Tissue transglutaminase
Ensembl | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 20: 38.13 – 38.17 Mb | Chr 2: 157.96 – 157.99 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
| Protein-glutamine gamma-glutamyltransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Tissue transglutaminase (abbreviated as tTG or TG2) is a 78-kDa, calcium-dependent
tTG is the
Structure
Gene
The human tTG gene is located on the 20th chromosome (20q11.2-q12).
Protein
TG2 is a multifunctional enzyme that belongs to
Mechanism
The catalytic mechanism for crosslinking in human tTG involves the
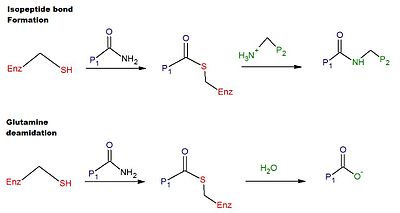
Regulation
The expression of tTG is regulated at the transcriptional level depending on complex signal cascades. Once synthesized, most of the protein is found in the cytoplasm, plasma membrane and ECM, but a small fraction is translocated to the nucleus, where it participates in the control of its own expression through the regulation of transcription factors.[22]
Crosslinking activity by tTG requires the binding of Ca2+ ions.

Recent studies have suggested that interferon-γ may serve as an activator of extracellular tTG in the small intestine; these studies have a direct implication to the pathogenesis of celiac disease.[12] Activation of tTG has been shown to be accompanied by large conformational changes, switching from a compact (inactive) to an extended (active) conformation. (see Figure 3)[23][25][26]

In the extracellular matrix, TG2 is "turned off", due primarily to the oxidizing activity of endoplasmic reticulum protein 57 (ERp57).[24] Thus, tTG is allosterically regulated by two separate proteins, Erp57 and TRX-1.[24] (See Figure 4).

Function
tTG is expressed ubiquitously and is present in various cellular compartments, such as the cytosol, the nucleus, and the plasma membrane.[14] It requires calcium as a cofactor for transamidation activity. Transcription is increased by retinoic acid. Among its many supposed functions, it appears to play a role in wound healing, apoptosis, and extracellular matrix development[11] as well as differentiation and cell adhesion.[14] It has been noted that tTG may have very different activity in different cell types. For example, in neurons, tTG supports the survival of cells subjected to injury whereas in astrocytes knocking out the gene expression for tTG is beneficial to cell survival.[27]
tTG is thought to be involved in the regulation of the cytoskeleton by crosslinking various cytoskeletal proteins including myosin, actin, and spectrin.[28] Evidence shows that intracellular tTG crosslinks itself to myosin. It is also believed that tTG may stabilize the structure of the dying cells during apoptosis by polymerizing the components of the cytoskeleton, therefore preventing the leakage of the cellular contents into the extracellular space.[7]
tTG also has GTPase activity:[5] In the presence of GTP, it suggested to function as a G protein participating in signaling processes.[29] Besides its transglutaminase activity, tTG is proposed to also act as kinase,[30] and protein disulfide isomerase,[31] and deamidase.[32] This latter activity is important in the deamidation of gliadin peptides, thus playing important role in the pathology of coeliac disease.
tTG also presents PDI (Protein Disulfide Isomerase) activity.[33][34] Based on its PDI activity, tTG plays an important role in the regulation of proteostasis, by catalyzing the trimerization of HSF1 (Heat Shock Factor 1) and thus the body's response to heat shock. In the absence of tTG, the response to heat shock is impaired since the necessary trimer is not formed.[34]
Clinical significance
tTG is the most comprehensively studied transglutaminase and has been associated with many diseases. However, none of these diseases are related to an enzyme deficiency. Indeed, thus far no disease has been attributed to the lack of tTG activity and this has been attested through the study of tTG knockout mice.[35]
Celiac Disease
tTG is best known for its link with
Cancer
Recent studies suggest that tTG also plays a role in
Other Diseases
tTG is believed to contribute to several neurodegenerative disorders including
tTG has also been linked to the pathogenesis of fibrosis in various organs including the lung and the kidney. Specifically, in kidney fibrosis, tTG contributes to the stabilization and accumulation of the ECM affecting TGF beta activity.[16]
Diagnostic
Therapeutic
It's still experimental to use tTG as a form of surgical glue. It is also being studied as an attenuator of
Interactions
TG2 participates in both enzymatic and non-enzymatic
| Mouse Mutant Alleles for Tgm2 | |
|---|---|
| Marker Symbol for Mouse Gene. This symbol is assigned to the genomic locus by the MGI | Tgm2 |
| Mutant Mouse Embryonic Stem Cell Clones. These are the known targeted mutations for this gene in a mouse. | Tgm2tm1a(KOMP)Wtsi |
| Example structure of targeted conditional mutant allele for this gene | |
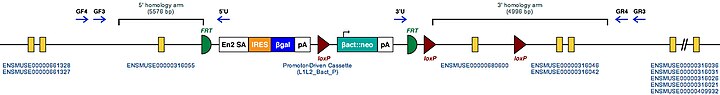
| |
| These Mutant ES Cells can be studied directly or used to generate mice with this gene knocked out. Study of these mice can shed light on the function of Tgm2: see Knockout mouse | |
Erp57
Endoplasmic reticulum protein 57 (Erp57), is a
Transglutaminase 2 (TG2) is a ubiquitously expressed (intracellular as well as extracellular) protein, with multiple modes of Post-translational regulation, including an allosteric disulfide bond between Cys-370-Cys-371 that renders the enzyme inactive in the extracellular matrix.[24]
Endoplasmic reticulum (ER)-resident protein 57 (ERp57), a protein in the ER that promotes folding of nascent proteins and is also present in the extracellular environment, has the cellular and biochemical characteristics for inactivating TG2. We found that ERp57 colocalizes with extracellular TG2 in cultured human umbilical vein endothelial cells (HUVECs). ERp57 oxidized TG2 with a rate constant that was 400-2000-fold higher than those of the aforementioned small molecule oxidants. Moreover, its specificity for TG2 was also markedly higher than those of other secreted redox proteins, including protein disulfide isomerase (PDI), ERp72, TRX, and quiescin sulfhydryl oxidase 1 (QSOX1).
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000198959 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000037820 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ S2CID 19217277.
- ^ PMID 22437759.
- ^ PMID 16368554.
- S2CID 11242870.
- PMID 22442151.
- PMID 10684262.
- ^ PMID 12366374.
- ^ PMID 22228808.
- ^ PMID 22326684.
- ^ S2CID 21883387.
- ^ "Entrez Gene: TGM2 transglutaminase 2".
- ^ )
- PMID 18092889.
- PMID 11867708.
- PMID 20450932.
- ^ PMID 20547769.
- PMID 26160175.
- PMID 29764956.
- ^ PMID 21908620.
- ^ PMID 29305423.
- PMID 18092889.
- PMID 21304968.
- PMID 29570839.
- PMID 22364871.
- PMID 12368090.
- PMID 15069073.
- PMID 12737632.
- PMID 17100777.
- ^ PMID 30691081.
- ^ PMID 29752334.
- ^ S2CID 58551851.
- S2CID 20033968.
- PMID 29550590.
- S2CID 59341070.
- S2CID 586174.
- PMID 21541002.
- S2CID 16143202.
- PMID 22698685.
- S2CID 11018740.
- PMID 29795262.
- ^ PMID 29187283.
- S2CID 14849506.
External links
- Endomysial antibodies Archived 2021-05-12 at the Wayback Machine
- A collection of substrates and interaction partners of TG2 is accessible in the TRANSDAB, an interactive transglutaminase substrate database.

