Tocopherol
Tocopherols (/toʊˈkɒfəˌrɒl/;[1] TCP) are a class of organic compounds comprising various methylated phenols, many of which have vitamin E activity. Because the vitamin activity was first identified in 1936 from a dietary fertility factor in rats, it was named tocopherol, from Greek τόκος tókos 'birth' and φέρειν phérein 'to bear or carry', that is 'to carry a pregnancy', with the ending -ol signifying its status as a chemical alcohol.
Tocotrienols, which are related compounds, also have vitamin E activity. All of these various derivatives with vitamin activity may correctly be referred to as "vitamin E". Tocopherols and tocotrienols are fat-soluble antioxidants but also seem to have many other functions in the body.
Forms
| Form | Structure |
|---|---|
| α-Tocopherol | 
|
| β-Tocopherol | 
|
| γ-Tocopherol | 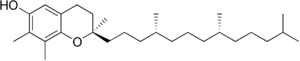
|
| δ-Tocopherol | 
|
The
Each form has a different biological activity.[4][5] In general, the unnatural l-isomers of tocotrienols lack almost all vitamin activity, and half of the possible 8 isomers of the tocopherols (those with 2S chirality at the ring-tail junction) also lack vitamin activity. Of the stereoisomers that retain activity, increasing methylation, especially full methylation to the alpha-form, increases vitamin activity. In tocopherols, this is due to the preference of the tocopherol binding protein for the α-tocopherol form of the vitamin.
As a food additive, tocopherol is labeled with these E numbers: E306 (tocopherol), E307 (α-tocopherol), E308 (γ-tocopherol), and E309 (δ-tocopherol). All of these are approved in the US,[6] EU,[7] and Australia and New Zealand[8] for use as antioxidants.
α-Tocopherol
α-Tocopherol is the form of vitamin E that is preferentially absorbed and accumulated in humans.[9] The measurement of "vitamin E" activity in international units (IU) was based on fertility enhancement by the prevention of miscarriages in pregnant rats relative to α-tocopherol.
Although the mono-methylated form ddd-γ-tocopherol is the most prevalent form of vitamin E in oils, there is evidence that rats can methylate this form to the preferred α-tocopherol, since several generations of rats retained α-tocopherol tissue levels, even when those generations were fed only γ-tocopherol through their lives.
There are three stereocenters in α-tocopherol, so this is a chiral molecule.[10] The eight stereoisomers of α-tocopherol differ in the arrangement of groups around these stereocenters. In the image of RRR-α-tocopherol below, all three stereocenters are in the R form. However, if the middle of the three stereocenters were changed (so the hydrogen was now pointing down and the methyl group pointing up), this would become the structure of RSR-α-tocopherol. These stereoisomers also may be named in an alternative older nomenclature, where the stereocenters are either in the d or l form.[11]

1 IU of tocopherol is defined as ⅔ milligrams of RRR-α-tocopherol (formerly named d-α-tocopherol or sometimes ddd-α-tocopherol). 1 IU is also defined as 1 milligram of an equal mix of the eight stereoisomers, which is a
Tocotrienols
Tocotrienols, although less commonly known, also belong to the vitamin E family. Tocotrienols have four natural 2' d-isomers (they have a stereoisomeric carbon only at the 2' ring-tail position). The four tocotrienols (in order of decreasing methylation: d-α-, d-β-, d-γ-, and d-δ-tocotrienol) have structures corresponding to the four tocopherols, except with an unsaturated bond in each of the three isoprene units that form the hydrocarbon tail, whereas tocopherols have a saturated phytyl tail (the phytyl tail of tocopherols gives the possibility for 2 more stereoisomeric sites in these molecules that tocotrienols do not have). Tocotrienol has been subject to fewer clinical studies and seen less research as compared to tocopherol. However, there is growing interest in the health effects of these compounds.[13]
Function and dietary recommendations

Mechanism of action
Tocopherols are radical scavengers, delivering an H atom to quench free radicals. At 323 kJ/mol, the O-H bond in tocopherols is approximately 10% weaker than in most other
Dietary considerations
The U.S. Recommended Dietary Allowance (RDA) for adults is 15 mg/day.[16] The RDA is based on the α-tocopherol form because it is the most active form as originally tested. Vitamin E supplements are absorbed best when taken with meals.[17] The U.S. Institute of Medicine has set an upper tolerable intake level (UL) for vitamin E at 1,000 mg (1,500 IU) per day.[18] The European Food Safety Authority sets UL at 300 mg α-tocopherol equivalents /day.[19]
α-Tocopherol equivalents
For dietary purposes, vitamin E activity of vitamin E
Sources
The U.S. Department of Agriculture (USDA), Agricultural Research Services, maintains a food composition database. The last major revision was Release 28, September 2015.[12] In general, food sources with the highest concentrations of vitamin E are vegetable oils, followed by nuts and seeds. Adjusting for typical portion sizes, however, for many people in the United States the most important sources of vitamin E include fortified breakfast cereals.[12]
Deficiency
Vitamin E deficiency is rare, and in almost all instances caused by an underlying disease rather than a diet low in vitamin E.[18] Vitamin E deficiency causes neurological problems due to poor nerve conduction. These include neuromuscular problems such as spinocerebellar ataxia and myopathies.[11] Deficiency also may cause anemia, due to oxidative damage to red blood cells.
Supplements
Commercial vitamin E supplements may be classified into several distinct categories:
- Fully synthetic vitamin E, "dl-α-tocopherol", the most inexpensive, most commonly sold supplement form usually as the acetate ester
- Semi-synthetic "natural source" vitamin E esters, the "natural source" forms used in tablets and multiple vitamins; these are highly fractionated d-α-tocopherol or its esters, often made by synthetic methylation of gamma and beta d,d,d tocopherol vitamers extracted from plant oils.
- Less fractionated "natural mixed tocopherols" and high d-γ-tocopherol fraction supplements
Synthetic all-racemic
Synthetic vitamin E derived from petroleum products is manufactured as all-
The 8-isomer all-rac vitamin E is always marked on labels simply as dl-tocopherol or dl-tocopheryl acetate, even though it is (if fully written out) dl,dl,dl-tocopherol. The present largest manufacturers of this type are DSM and BASF.
Natural α-tocopherol is the RRR-α (or ddd-α) form. The synthetic dl,dl,dl-α ("dl-α") form is not so active as the natural ddd-α ("d-α") tocopherol form. This is mainly due to reduced vitamin activity of the four possible stereoisomers that are represented by the l or S enantiomer at the first stereocenter (an S or l configuration between the chromanol ring and the tail, i.e., the SRR, SRS, SSR, and SSS stereoisomers).[10] The three unnatural "2R" stereoisomers with natural R configuration at this 2' stereocenter, but S at one of the other centers in the tail (i.e., RSR, RRS, RSS), appear to retain substantial RRR vitamin activity, because they are recognized by the alpha-tocopherol transfer protein, and thus maintained in the plasma, where the other four stereoisomers (SRR, SRS, SSR, and SSS) are not. Thus, the synthetic all-rac-α-tocopherol, in theory, would have approximately half the vitamin activity of RRR-α-tocopherol in humans. Experimentally, the ratio of activities of the 8 stereoisomer racemic mixture to the natural vitamin, is 1 to 1.36 in the rat pregnancy model (suggesting a measured activity ratio of 1/1.36 = 74% of natural, for the 8-isomer racemic mix).[23]
Although it is clear that mixtures of stereoisomers are not so active as the natural RRR-α-tocopherol form, in the ratios discussed above, specific information on any side effects of the seven synthetic vitamin E stereoisomers is not readily available.
Esters

Manufacturers also commonly convert the phenol form of the vitamins (with a free
Mixed tocopherols
"Mixed tocopherols" in the USA contain at least 20% w/w other natural R, R,R- tocopherols, i.e. R, R,R-α-tocopherol content plus at least 25% R, R,R-β-, R, R,R-γ-, R, R,R-δ-tocopherols.[citation needed]
Some brands may contain 20.0% w/w or more of the other tocopherols and measurable tocotrienols. Some mixed tocopherols with higher γ-tocopherol content are marketed as "High Gamma-Tocopherol". The label should report each component in milligrams, except R, R,R-α-tocopherol may still be reported in IU. Mixed tocopherols also may be found in other nutritional supplements.[citation needed]
Uses
Observational studies that measure dietary intake and/or serum concentration, and experimental studies that ideally are
Supplement popularity over time
In the US, the popularity for vitamin E as a dietary supplement may have peaked around 2000. The Nurses' Health Study (NHS) and the Health Professionals Follow-up Study (HPFS) tracked dietary supplement use by people over the age of 40 during years 1986–2006. For women, user prevalence was 16.1% in 1986, 46.2% in 1998, 44.3% in 2002, but had decreased to 19.8% in 2006. Similarly, for men, prevalence for same years was 18.9%, 52.0%, 49.4%, and 24.5%. The authors theorized that declining use in these health science aware populations may have due to publications of studies that showed either no benefits or negative consequences from vitamin E supplements.[29] There is other evidence for declining use of vitamin E. Within the U.S. military services, vitamin prescriptions written for active, reserve and retired military, and their dependents, were tracked over years 2007–2011. Vitamin E prescriptions decreased by 53% while vitamin C remained constant and vitamin D increased by 454%.[30] A report on vitamin E sales volume in the USA documented a 50% decrease between 2000 and 2006,[31] with a significant cause attributed to a well-publicized meta-analysis that had concluded that high-dosage vitamin E increased all-cause mortality.[32]
A Cochrane review published in 2017 (updated in 2023) on antioxidant vitamin and mineral supplements for slowing the progression of
Complementary and alternative medicine
Proponents of megavitamin therapy and orthomolecular medicine advocate natural tocopherols.[3] Meanwhile, clinical trials have largely concentrated on use of either a synthetic, all-racemic d-α-tocopheryl acetate or synthetic dl-α-tocopheryl acetate.[citation needed]
Antioxidant theory
Tocopherol is described as functioning as an antioxidant. A dose-ranging trial was conducted in people with chronic oxidative stress attributed to elevated serum cholesterol. Plasma F2-isoprostane concentration was selected as a biomarker of free radical-mediated lipid peroxidation. Only the two highest doses - 1600 and 3200 IU/day - significantly lowered F2-isoprostane.[36]
Alzheimer's disease
Cancer
From reviews of observational studies, diets higher in vitamin E content were associated with a lower relative risk of
The U.S. Food and Drug Administration initiated a process of reviewing and approving food and dietary supplement health claims in 1993. A Qualified Health Claim issued in 2012 allows product label claims that vitamin E may reduce risk of renal, bladder, and colorectal cancers, with a stipulation that the label must include a mandatory qualifier sentence: “FDA has concluded that there is very little scientific evidence for this claim.”[52] The European Food Safety Authority (EFSA) reviews proposed health claims for the European Union countries. As of March 2018, EFSA has not evaluated any vitamin E and cancer prevention claims.
Cataracts
A meta-analysis from 2015 reported that for studies that reported serum tocopherol, higher serum concentration was associated with a 23% reduction in relative risk of age-related cataracts (ARC), with the effect due to differences in nuclear cataract rather than cortical or posterior subcapsular cataract - the three major classifications of age-related cataracts.[53] However, this article and a second meta-analysis reporting on clinical trials of α-tocopherol supplementation reported no statistically significant change to risk of ARC when compared to placebo.[53][54]
Cardiovascular diseases
Research on the effects of vitamin E on
In 2001 the U.S. Food and Drug Administration rejected proposed health claims for vitamin E and cardiovascular health.[60] The U.S. National Institutes of Health also reviewed the literature and concluded there was not sufficient evidence to support the idea that routine use of vitamin E supplements prevents cardiovascular disease or reduces its morbidity and mortality.[18] In 2010 the European Food Safety Authority reviewed and rejected claims that a cause and effect relationship has been established between the dietary intake of vitamin E and maintenance of normal cardiac function or of normal blood circulation.[61]
Pregnancy
Antioxidant vitamins as dietary supplements have been proposed as having benefits if consumed during pregnancy. For the combination of vitamin E with vitamin C supplemented to pregnant women, a Cochrane review of 21 clinical trials concluded that the data do not support vitamin E supplementation - majority of trials α-tocopherol at 400 IU/day plus vitamin C at 1000 mg/day - as being efficacious for reducing risk of
Topical
Although there is widespread use of vitamin E as a topical medication, with claims for improved wound healing and reduced scar tissue, reviews have repeatedly concluded that there is insufficient evidence to support these claims.[63][64]
Side effects
The U.S. Food and Nutrition Board set a
Drug interactions
The amounts of α-tocopherol, other tocopherols and tocotrienols that are components of dietary vitamin E, when consumed from foods, do not appear to cause any interactions with drugs. Consumption of α-tocopherol as a dietary supplement in amounts in excess of 300 mg/day may lead to interactions with
Synthesis
Naturally sourced d-α-tocopherol can be extracted and purified from seed oils, or γ-tocopherol can be extracted, purified, and methylated to create d-alpha-tocopherol. In contrast to α-tocopherol extracted from plants, which also is called d-α-tocopherol, industrial synthesis creates dl-α-tocopherol. "It is synthesized from a mixture of toluene and 2,3,5-trimethyl-hydroquinone that reacts with isophytol to all-rac-α-tocopherol, using iron in the presence of hydrogen chloride gas as a catalyst. The reaction mixture obtained is filtered and extracted with aqueous caustic soda. Toluene is removed by evaporation and the residue (all rac-α-tocopherol) is purified by vacuum distillation." Specification for the ingredient is >97% pure.[69] This synthetic dl-α-tocopherol has approximately 50% of the potency of d-α-tocopherol. Manufacturers of dietary supplements and fortified foods for humans or domesticated animals convert the phenol form of the vitamin to an ester using either acetic acid or succinic acid because the esters are more chemically stable, providing for a longer shelf-life. The ester forms are de-esterified in the gut and absorbed as free α-tocopherol.
History
During feeding experiments with rats Herbert McLean Evans concluded in 1922 that besides vitamins B and C, an unknown vitamin existed.[70] Although every other nutrition was present, the rats were not fertile. This condition could be changed by additional feeding with wheat germ. It took several years until 1936 when the substance was isolated from wheat germ and the formula C29H50O2 was determined. Evans also found that the compound reacted like an alcohol and concluded that one of the oxygen atoms was part of an OH (hydroxyl) group. As noted in the introduction, the vitamin was given its name by Evans from Greek words meaning "to bear young" with the addition of the -ol as an alcohol.[71] The structure was determined shortly thereafter in 1938.[72]
See also
References
- ^ "Tocopherol". Dictionary.com Unabridged (Online). n.d. Retrieved 28 February 2018.
- ^ S2CID 24827255.
In North America, the intake of γ-tocopherol has been estimated to exceed that of α-tocopherol by a factor of 2–4 ... due to the fact that soybean oil is the predominant vegetable oil in the American diet (76.4%) followed by corn oil and canola oil (both 7%) ... The supply of dietary fats ... is much more diverse in Europe ... The oils mainly consumed in Europe, i.e. sunflower, olive and canola oil, provide less γ-tocopherol but more α-tocopherol ... [T]he ratio of α-:γ-tocopherol is at least 1:2. Therefore, the average γ-tocopherol intake may be estimated as 4–6 mg/day, which is about 25–35% of the USA intake. In accordance with the lower estimated European intake of γ-tocopherol, the serum levels of γ-tocopherol in European populations are 4–20 times lower than that of α-tocopherol
- ^ PMID 11722951.
- ^ a b Food and Agriculture Organization, World Health Organization (2001). "9. Vitamin E". Joint FAO/WHO Expert Consultation on Human Vitamin and Mineral Requirements (Report). Bangkok, Thailand: FAO Rome.
- .
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". Food and Drug Administration. Archived from the original on November 8, 2011. Retrieved 2011-10-27.
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Archived from the original on 2010-10-07. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code "Standard 1.2.4 - Labelling of ingredients". 8 September 2011.
- PMID 17320165.
- ^ )
- ^ S2CID 7031925.
- ^ a b c Nutrient Data Laboratory (September 2015). Composition of Foods Raw, Processed, Prepared: USDA National Nutrient Database for Standard Reference, Release 28 (PDF) (Report) (Slightly revised ed.). Beltsville, MA: Beltsville Human Nutrition Research Center, Agricultural Research Service, United States Department of Agriculture. pp. 20–21. SR28.
- PMID 16458936.
- ISBN 0-8493-0487-3.
- PMID 21664268.
- ^ from the original on 2018-02-26.
- PMID 11597949.
- ^ National Institutes of Health (26 March 2021). "Vitamin E: Facts Sheet for Health Professionals". Dietary Supplement Fact Sheets. Office of Dietary Supplements. Retrieved 19 May 2021.
- ^ a b Tolerable Upper Intake Levels For Vitamins And Minerals (PDF), European Food Safety Authority, 2006, archived (PDF) from the original on 2016-03-16
- ^ "Vitamins". Archived from the original on 2013-03-20. Retrieved 2013-03-26. University of Minnesota Nutrition Coordinating Center on Vitamins
- PMID 11160563.
- PMID 8857515.
- PMID 11890405. Archived from the original(PDF) on 2008-12-17. Retrieved 2008-03-12.
- PMID 7288289.
- PMID 17254374.
- PMID 29262004.
- PMID 38174786.
- PMID 27414419.
- PMID 24119503.
- PMID 26126244.
- PMID 18618194.
- S2CID 35030072.
- ^ PMID 37702300.
- PMID 28756617.
- PMID 23582353.
- PMID 17936185.
- S2CID 8570146.
- S2CID 20893019.
- PMID 16291923.
- PMID 22543848.
- S2CID 44859128.
- S2CID 52848530.
- ^ PMID 26547129.
- ^ PMID 25550926.
- ^ PMID 28244705.
- PMID 22498220.
- PMID 8127329.
- PMID 20363687.
- PMID 9521168.
- PMID 21990298.
- PMID 15998891.
- ^ Alliance for Natural Health v. Sebelius, Case No. 09-1546 (D.D.C.) U.S. Food & Drug Administration May 17, 2012
- ^ PMID 25591715.
- PMID 22696344.
- ^ PMID 19774218.
- ^ PMID 8479463.
- PMID 25779938.
- PMID 18997197.
- S2CID 23237227.
- ^ Letter Regarding Dietary Supplement Health Claim for Vitamin E and Heart Disease (Docket No 99P-4375) U.S. Food and Drug Administration.
- ^ Scientific Opinion on the substantiation of health claims related to vitamin E and protection of DNA, proteins and lipids from oxidative damage (ID 160, 162, 1947),... maintenance of normal cardiac function (ID 166),... maintenance of normal blood circulation (ID 216)... pursuant to Article 13(1) of Regulation (EC) No 1924/2006 European Food Safety Authority EFSA Journal 2010;8(10):1816.
- ^ PMID 26343254.
- PMID 26044054.
- PMID 26977069.
- PMID 24040282.
- S2CID 23820017.
- S2CID 38212099.
- ^ PMID 25225959.
- ^ Scientific Opinion on the safety and efficacy of synthetic α-tocopherol for all animal species (2012) European Food Safety Authority EFSA Journal 2012;10(7):2784
- PMID 17838496.
- from the original on 29 September 2007.
- .
External links
- US Office of Dietary Supplements article on Vitamin E
- Vitamin E risk assessment Archived 2006-01-02 at the Wayback Machine, Expert Group on Vitamins and Minerals, UK Food Standards Agency, 2003
