Toll-like receptor
| Toll-like receptor | |
|---|---|
 The curved leucine-rich repeat region of toll-like receptors, represented here by TLR3 | |
| Identifiers | |
| Symbol | Toll-like receptor |
| Membranome | 7 |
| PIRSF037595 | |
Toll-like receptors (TLRs) are a class of
TLRs received their name from their similarity to the protein coded by the toll gene.[4]
Function
The ability of the immune system to recognize
The binding of
Upon activation, TLRs recruit
Toll-like receptors have also been shown to be an important link between innate and adaptive immunity through their presence in dendritic cells.[8] Flagellin, a TLR5 ligand, induces cytokine secretion on interacting with TLR5 on human T cells.[8]
Superfamily

TLRs are a type of
Three subgroups of TIR domains exist. Proteins with subgroup 1 TIR domains are receptors for
Extended family
This section is missing information about choanoflagellate TLR (pmid29848444) . (December 2021) |
TLRs are present in
In recent years TLRs were identified also in the mammalian nervous system. Members of the TLR family were detected on glia, neurons and on neural progenitor cells in which they regulate cell-fate decision.[10]
It has been estimated that most mammalian species have between ten and fifteen types of toll-like receptors. Thirteen TLRs (named simply TLR1 to TLR13) have been identified in humans and mice together, and equivalent forms of many of these have been found in other mammalian species.
Vertebrate TLRs are divided by similarity into the families of TLR 1/2/6/10/14/15, TLR 3, TLR 4, TLR 5, TLR 7/8/9, and TLR 11/12/13/16/21/22/23.[14]
TLRs in Drosophila immunity
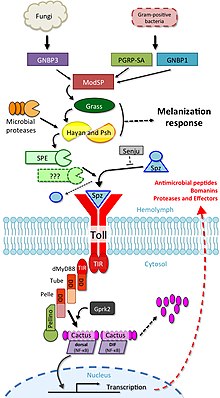
The involvement of toll signalling in immunity was first demonstrated in the fruit fly,
The toll pathway is activated by different stimuli, such as
Drosophila have a total of 9 toll family and 6 spz family genes that interact with each other to differing degrees.[22]
TLR2
TLR3
TLR11
As noted above, human cells do not express
As mouse TLR11 is able to recognize Salmonella effectively, normal mice do not get infected by oral
Summary of known mammalian TLRs
Toll-like receptors bind and become activated by different ligands, which, in turn, are located on different types of organisms or structures. They also have different adapters to respond to activation and are located sometimes at the cell surface and sometimes to internal
| Receptor | Ligand(s)[26] | Ligand location[26] | Adapter(s) | Location | Cell types[26] |
|---|---|---|---|---|---|
TLR 1 |
multiple triacyl lipopeptides | Bacterial lipoprotein | MyD88 /MAL |
cell surface |
|
TLR 2 |
multiple glycolipids | Bacterial peptidoglycans | MyD88/MAL | cell surface | |
| multiple lipopeptides and proteolipids | Bacterial peptidoglycans | ||||
| lipoteichoic acid | Gram-positive bacteria | ||||
HSP70 |
Host cells
| ||||
| zymosan (Beta-glucan) | Fungi | ||||
| Numerous others | |||||
TLR 3
|
poly I:C
|
viruses | TRIF
|
cell compartment |
|
TLR 4
|
lipopolysaccharide | Gram-negative bacteria | MyD88/MAL/ TRIF /TRAM |
cell surface |
|
| several heat shock proteins | Bacteria and host cells | ||||
| fibrinogen | host cells | ||||
| heparan sulfate fragments | host cells | ||||
| hyaluronic acid fragments | host cells | ||||
| nickel[31] | |||||
| Various opioid drugs | |||||
TLR 5 |
Bacterial flagellin |
Bacteria | MyD88 | cell surface |
|
| Profilin[32] | Toxoplasma gondii | ||||
TLR 6 |
multiple diacyl lipopeptides | Mycoplasma | MyD88/MAL | cell surface |
|
TLR 7 |
imidazoquinoline | small synthetic compounds | MyD88 | cell compartment |
|
| loxoribine (a guanosine analogue) | |||||
| bropirimine | |||||
| resiquimod | |||||
| single-stranded RNA | RNA viruses | ||||
TLR 8 |
small synthetic compounds; single-stranded Viral RNA, phagocytized bacterial RNA(24) | MyD88 | cell compartment |
| |
TLR 9 |
unmethylated CpG Oligodeoxynucleotide DNA |
Bacteria, DNA viruses | MyD88 | cell compartment |
|
TLR 10 |
triacylated lipopeptides[34] | unknown | cell surface | ||
TLR 11 |
Profilin | Toxoplasma gondii[38] | MyD88 | cell compartment[39] |
|
| Flagellin | Bacteria (E. coli, Salmonella)[24] | ||||
| TLR 12 | Profilin | Toxoplasma gondii[40] | MyD88 | cell compartment |
|
| TLR 13[42][43] | bacterial ribosomal RNA sequence "CGGAAAGACC" (but not the methylated version)[44] | Virus, bacteria | MyD88, TAK-1 | cell compartment |
|
Ligands

Because of the specificity of toll-like receptors (and other innate immune receptors) they cannot easily be changed in the course of evolution, these receptors recognize molecules that are constantly associated with threats (i.e., pathogen or cell stress) and are highly specific to these threats (i.e., cannot be mistaken for self molecules that are normally expressed under physiological conditions). Pathogen-associated molecules that meet this requirement are thought to be critical to the pathogen's function and difficult to change through mutation; they are said to be evolutionarily conserved. Somewhat conserved features in pathogens include
Endogenous ligands
The stereotypic inflammatory response provoked by toll-like receptor activation has prompted speculation that endogenous activators of toll-like receptors might participate in autoimmune diseases. TLRs have been suspected of binding to host molecules including
Signaling

TLRs are believed to function as
A set of endosomal TLRs comprising TLR3, TLR7, TLR8 and TLR9 recognize
The adapter proteins and kinases that mediate TLR signaling have also been targeted. In addition, random germline mutagenesis with
TLR signaling is divided into two distinct signaling pathways, the MyD88-dependent and TRIF-dependent pathway.
MyD88-dependent pathway
The MyD88-dependent response occurs on dimerization of TLRs, and is used by every TLR except TLR3. Its primary effect is activation of NFκB and
TRIF-dependent pathway
Both TLR3 and TLR4 use the TRIF-dependent pathway, which is triggered by
TLR signaling ultimately leads to the induction or suppression of genes that orchestrate the inflammatory response. In all, thousands of genes are activated by TLR signaling, and collectively, the TLRs constitute one of the most
TLR4 is the only TLR that uses all four adaptors. Complex consisting of TLR4, MD2 and LPS recruits TIR domain-containing adaptors TIRAP and MyD88 and thus initiates activation of NFκB (early phase) and MAPK. TLR4-MD2-LPS complex then undergoes endocytosis and in endosome it forms a signalling complex with TRAM and TRIF adaptors. This TRIF-dependent pathway again leads to IRF3 activation and production of type I interferons, but it also activates late-phase NFκB activation. Both late and early phase activation of NFκB is required for production of inflammatory cytokines.[49]
Medical relevance
Imiquimod (cardinally used in dermatology) is a TLR7 agonist, and its successor resiquimod, is a TLR7 and TLR8 agonist.[53] Recently, resiquimod has been explored as an agent for cancer immunotherapy,[54] acting through stimulation of tumor-associated macrophages.
Several TLR ligands are in clinical development or being tested in animal models as vaccine adjuvants,[55] with the first clinical use in humans in a recombinant herpes zoster vaccine in 2017, which contains a monophosphoryl lipid A component.
TLR7 messenger RNA expression levels in dairy animals in a natural outbreak of foot-and-mouth disease have been reported.[56]
The "unnatural" enantiomers of opioid drugs such as (+)-morphine and (+)-naloxone lack affinity for opioid receptors, still produce the same activity at TLR4 as their "normal" enantiomers.[64][65] So, "unnatural" entianomers of opioids such as (+)-naloxone, can be used to block the TLR4 activity of opioid analgesic drugs without having any affinity for μ-opioid receptor[66][65][67]
Discovery
When microbes were first recognized as the cause of infectious diseases, it was immediately clear that multicellular organisms must be capable of recognizing them when infected and, hence, capable of recognizing molecules unique to microbes. A large body of literature, spanning most of the last century, attests to the search for the key molecules and their receptors. More than 100 years ago,
The prototypic member of the family, the toll receptor (P08953; Tl) in the fruit fly
The first reported human toll-like receptor was described by Nomura and colleagues in 1994,
In 1997,

In turn, the other TLR genes were ablated in mice by gene targeting, largely in the laboratory of Shizuo Akira and colleagues. Each TLR is now believed to detect a discrete collection of molecules – some of microbial origin, and some products of cell damage – and to signal the presence of infections.[73]
Plant homologs of toll were discovered by Pamela Ronald in 1995 (rice XA21)[74] and Thomas Boller in 2000 (Arabidopsis FLS2).[75]
In 2011, Beutler and Hoffmann were awarded the Nobel Prize in Medicine or Physiology for their work.[76] Hoffmann and Akira received the Canada Gairdner International Award in 2011.[77]
Notes and references
- PMID 24032031.
- PMID 32655969.
- PMID 20860480.
- ^ PMID 15923538.
- PMID 17212934.
- PMID 15585605.
- ^ PMID 9237759.
- ^ PMID 23345392.
- ^ Sameer AS, Nissar S. Toll-Like Receptors (TLRs): Structure, Functions, Signaling, and Role of Their Polymorphisms in Colorectal Cancer Susceptibility. Biomed Res Int. 2021 Sep 12;2021:1157023. doi: 10.1155/2021/1157023. PMID 34552981; PMCID: PMC8452412.
- S2CID 12517461.
- PMID 11022119.
- PMID 11022120.
- PMID 14993594.
- ^ PMID 15976025.
- PMID 17201680.
- PMID 21209287.
- ^ PMID 31018123.
- PMID 27776480.
- ^ S2CID 10736743.
- PMID 29452635.
- S2CID 11104900.
- PMID 31088910.
- PMID 23101625.
- ^ PMID 26859749.
- PMID 23101627.
- ^ ISBN 978-0-7817-9543-2.
- ^ PMID 16237641.
- ^ PMID 12110131.
- S2CID 30443009.
- PMID 10623846.
- PMID 28965571.
- PMID 24861338.
- ^ Seizer L, Rahimi S, Santos-Sierra S, Drexel M (2022) Expression of toll like receptor 8 (TLR8) in specific groups of mouse hippocampal interneurons. PLoS ONE 17(5): e0267860. https://doi.org/10.1371/journal.pone.0267860
- PMID 20348427.
- PMID 11267672.
- PMID 11970999.)
{{cite journal}}: CS1 maint: location (link - ^ PMID 24198280.
- S2CID 34165967.
- PMID 21097503.
- PMID 23246311.
- PMID 19077284.
- PMID 21131352.
- S2CID 206540638.
- PMID 23802068.
- PMID 20424184.
- PMID 37487664.
- S2CID 15608748.
- S2CID 4405163.
- ^ S2CID 39414949.
- PMID 17475853.
- S2CID 13016860.
- S2CID 16163262.
- ISBN 3-540-00332-0.
- PMID 31015631.
- PMID 26344622.
- ^ Audarya, S.D.; Pattnaik, B.; Sanyal, A.; Mohapatra, J.K. (2017). "Toll like Receptor 7 Messenger Ribonucleic Acid Expression Levels in Dairy Animals in an Outbreak of Foot-and-mouth disease" (PDF). Buffalo Bulletin. 36 (3). Archived from the original (PDF) on 28 April 2021.
- S2CID 7286123.
- PMID 20615556.
- PMID 19607972.
- PMID 19833175.
- ^ Drahl C (22 August 2012). "Small Molecules Target Toll-Like Receptors". Chemical & Engineering News.
- S2CID 12295407.
- PMID 20974246.
- PMID 19762094.
- ^ PMID 18662331.
- PMID 18599265.
- PMID 20178837.
- S2CID 19439405.
- PMID 7584026.
- PMID 8838819.
- S2CID 1700458.
- PMID 9851930.
- S2CID 7419784.
- S2CID 10548988.
- PMID 10911994.
- ^ "The Nobel Prize in Physiology or Medicine 2011". Nobel Media AB. 3 October 2011.
- ^ Mitchell B (23 March 2011). "B.C. doctor wins prestigious medical prize". The Star.
See also
External links
- Toll-Like+Receptors at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Toll+protein,+Drosophila at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- TollML: Toll-like receptors and ligands database at University of Munich
- The Toll-Like Receptor Family of Innate Immune Receptors (pdf)
- Toll-Like receptor Pathway
- BioScience Animations
