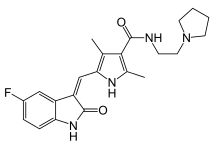
Toceranib
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Palladia |
| AHFS/Drugs.com | Veterinary Use |
| License data | |
Antineoplastic agent | |
| ATCvet code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 77% |
| Protein binding | 91%-93% |
| Elimination half-life | 16 h |
| Identifiers | |
| |
JSmol) | |
| |
| |
Toceranib is a
kit tyrosine kinase, though it may also have an anti-angiogenic
effect.
References
- PMID 19470739.
- ^ CBS News FDA Approves First-Ever Dog Cancer Drug
- ^ "FDA: First Drug to Treat Cancer in Dogs Approved". U.S. Food and Drug Administration (FDA) (Press release). 3 June 2009. Archived from the original on 22 July 2010. Retrieved 2 October 2021.
- ^ "Palladia New Animal Drug Application" (PDF). U.S. Food and Drug Administration (FDA). 22 May 2009. Archived from the original (PDF) on 16 November 2010. Retrieved 2 October 2021.
- ^ "In Trials for New Cancer Drugs, Family Pets Are Benefiting, Too". The New York Times. 24 November 2006. Retrieved 2 October 2021.
External links
- "Toceranib". Drug Information Portal. U.S. National Library of Medicine.
