Thogotovirus
| Thogotovirus | |
|---|---|

| |
Electron micrograph of Bourbon virus (scale bar: 100 nm)
| |
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Kingdom: | Orthornavirae |
| Phylum: | Negarnaviricota
|
| Class: | Insthoviricetes |
| Order: | Articulavirales |
| Family: | Orthomyxoviridae |
| Genus: | Thogotovirus |
| Species | |
| |
| Synonyms[1] | |
|
Thogoto-like viruses | |
Thogotovirus is a
The genus contains the species Thogoto thogotovirus and Dhori virus (DHOV), and the latter's subtype Batken virus, as well as the species or strains Araguari virus, Aransas Bay virus (ABV), Bourbon virus, Jos virus (JOSV) and Upolu virus (UPOV), which have yet to be confirmed by the International Committee on Taxonomy of Viruses (ICTV). A wide range of mammals are infected by members of the genus; some types also infect birds. THOV causes disease in livestock. THOV, DHOV and Bourbon virus can infect humans, and have occasionally been associated with human disease.
History
Virology


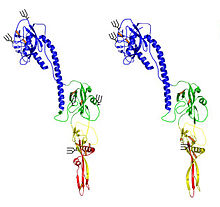
The virus particle is
Viral proteins
The genome encodes 7–9 proteins, including the trimeric
The thogotovirus glycoprotein is not similar to the influenza virus glycoproteins (
THOV and JOSV
As in all orthomyxoviruses, the largest three segments (1–3) encode the three subunits of the RNA polymerase. In thogotoviruses, segment 4 encodes the glycoprotein and segment 5 the nucleoprotein. The messenger RNA (mRNA) from segment 6 can be spliced to encode the matrix protein or unspliced to encode ML, which has 38 additional amino acids at its C-terminus.[4][15][16][18] No product has yet been identified for the seventh segment, observed in DHOV.[18]
Life cycle

The receptor on the vertebrate host cell is
As orthomyxoviruses do not encode a capping enzyme, initiation of transcription involves the virus cutting the cap off the 5′-end of host mRNAs, so that the mRNA is recognised by the host translation machinery. A similar "cap snatching" process is used by other orthomyxoviruses, but a much longer host RNA sequence is cleaved along with the cap and incorporated into the viral mRNA.[28][29]
The virus assembles by the cell membrane and leaves the cell by budding.[16] For THOV grown in baby hamster kidney cells, virus particles start to be released 6–8 hours after infection, with substantial quantities still being produced 24 hours after infection. This growth rate is slower than that of influenza viruses, and is more similar to Quaranfil virus.[17][21]
Epidemiology
Most thogotoviruses have been shown to infect arthropods, generally
Transmission to vertebrates usually occurs via a tick
Host interaction and disease
Ticks
No major pathological changes are observed in Rhipicephalus appendiculatus ticks infected with THOV.[17] The virus is concentrated in the synganglion (the tick brain) early on in the blood-feeding process, with the proportion of virus located in the salivary glands increasing during the late phase of blood-feeding.[17][33] Lower levels of virus are found in the trachea, digestive tract and female sex organs, but not in the male sex organs or the excretory system. The high level of virus present in the synganglion has been proposed to help the virus persist through the metamorphosis of the tick, as the nervous system undergoes less remodeling than other systems.[17]
Vertebrates
In the laboratory setting, several members of the genus cause severe disease in mice and hamsters.[5][6] Systemic spread of the virus occurs, with pathological effects present in multiple organs and systems, including the brain, liver, lymphatic system, and sometimes the lungs and small intestine.[5][6] Lymphocytes are a major target cell for DHOV.[6] DHOV infection in mice resembles experimental influenza infection in mice and ferrets as well as fatal H5N1 influenza infection of humans, and has been proposed as a model for this disease.[6]
Natural infections with thogotoviruses in mammals generally do not appear to result in symptoms. THOV is a significant veterinary pathogen, for example, causing a febrile illness and
Treatment and prevention
No specific treatment or vaccine is available for thogotoviruses, as of February 2015. The antiviral drug ribavirin, which has a broad spectrum of activity that includes some other orthomyxoviruses,[40] has been shown to inhibit DHOV replication in vitro in a single study.[41] Supportive therapy is used for THOV disease,[39] and has been recommended by the US Centers for Disease Control and Prevention for infection with Bourbon virus. As with other arboviruses, avoidance of contact with the vector is central to prevention.[42]
Species and strains
Two species have been confirmed by the ICTV, THOV and DHOV.[43] The two viruses have a low degree of sequence identity (37% for the nucleoprotein; 31% for the envelope glycoprotein), and their antibodies do not crossreact.[13] Batken virus is a subtype of DHOV.[13] As of February 2015, a further five species or strains have been suggested as belonging to the genus.[7]
| Species/strain | RNA segments | Diameter (nm) | Vectors | Vertebrate hosts | Distribution |
|---|---|---|---|---|---|
| Araguari | 6 | 105 | Unknown | Gray four-eyed opossum, mouse | S. America |
| Aransas Bay | 6 | 75–140 | Ornithodoros ticks | Mouse | N. America |
| Batken | 50–100 | Hyalomma ticks, Aedes and Culex mosquitoes | Chicken, hamster, mouse | Asia | |
| Bourbon | ≥6 | ~100–130 | Unknown | Human | N. America |
| Dhori | 7 | Hyalomma ticks | Birds, hare, horse, human, mouse, ruminants | Africa, Asia, Europe | |
| Jos | ≥6 | 85–120 | Amblyomma and Rhipicephalus ticks | Mouse, zebu | Africa |
| Thogoto | 6 | 100 | Amblyomma, Hyalomma and Rhipicephalus ticks | Banded mongoose, donkey, human, rodents, ruminants | Africa, Asia, Europe |
| Upolu | 6 | 75–120 | Ornithodoros ticks | Mouse | Australia |
THOV-like viruses

Thogoto virus (THOV)
Antibodies have been found to THOV in rats and many domestic animals, including goats, sheep, donkeys, camels, cattle and buffaloes, and the virus has been isolated from the wild banded mongoose (Mongos mungo).[13][32] It causes significant livestock disease, including a febrile illness and abortion in sheep.[5][36][37][38] In artificial laboratory infections, it is highly pathogenic in hamsters and also infects mice.[5] The virus is known to infect humans in natural settings.[7][31]
The virus particle is generally spherical with some filamentous forms; the diameter is around 100 nm.[17] The genome has six RNA segments.[4]
Araguari virus
The Araguari virus was first isolated from a Gray four-eyed opossum (Philander opossum) in Serra do Navio, Amapá, Brazil in 1969.[14] Its method of transmission is unknown.[6] In laboratory infections, it is pathogenic to mice. The virion is around 105 nm in diameter. The genome has six RNA segments. Based on partial sequence data the virus was found to be most closely related to THOV.[14]
Aransas Bay virus (ABV)
ABV was found in the soft-bodied tick genus Ornithodoros in seabird nests in southern Texas, USA, in 1975; it was the first member of the genus to be found in North America.[7][15] No natural vertebrate host has been identified, but the virus is highly pathogenic to mice in laboratory infections. The virus particle is spherical or ovoid, with a range of sizes, from 75 nm × 85 nm to 120 nm × 140 nm. The genome has six RNA segments. It is most similar to UPOV, with some similarity to THOV and JOSV.[15]
Jos virus (JOSV)

JOSV was first isolated from the zebu (Bos indicus) in Jos, Nigeria in 1967. It has since been found infecting Amblyomma and Rhipicephalus hard-bodied ticks in several countries across Africa. In the laboratory it causes severe pathology in mice. The virus particle has a variable, usually ovoid, morphology with a diameter of 85–120 nm. The genome contains at least six RNA segments.[2] It has some sequence similarities with UPOV and ABV.[15]
Upolu virus (UPOV)
UPOV was first isolated on
DHOV-like viruses
Dhori virus (DHOV)
DHOV was first isolated from Hyalomma dromedarii hard-bodied ticks infesting camels in Gujarat, India, in 1961.[6] It has since been observed in eastern Russia, Pakistan, Egypt, Saudi Arabia, Kenya and southern Portugal. The vector is usually a species of Hyalomma, such as H. marginatum.[6][13][31][46][47]
Where DHOV is prevalent, antibodies to the virus have been documented in camels, goats, horses, cattle and humans.
DHOV has seven RNA segments.[13]

Batken virus
Bourbon virus
Oz virus
Oz virus was first characterised in 2018 after isolation from the hard tick Amblyomma testudinarium in Ehime, Japan. [DOI: 10.1016/j.virusres.2018.03.004]. The first human case, a 70 year old female patient who died of myocarditis with isolation of Oz virus on autopsy, was reported on 23.6.2023 by the Japanese Ministry of Heath.
Notes and references
- PMID 8992952. Retrieved 4 June 2019.
- ^ PMID 21994326
- ^ Members of the recently ratified genus Quaranjavirus are also transmitted by ticks.[2]
- ^ PMID 20861272
- ^ PMID 14329965
- ^ PMID 17426188
- ^ PMID 25899080.
- ^ PMID 3445590
- ^ PMID 9349464
- PMID 6858001.
- ^ ICTV 6th report (PDF), International Committee on Taxonomy of Viruses, retrieved 9 March 2015
- ^ Pringle CR, Virus Taxonomy 1996 - A Bulletin from the Xth International Congress of Virology in Jerusalem (PDF), International Committee on Taxonomy of Viruses, retrieved 9 March 2015
- ^ a b c d e f g h i j k Büchen-Osmond C, ed. (2006), "Index of Viruses - Orthomyxoviridae", ICTVdB - The Universal Virus Database, version 4, Columbia University, archived from the original on 2 April 2015, retrieved 3 July 2020
- ^ PMID 16354811.
- ^ PMID 24574415
- ^ a b c d e f g "Thogotovirus", ViralZone, Swiss Institute of Bioinformatics, retrieved 6 March 2015
- ^ PMID 2543769.
- ^ a b c King AM, Adams MJ, Carstens EB, Lefkowitz EJ, eds. (2011), Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses, Academic Press, p. 757
- ^ PMID 3655743
- PMID 1733105
- ^ PMID 19726499
- PMID 25392223.
- PMID 18282283
- PMID 16364743
- ^ Thogoto virus: Molecular biology of a tick-transmitted Orthomyxovirus, Universitäts Klinikum Freiburg, retrieved 7 March 2015
- ^ PMID 16223950
- PMID 10051598
- PMID 9343188
- PMID 24454773
- ^ S2CID 33344597
- ^ a b c d e f g Gratz N (2006), Vector- and Rodent-Borne Diseases in Europe and North America: Distribution, Public Health Burden, and Control, Cambridge University Press, pp. 108–9
- ^ PMID 10466973
- ^ PMID 12932755
- PMID 3616608.
- PMID 2617637.
- ^ CABI, p. 110
- ^ a b Maramorosch K, McIntosh AH (1994), Arthropod Cell Culture Systems, CRC Press, p. 165
- ^ PMID 7745744
- ^ a b c d e Berger SA, Calisher CH, Keystone JS (2003), Exotic Viral Diseases: A Global Guide, BC Decker, pp. 179–80
- S2CID 43106875
- PMID 18318134
- ^ "Bourbon virus", CDC website, Division of Vector-Borne Diseases, CDC, retrieved 4 March 2015
- ^ ICTV Master Species List 2014 v2, International Committee on Taxonomy of Viruses, 2014, archived from the original on 9 March 2015, retrieved 7 March 2015
- ^ PMID 3688319
- PMID 16836823
- PMID 6415873
- PMID 18060218
- ^ L'vov DN, Dzharkenov AF, Aristova VA, Kovtunov AI, Gromashevskiĭ VL (2002), "[The isolation of Dhori viruses (Orthomyxoviridae, Thogotovirus) and Crimean-Congo hemorrhagic fever virus (Bunyaviridae, Nairovirus) from the hare (Lepus europaeus) and its ticks Hyalomma marginatum in the middle zone of the Volga delta, Astrakhan region, 2001]", Voprosy virusologii (in Russian), 47: 32–36
- PMID 18590134
- ^ PMID 25069283
