Thallium(III) oxide
| |
| Names | |
|---|---|
| Other names
thallium trioxide, thallium sesquioxide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.013.846 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| Tl2O3 | |
| Molar mass | 456.76 g/mol |
| Appearance | dark brown solid |
| Density | 10.19 g/cm3, solid (22 °C) |
| Melting point | 717 °C (1,323 °F; 990 K) |
| Boiling point | 875 °C (1,607 °F; 1,148 K) (decomposes) |
| insoluble | |
| +76.0·10−6 cm3/mol | |
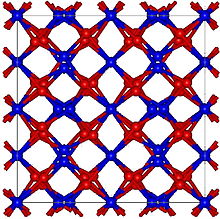
| Structure | |
| Cubic, (Bixbyite) cI80[2] | |
| Ia3 (No. 206) | |
| Hazards | |
| GHS labelling:[3] | |
  
| |
| Danger | |
| H300+H330, H373, H411 | |
| P273, P301+P310+P330, P304+P340+P310, P314 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
44 mg/kg (oral, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thallium(III) oxide, also known as thallic oxide, is a chemical compound of
MOCVD is known.[6] Any practical use of thallium(III) oxide will always have to take account of thallium's poisonous nature. Contact with moisture and acids
may form poisonous thallium compounds.
Production
It is produced by the reaction of thallium with oxygen or hydrogen peroxide in an alkaline thallium(I) solution. Alternatively, it can be created by the oxidation of thallium(I) nitrate by chlorine in an aqueous potassium hydroxide solution.[7]
References
- ISBN 0-8493-0462-8..
- .
- ^ GHS: Sigma-Aldrich 204617
- ^ http://www.handbookofmineralogy.org/pdfs/avicennite.pdf Handbook of Mineralogy
- S2CID 96808351.
- .
- ISBN 3-432-87813-3(in German)

