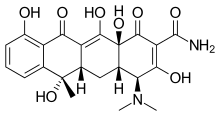
Tetracycline
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌtɛtrəˈsaɪkliːn/ |
| Other names | TE/TET/TC/TCY[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682098 |
| License data |
|
| Pregnancy category |
|
QJ51AA07 (WHO) | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80% |
| Metabolism | Not metabolized |
| Elimination half-life | 8–11 hours, 57–108 hours (kidney impairment) |
| Excretion | Urine (>60%), feces |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Tetracycline, sold under various brand names, is an oral
Common side effects include vomiting,
Tetracycline was patented in 1953
Medical uses
Spectrum of activity
Tetracyclines have a broad spectrum of antibiotic action. Originally, they possessed some level of bacteriostatic activity against almost all medically relevant
It is also one of a group of antibiotics which together may be used to treat
The following list presents MIC susceptibility data for some medically significant microorganisms:
- Escherichia coli: 1 μg/mL to >128 μg/mL
- Shigella spp.: 1 μg/mL to 128 μg/mL[11]
Anti-eukaryote use
The tetracyclines also have activity against certain
Use as a biomarker

Since tetracycline is absorbed into bone, it is used as a marker of bone growth for
Tetracycline is also used as a biomarker in wildlife to detect consumption of medicine- or vaccine-containing baits.[14]
Side effects
This section needs more primary sources. (November 2022) |  |
Use of tetracycline antibiotics can:[15]
- Discolor permanent teeth (yellow-gray-brown), from prenatal period through childhood and adulthood.[16] Children receiving long- or short-term therapy with a tetracycline or glycylcycline may develop permanent brown discoloration of the teeth.
- Be inactivated by calcium ions, so are not to be taken with milk, yogurt, and other dairy products
- Be inactivated by aluminium, iron, and zinc ions, not to be taken at the same time as indigestion remedies (some common antacids and over-the-counter heartburn medicines)
- Cause skin photosensitivity[17], so exposure to the sun or intense light is not recommended
- Cause drug-induced lupus, and hepatitis
- Cause microvesicular fatty liver[18]
- Cause tinnitus[19]
- Interfere with methotrexate by displacing it from the various protein-binding sites
- Cause breathing complications, as well as anaphylactic shock, in some individuals
- Affect bone growth of the fetus, so should be avoided during pregnancy
- Fanconi syndrome may result from ingesting expired tetracyclines.
Caution should be exercised in long-term use when breastfeeding. Short-term use is safe; bioavailability in milk is low to nil.[20] According to the U.S. Food and Drug Administration (FDA), cases of Stevens–Johnson syndrome, toxic epidermal necrolysis, and erythema multiforme associated with doxycycline use have been reported, but a causative role has not been established.[21]
Pharmacology
Mechanism of action
Tetracycline inhibits protein synthesis by blocking the attachment of charged
Mechanisms of resistance
Bacteria usually acquire resistance to tetracycline from horizontal transfer of a gene that either encodes an efflux pump or a ribosomal protection protein. Efflux pumps actively eject tetracycline from the cell, preventing the build up of an inhibitory concentration of tetracycline in the cytoplasm.[24] Ribosomal protection proteins interact with the ribosome and dislodge tetracycline from the ribosome, allowing for translation to continue.[25]
History
Discovery
The tetracyclines, a large family of antibiotics, were discovered by
Evidence in antiquity
Tetracycline has a high affinity for calcium and is incorporated into bones during the active mineralization of hydroxyapatite. When incorporated into bones, tetracycline can be identified using ultraviolet light.[30]
There is evidence that early inhabitants of Northeastern Africa consumed tetracycline antibiotics.
It is conjectured that the beer brewed by the Nubians was the source of the tetracycline found in these bones.[32]
Society and culture
Price
According to data from EvaluatePharma and published in the
Names
It is marketed under the brand names Sumycin, Tetracyn, and Panmycin, among others. Actisite is a thread-like fiber formulation used in dental applications.[citation needed]
It is also used to produce several semisynthetic derivatives, which together are known as the tetracycline antibiotics. The term "tetracycline" is also used to denote the four-ring system of this compound; "tetracyclines" are related substances that contain the same four-ring system.[citation needed]
Media
Due to the drug's association with fighting infections, it serves as the main "commodity" in the science fiction series Aftermath, with the search for tetracycline becoming a major preoccupation in later episodes.[34]
Tetracycline is also represented in Bohemia Interactive's survival sandbox, DayZ. In the game, players may find the antibiotic to treat the common cold, influenza, cholera and infected wounds, but does not portray any side effects associated with tetracycline.
Research
Genetic engineering
In genetic engineering, tetracycline is used in transcriptional activation. It has been used as an engineered "control switch" in chronic myelogenous leukemia models in mice. Engineers were able to develop a retrovirus that induced a particular type of leukemia in mice, and could then "switch" the cancer on and off through tetracycline administration. This could be used to grow the cancer in mice and then halt it at a particular stage to allow for further experimentation or study.[35]
A technique being developed for the control of the
References
- ^ "Antibiotic abbreviations list". Retrieved 22 June 2023.
- ^ "Tetracycline". PubChem. National Center for Biotechnology Information.
- ^ a b c d e f g h i "Tetracycline". The American Society of Health-System Pharmacists. Archived from the original on 28 December 2016. Retrieved 8 December 2016.
- ^ U.S. patent 2699054A
- ^ S2CID 34647314.
- ISBN 9783527607495. Archivedfrom the original on 2016-12-20.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Lippincott's Illustrated Reviews: Pharmacology, 4th ed. Harvery RA, Champe, PC. Lippincott, Williams & Wilkins, 2009
- PMID 25772356.
- PMID 26475870.
- ^ "Tetracycline hydrochloride" (PDF). Susceptibility and Minimum Inhibitory Concentration (MIC) Data. TOKU-E. 8 September 2015. Archived from the original (PDF) on 8 September 2015.
- ^ Mayton CA. "Tetracycline labeling of bone". Archived from the original on 2007-03-12.
- ^ "Tetracycline Labeling". The Johns Hopkins Medical Institutions. 8 January 2001. Archived from the original on 2012-12-15.
- S2CID 35102508. Archived from the originalon 2013-04-15.
- ^ "Tetracycline: MedlinePlus Drug Information". medlineplus.gov. Archived from the original on 2017-05-10. Retrieved 2017-05-19.
- PMID 15485524.
- PMID 31751095, retrieved 2024-03-19
- PMID 31644155, retrieved 2024-03-20
- ^ Schlossberg DL, Samuel R (2017). Antibiotics Manual : A Guide to Commonly Used Antimicrobials. John Wiley & Sons, Inc. p. 367 – via ProQuest Ebook Central.
- ^ Riordan J, Wambach K, eds. (November 2010). Breastfeeding and Human Lactation. Jones & Bartlett Learning. p. 179.
- ^ "FDA Adverse Events Reporting System". Food and Drug Administration. 27 August 2010. Archived from the original on 2011-01-17. Retrieved 14 January 2011.
- ^ Mehta A (2011-05-27). "Mechanism of Action of Tetracyclines". Pharmaxchange.info. Archived from the original on 2012-06-05. Retrieved 2012-06-07.
- ^ Todar K (2012). "Antimicrobial Agents in the Treatment of Infectious Disease.". Todars Online Text Book of Bacteriology. Archived from the original on 2013-10-08. Retrieved 2013-08-27.
- PMID 11381101.
- PMID 14638464.
- better source needed]
- PMID 3903946.
- ISSN 0002-7863.
- ISSN 0002-7863.
- PMID 20456523.
- PMID 7001623.
- ^ Armelagos G (2000). "Take Two Beers and Call Me in 1,600 Years: Use of Tetracycline by Nubians and Ancient Egyptians". Natural History. 109 (4): 50–53.
- ^ a b McCluskey PD (6 November 2015). "As competition wanes, prices for generics skyrocket". Boston Globe. Archived from the original on 19 November 2015. Retrieved 18 November 2015.
- ^ "Aftermath Episode Recap". SyFy Channel. Archived from the original on 13 October 2017. Retrieved 15 April 2020.
- S2CID 40155100.
- ^ Urquhart C (15 July 2012). "Can GM mosquitoes rid the world of a major killer?". The Observer. Archived from the original on 5 December 2013. Retrieved 2012-07-15.
External links
- "Tetracycline". Drug Information Portal. U.S. National Library of Medicine.
