Thiocyanate

| |
| Names | |
|---|---|
| Preferred IUPAC name
Thiocyanate[1] | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
IUPHAR/BPS |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| [SCN]− | |
| Molar mass | 58.08 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thiocyanates are
Thiocyanate is analogous to the cyanate ion, [OCN]−, wherein oxygen is replaced by sulfur. [SCN]− is one of the pseudohalides, due to the similarity of its reactions to that of halide ions. Thiocyanate used to be known as rhodanide (from a Greek word for rose) because of the red colour of its complexes with iron.
Thiocyanate is produced by the reaction of elemental sulfur or thiosulfate with cyanide:
Oxidation of thiocyanate inevitably produces
Biological chemistry of thiocyanate in medicine
Thiocyanate[4] is known to be an important part in the biosynthesis of hypothiocyanite by a lactoperoxidase.[5][6][7] Thus the complete absence of thiocyanate or reduced thiocyanate[8] in the human body, (e.g., cystic fibrosis) is damaging to the human host defense system.[9][10]
Thiocyanate is a potent competitive inhibitor of the thyroid
In the early 20th century, thiocyanate was used in the treatment of hypertension, but it is no longer used because of associated toxicity.[13] Sodium nitroprusside, a metabolite of which is thiocyanate, is however still used for the treatment of a hypertensive emergency. Rhodanese catalyzes the reaction of sodium nitroprusside (like other cyanides) with thiosulfate to form the metabolite thiocyanate.
Coordination chemistry
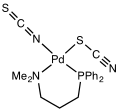
Thiocyanate shares its negative charge approximately equally between sulfur and nitrogen. As a consequence, thiocyanate can act as a nucleophile at either sulfur or nitrogen—it is an ambidentate ligand. [SCN]− can also bridge two (M−SCN−M) or even three metals (>SCN− or −SCN<). Experimental evidence leads to the general conclusion that class A metals (hard acids) tend to form N-bonded thiocyanate complexes, whereas class B metals (soft acids) tend to form S-bonded thiocyanate complexes. Other factors, e.g. kinetics and solubility, are sometimes involved, and linkage isomerism can occur, for example [Co(NH3)5(NCS)]Cl2 and [Co(NH3)5(SCN)]Cl2.[15] It [SCN] is considered as a weak ligand. ([NCS] is a strong ligand)[16]
Test for iron(III) and cobalt(II)
If [SCN]− is added to a solution with
Similarly, Co2+ gives a blue complex with thiocyanate.[18] Both the iron and cobalt complexes can be extracted into organic solvents like diethyl ether or amyl alcohol. This allows the determination of these ions even in strongly coloured solutions. The determination of Co(II) in the presence of Fe(III) is possible by adding KF to the solution, which forms uncoloured, very stable complexes with Fe(III), which no longer react with SCN−.[19]
Phospholipids or some detergents aid the transfer of thiocyanatoiron into chlorinated solvents like chloroform and can be determined in this fashion.[20]
See also
References
- ISBN 978-0-08-037941-8.
Citations
- ISBN 978-0-85404-182-4.
- PMID 25708890.
- .
- PMID 17404297.
- PMID 17204267.
- PMID 6340603.
- PMID 352945.
- PMID 18519245.[permanent dead link]
- PMID 17082494.
- PMID 19918082.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 15572417.
- ^ "Hypothyroidism". umm.edu. University of Maryland Medical Center. Retrieved 3 December 2014.
- PMID 18126744.
- ISSN 0020-1669.
- ^ Greenwood, p. 326
- ^ "coordination compounds" (PDF).
- ^ Greenwood, p. 1090
- PMID 18917685.
- ISSN 0369-0261.
- PMID 6892980.





