Tetrazole
| |||
| Identifiers | |||
|---|---|---|---|
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.005.477 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CH2N4 | |||
| Molar mass | 70.05 g/mol | ||
| Density | 1.477 g/mL | ||
| Melting point | 157 to 158 °C (315 to 316 °F; 430 to 431 K)[2] | ||
| Boiling point | 220 ± 23 °C (428 ± 41 °F; 493 ± 23 K) | ||
| Acidity (pKa) | 4.90[1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Tetrazoles are a class of synthetic organic heterocyclic compound, consisting of a 5-member ring of four nitrogen atoms and one carbon atom. The name tetrazole also refers to the parent compound with formula CH2N4, of which three isomers can be formulated.
Structure and bonding
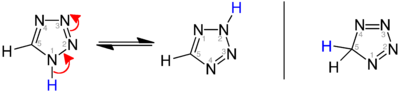
Three isomers of the parent tetrazole exist, differing in the position of the double bonds: 1H-, 2H-, and 5H-tetrazole. The 1H- and 2H- isomers are
Synthesis
1H-Tetrazole was first prepared by the reaction of anhydrous
2-Aryl-2H-tetrazoles are synthesized by a [3+2] cycloaddition reaction between an aryl diazonium and trimethylsilyldiazomethane.[10]
Uses
There are several pharmaceutical agents which are tetrazoles, including several cephalosporin-class antibiotics. Tetrazoles can act as bioisosteres for carboxylate groups because they have similar pKa and are deprotonated at physiological pH. Angiotensin II receptor blockers — such as losartan and candesartan, often are tetrazoles. A well-known tetrazole is dimethyl thiazolyl diphenyl tetrazolium bromide (MTT). This tetrazole is used in the
Some tetrazole
salts of nitrogen bases.Other tetrazoles are used for their explosive or combustive properties, such as tetrazole itself and
1H-Tetrazole and 5-(benzylthio)-1H-tetrazole (BTT) are widely used as acidic activators of the coupling reaction in oligonucleotide synthesis.[18]
2-Tetrazoles can undergo controlled

Related heterocycles
- Triazoles, analogs with three nitrogen atoms
- Pentazole, the analog with five nitrogen atoms (strictly speaking, an inorganic homocycle, not a heterocycle)
- Oxatetrazole
- Thiatetrazole
References
- doi:10.1039/b203118c.
- .
- ISSN 0108-2701.
- ^ PMID 21322546.
- J. Chem. Soc.Perkin Trans. 2 1983, 379.
- ISSN 0002-7863.
- ISSN 1364-5471.
- ISSN 0002-7863.
- ISSN 1521-3773.
- PMID 27041776.
- PMID 2922273.
- PMID 25224009.
- PMID 26787697.
- ^ "Greener explosives show promise". Chemistry World. 2 October 2008.
- .
- )
- PMID 22214278.
- .
- .
- .
- .



