Thrombophilia
| Thrombophilia | |
|---|---|
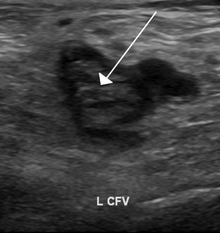 | |
| An ultrasound image demonstrating a blood clot in the left common femoral vein. | |
| Specialty | Hematology |
Thrombophilia (sometimes called hypercoagulability or a prothrombotic state) is an abnormality of
There is no specific treatment for most thrombophilias, but recurrent episodes of thrombosis may be an indication for long-term preventive
Signs and symptoms

The most common conditions associated with thrombophilia are
Venous thrombosis may also occur in more unusual places: in the
Thrombophilia has been linked to
Protein C deficiency may cause purpura fulminans, a severe clotting disorder in the newborn that leads to both tissue death and bleeding into the skin and other organs. The condition has also been described in adults. Protein C and protein S deficiency have also been associated with an increased risk of skin necrosis on commencing anticoagulant treatment with warfarin or related drugs.[2][12]
Causes
Thrombophilia can be congenital or acquired. Congenital thrombophilia refers to inborn conditions (and usually hereditary, in which case "hereditary thrombophilia" may be used) that increase the tendency to develop thrombosis, while, on the other hand, acquired thrombophilia refers to conditions that arise later in life.
Congenital
The most common types of congenital thrombophilia are those that arise as a result of overactivity of coagulation factors. They are relatively mild, and are therefore classified as "type II" defects.
The rare forms of congenital thrombophilia are typically caused by a deficiency of natural anticoagulants. They are classified as "type I" and are more severe in their propensity to cause thrombosis.
Blood group determines thrombosis risk to a significant extent. Those with blood groups other than type O are at a 2- to 4-fold relative risk. O blood group is associated with reduced levels of von Willebrand factor – because of increased clearance – and factor VIII, which is related to thrombotic risk .[5]
Acquired
A number of acquired conditions augment the risk of thrombosis. A prominent example is
Heparin-induced thrombocytopenia (HIT) is due to an immune system reaction against the anticoagulant drug heparin (or its derivatives).[1] Though it is named for associated low platelet counts, HIT is strongly associated with risk of venous and arterial thrombosis.[18] Paroxysmal nocturnal hemoglobinuria (PNH) is a rare condition resulting from acquired alterations in the PIGA gene, which plays a role in the protection of blood cells from the complement system. PNH increases the risk of venous thrombosis but is also associated with hemolytic anemia (anemia resulting from destruction of red blood cells).[19] Both HIT and PNH require particular treatment.[18][19]
Hematologic conditions associated with sluggish blood flow can increase risk for thrombosis. For example,
Cancer, particularly when metastatic (spread to other places in the body), is a recognised risk factor for thrombosis.[2][15] A number of mechanisms have been proposed, such as activation of the coagulation system by cancer cells or secretion of procoagulant substances. Furthermore, particular cancer treatments (such as the use of central venous catheters for chemotherapy) may increase the risk of thrombosis further.[21]
Nephrotic syndrome, in which protein from the bloodstream is released into the urine due to kidney diseases, can predispose to thrombosis;[1] this is particularly the case in more severe cases (as indicated by blood levels of albumin below 25 g/L) and if the syndrome is caused by the condition membranous nephropathy.[22] Inflammatory bowel disease (ulcerative colitis and Crohn's disease) predispose to thrombosis, particularly when the disease is active. Various mechanisms have been proposed.[2][23]
Unclear
A number of conditions that have been linked with venous thrombosis are possibly genetic and possibly acquired.[15] These include: elevated levels of factor VIII, factor IX, factor XI, fibrinogen and thrombin-activatable fibrinolysis inhibitor, and decreased levels of tissue factor pathway inhibitor. Activated protein C resistance that is not attributable to factor V mutations is probably caused by other factors and remains a risk factor for thrombosis.[15]
There is an association between the blood levels of
Mechanism
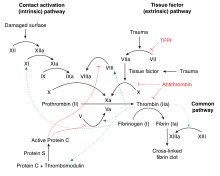
Thrombosis is a multifactorial problem because there are often multiple reasons why a person might develop thrombosis. These risk factors may include any combination of abnormalities in the blood vessel wall, abnormalities in the blood flow (as in immobilization), and abnormalities in the consistency of the blood. Thrombophilia is caused by abnormalities in blood consistency, which is determined by the levels of coagulation factors and other circulating blood proteins that participate in the "coagulation cascade".[15]
Normal coagulation is initiated by the release of
In thrombophilia, the balance between "procoagulant" and "anticoagulant" activity is disturbed. The severity of the imbalance determines the likelihood that someone develops thrombosis. Even small perturbances of proteins, such as the reduction of antithrombin to only 70–80% of the normal level, can increase the thrombosis risk; this is in contrast with
In addition to its effects on thrombosis, hypercoagulable states may accelerate the development of atherosclerosis, the arterial disease that underlies myocardial infarction and other forms of cardiovascular disease.[30][31]
Diagnosis

Tests for thrombophilia include
For hereditary cases, the patient must have at least two abnormal tests plus family history.
Screening
There are divergent views as to whether everyone with an unprovoked episode of thrombosis should be investigated for thrombophilia. Even those with a form of thrombophilia may not necessarily be at risk of further thrombosis, while recurrent thrombosis is more likely in those who have had previous thrombosis even in those who have no detectable thrombophilic abnormalities.
When venous thromboembolism occurs when a patient is experiencing transient major risk factors such as prolonged immobility, surgery, or trauma, testing for thrombophilia is not appropriate because the outcome of the test would not change a patient's indicated treatment.
In the United Kingdom, professional guidelines give specific indications for thrombophilia testing. It is recommended that testing be done only after appropriate counseling, and hence the investigations are usually not performed at the time when thrombosis is diagnosed but at a later time.
Recurrent miscarriage is an indication for thrombophilia screening, particularly antiphospholipid antibodies (anti-cardiolipin IgG and IgM, as well as lupus anticoagulant), factor V Leiden and prothrombin mutation, activated protein C resistance and a general assessment of coagulation through an investigation known as thromboelastography.[11]
Women who are planning to use oral contraceptives do not benefit from routine screening for thrombophilias, as the absolute risk of thrombotic events is low. If either the woman or a first-degree relative has had thrombosis, the risk of developing thrombosis is increased. Screening this selected group may be beneficial,[28] but even when negative may still indicate residual risk.[12] Professional guidelines therefore suggest that alternative forms of contraception be used rather than relying on screening.[12]
Thrombophilia screening in people with arterial thrombosis is generally regarded as unrewarding and is generally discouraged,
Treatment

There is no specific treatment for thrombophilia, unless it is caused by an underlying medical illness (such as nephrotic syndrome), where the treatment of the underlying disease is needed. In those with unprovoked and/or recurrent thrombosis, or those with a high-risk form of thrombophilia, the most important decision is whether to use anticoagulation medications, such as warfarin, on a long-term basis to reduce the risk of further episodes.[3] This risk needs to weighed against the risk that the treatment will cause significant bleeding, as the reported risk of major bleeding is over 3% per year, and 11% of those with major bleeding may die as a result.[3]
Apart from the abovementioned forms of thrombophilia, the risk of recurrence after an episode of thrombosis is determined by factors such as the extent and severity of the original thrombosis, whether it was provoked (such as by immobilization or pregnancy), the number of previous thrombotic events, male sex, the presence of an inferior vena cava filter, the presence of cancer, symptoms of post-thrombotic syndrome, and obesity.[3] These factors tend to be more important in the decision than the presence or absence of a detectable thrombophilia.[12][38]
Those with antiphospholipid syndrome may be offered long-term anticoagulation after a first unprovoked episode of thrombosis. The risk is determined by the subtype of antibody detected, by the
Women with a thrombophilia who are contemplating pregnancy or are pregnant usually require alternatives to warfarin during pregnancy, especially in the first 13 weeks, when it may produce abnormalities in the unborn child.
When women experience recurrent pregnancy loss secondary to thrombophilia, some studies have suggested that low molecular weight heparin reduces the risk of miscarriage. When the results of all studies are analysed together, no statistically significant benefit could be demonstrated.[40]
Prognosis
In people without a detectable thrombophilia, the cumulative risk of developing thrombosis by the age of 60 is about 12%. About 60% of people who are deficient in antithrombin will have experienced thrombosis at least once by age 60, as will about 50% of people with protein C deficiency and about a third of those with protein S deficiency. People with activated protein C resistance (usually resulting from factor V Leiden), in contrast, have a slightly raised absolute risk of thrombosis, with 15% having had at least one thrombotic event by the age of sixty.[13] In general, men are more likely than women to experience repeated episodes of venous thrombosis.[5]
People with factor V Leiden are at a relatively low risk of thrombosis, but may develop thrombosis in the presence of an additional risk factor, such as immobilization. Most people with the prothrombin mutation (G20210A) never develop thrombosis.[13]
Epidemiology
The major ("type 1") thrombophilias are rare. Antithrombin deficiency is present in 0.2% of the general population and 0.5–7.5% of people with venous thrombosis. Protein C deficiency, too, is present in 0.2% of the population, and can be found in 2.5–6% of people with thrombosis. The exact prevalence of protein S deficiency in the population is unknown; it is found 1.3–5% of people with thrombosis.[13]
The minor ("type 2") thrombophilias are much more common. Factor V Leiden is present in 5% of the population of Northern European descent, but much rarer in those of Asian or African extraction. In people with thrombosis, 10% have factor V Leiden. In those who are referred for thrombophilia testing, 30–50% have the defect. The prothrombin mutation occurs at rates of 1–4% in the general population, 5–10% of people with thrombosis, and 15% of people referred for thrombophilia testing. Like factor V Leiden, this abnormality is uncommon in Africans and Asians.[13]
The exact prevalence of antiphospholipid syndrome is not well known, as different studies employ different definitions of the condition. Antiphospholipid antibodies are detected in 24% of those referred to thrombophilia testing.[17]
History

German physician
Antiphospholipid syndrome was described in full in the 1980s, after various previous reports of specific antibodies in people with systemic lupus erythematosus and thrombosis.[17][44] The syndrome is often attributed to the British rheumatologist Graham R.V. Hughes, and is often referred to as Hughes syndrome for that reason.[45]
The more common genetic thrombophilias were described in the 1990s. Many studies had previously indicated that many people with thrombosis showed resistance activated protein C. In 1994 a group in Leiden, The Netherlands, identified the most common underlying defect—a mutation in factor V that made it resistant to the action of activated protein C. The defect was called factor V Leiden, as genetic abnormalities are typically named after the place where they are discovered.[46] Two years later, the same group described a common mutation in the prothrombin gene that caused elevation of prothrombin levels and a mild increase in thrombosis risk.[4][5][47]
It is suspected that other genetic abnormalities underlying familial thrombosis will in future be discovered through studies of the entire genetic code, looking for small alternations in genes.[4][5]
References
- ^ ISBN 978-1-4160-2973-1.
- ^ PMID 18024620.
- ^ S2CID 31610364.
- ^ PMID 18574041.
- ^ PMID 19630821.
- PMID 17060659.
- PMID 20592294.
- ^ PMID 18710381.
- ^ PMID 17433903.
- PMID 31549567.
- ^ S2CID 42968924.
- ^ PMID 20128794.
- ^ S2CID 43161448.
- S2CID 53089216.
- ^ PMID 16304352.
- PMID 19141167.
- ^ S2CID 25554663.
- ^ PMID 16643427.
- ^ S2CID 19715590.
- PMID 20817333.
- PMID 15925818.
- PMID 18497417.
- S2CID 11391129.
- PMID 26560059.
- S2CID 8633455.
- S2CID 201995636.
- PMID 15477430.
- ^ PMID 20163835.
- PMID 19700047.
- PMID 19228706.
- PMID 21542745.
- ^ PMID 18501222.
- PMID 16173967.
- ^ ABIM Foundation, American Society of Hematology, retrieved 10 December 2013, which cites
- Chong LY, Fenu E, Stansby G, Hodgkinson S, Guideline Development G (2012). "Management of venous thromboembolic diseases and the role of thrombophilia testing: Summary of NICE guidance". BMJ. 344: e3979. S2CID 42342532.
- Baglin T, Gray E, Greaves M, Hunt BJ, Keeling D, Machin S, MacKie I, Makris M, Nokes T, Perry D, Tait RC, Walker I, Watson H, British Committee for Standards in Haematology (2010). "Clinical guidelines for testing for heritable thrombophilia". British Journal of Haematology. 149 (2): 209–220. PMID 20128794.
- Chong LY, Fenu E, Stansby G, Hodgkinson S, Guideline Development G (2012). "Management of venous thromboembolic diseases and the role of thrombophilia testing: Summary of NICE guidance". BMJ. 344: e3979.
- PMID 24307720.
- PMID 19080721.
- S2CID 22896788.
- PMID 18574272. Archived from the originalon 2013-01-12.
- ^ PMID 18574280. Archived from the originalon 2013-01-12.
- PMID 26837697.
- S2CID 42594050.
- PMID 6895379.
- PMID 6239102.
- PMID 6414579.
- PMID 16880079.
- S2CID 4314040.
- PMID 8916933.
External links
- "Thrombophilia". Patient UK.
