically referred to as "troostite").
Embrittlement
Embrittlement occurs during tempering when, through a specific temperature range, the steel experiences an increase in hardness and a reduction in ductility, as opposed to the normal decrease in hardness that occurs on either side of this range. The first type is called tempered martensite embrittlement (TME) or one-step embrittlement. The second is referred to as temper embrittlement (TE) or two-step embrittlement.
One-step embrittlement usually occurs in carbon steel at temperatures between 230 °C (446 °F) and 290 °C (554 °F), and was historically referred to as "500 degree [Fahrenheit] embrittlement." This embrittlement occurs due to the precipitation of
Two-step embrittlement typically occurs by aging the metal within a critical temperature range, or by slowly cooling it through that range, For carbon steel, this is typically between 370 °C (698 °F) and 560 °C (1,040 °F), although impurities like phosphorus and sulfur increase the effect dramatically. This generally occurs because the impurities are able to migrate to the grain boundaries, creating weak spots in the structure. The embrittlement can often be avoided by quickly cooling the metal after tempering. Two-step embrittlement, however, is reversible. The embrittlement can be eliminated by heating the steel above 600 °C (1,112 °F) and then quickly cooling.[19]
Alloy steels
Many elements are often alloyed with steel. The main purpose for alloying most elements with steel is to increase its
Most alloying elements (solutes) have the benefit of not only increasing hardness, but also lowering both the martensite start temperature and the temperature at which austenite transforms into ferrite and cementite. During quenching, this allows a slower cooling rate, which allows items with thicker cross-sections to be hardened to greater depths than is possible in plain carbon steel, producing more uniformity in strength.
Tempering methods for alloy steels may vary considerably, depending on the type and amount of elements added. In general, elements like
However, during tempering, elements like chromium, vanadium, and molybdenum precipitate with the carbon. If the steel contains fairly low concentrations of these elements, the softening of the steel can be retarded until much higher temperatures are reached, when compared to those needed for tempering carbon steel. This allows the steel to maintain its hardness in high-temperature or high-friction applications. However, this also requires very high temperatures during tempering, to achieve a reduction in hardness. If the steel contains large amounts of these elements, tempering may produce an increase in hardness until a specific temperature is reached, at which point the hardness will begin to decrease.[20][21] For instance, molybdenum steels will typically reach their highest hardness around 315 °C (599 °F) whereas vanadium steels will harden fully when tempered to around 371 °C (700 °F). When very large amounts of solutes are added, alloy steels may behave like precipitation-hardening alloys, which do not soften at all during tempering.[22]
Cast iron
Cast iron comes in many types, depending on the carbon content. However, they are usually divided into grey and white cast iron, depending on the form that the carbides take. In grey cast iron, the carbon is mainly in the form of graphite, but in white cast iron, the carbon is usually in the form of cementite. Grey cast iron consists mainly of the microstructure called pearlite, mixed with graphite and sometimes ferrite. Grey cast iron is usually used as cast, with its properties being determined by its composition.
White cast iron is composed mostly of a microstructure called
White tempering
Malleable (porous) cast iron is manufactured by white tempering. White tempering is used to burn off excess carbon, by heating it for extended amounts of time in an oxidizing environment. The cast iron will usually be held at temperatures as high as 1,000 °C (1,830 °F) for as long as 60 hours. The heating is followed by a slow cooling rate of around 10 °C (18 °F) per hour. The entire process may last 160 hours or more. This causes the cementite to decompose from the ledeburite, and then the carbon burns out through the surface of the metal, increasing the malleability of the cast iron.[23]
Black tempering
Ductile (non-porous) cast iron (often called "black iron") is produced by black tempering. Unlike white tempering, black tempering is done in an inert gas environment, so that the decomposing carbon does not burn off. Instead, the decomposing carbon turns into a type of graphite called "temper graphite" or "flaky graphite," increasing the malleability of the metal. Tempering is usually performed at temperatures as high as 950 °C (1,740 °F) for up to 20 hours. The tempering is followed by slow cooling through the lower critical temperature, over a period that may last from 50 to over 100 hours.[23]
Precipitation hardening alloys
Precipitation-hardening alloys first came into use during the early 1900s. Most heat-treatable alloys fall into the category of precipitation-hardening alloys, including alloys of
are also precipitation-hardening alloys. These alloys become softer than normal when quenched and then harden over time. For this reason, precipitation hardening is often referred to as "aging."Although most precipitation-hardening alloys will harden at room temperature, some will only harden at elevated temperatures and, in others, the process can be sped up by aging at elevated temperatures. Aging at temperatures higher than room-temperature is called "artificial aging". Although the method is similar to tempering, the term "tempering" is usually not used to describe artificial aging, because the physical processes, (i.e.: precipitation of
See also
- Annealing (metallurgy)
- Austempering
- Precipitation strengthening
- Tempered glass
References
- ^ Light, its interaction with art and antiquities By Thomas B. Brill - Plenum Publishing 1980 Page 55
- ^ Andrews, Jack (1994). New Edge of the Anvil: a resource book for the blacksmith. pp. 98–99
- ^ a b c d e Steel metallurgy for the non-metallurgist By John D. Verhoeven - ASM International 2007 Page 99-105
- ^ The Medieval Sword in the Modern World By Michael 'Tinker' Pearce - 2007 Page 39
- ^ Tool steels By George Adam Roberts, George Krauss, Richard Kennedy, Richard L. Kennedy - ASM International 1998 Page 2
- ^ Roberts-Austen By Sir William Chandler Roberts-Austen, Sydney W. Smith - Charles Griffin & Co. 1914 Page 155-156
- S2CID 135890256.
- ^ Steel castings handbook By Malcolm Blair, Thomas L. Stevens - Steel Founders' Society of America and ASM International Page 24-9
- ^ Practical heat treating By Jon L. Dossett, Howard E. Boyer - ASM International 2006 Page 112
- ^ How To Weld By Todd Bridigum - Motorbook 2008 Page 37
- ^ Practical Blacksmithing and Metalworking By Percy W. Blandford - TAB Books 1988 Page 3, 74–75
- ^ Practical Blacksmithing and Metalworking By Percy W. Blandford - TAB Books 1988 Page 74-75
- ^ Knife Talk II: The High Performance Blade By Ed Fowler - Krause Publications 2003 Page 114
- ^ a b c Elements of metallurgy and engineering alloys By Flake C. Campbell - ASM International 2008 Page 195-196
- ^ Steel Heat Treatment Handbook By George E. Totten -- Marcel Dekker 1997 Page 659
- ^ Phase Transformations in Steels, Volume 1: Fundamentals and Diffusion-Controlled Transformations by Elena Pereloma, David V Edmonds -- Woodhead Publishing 2012 Page 20--39
- ^ Light Microscopy of Carbon Steels by Leonard Ernest Samuels ASM International 1999 Page 20--25
- ^ Principles of Heat Treatment of Steel By Romesh C. Sharma - New Age International (P) Limited 2003 Page 101-110
- ^ Elements of metallurgy and engineering alloys By Flake C. Campbell - ASM International 2008 Page 197
- ^ "Hardenable Alloy Steels :: Total Materia Article". www.keytometals.com.
- ^ Steel Heat Treatment: Metallurgy and Technologies By George E. Totten -- CRC Press 2007 Page 6, 200--203
- ^ Steels: Microstructure and Properties: Microstructure and Properties By Harry Bhadeshia, Robert Honeycombe -- Elsevier 2006Page 191--207
- ^ a b c Physical metallurgy for engineers By Miklós Tisza - ASM International 2002 Page 348-350
Further reading
- Manufacturing Processes Reference Guide by Robert H. Todd, Dell K. Allen, and Leo Alting pg. 410

