Transfer hydrogenation
In
Organometallic catalysts
In the area of
where RR'C*H−OH is a chiral product. A typical catalyst is (cymene)R,R-HNCHPhCHPhNTs, where Ts refers to a tosyl group (SO2C6H4Me) and R,R refers to the absolute configuration of the two chiral carbon centers. This work was recognized with the 2001 Nobel Prize in Chemistry to Ryōji Noyori.[5]
Another family of hydrogen-transfer agents are those based on aluminium alkoxides, such as aluminium isopropoxide in the MPV reduction; however their activities are relatively low by comparison with the transition metal-based systems.

The catalytic asymmetric hydrogenation of
Even though the BINAP-Ru dihalide catalyst could reduce functionalized ketones, the hydrogenation of simple

Metal-free routes
Prior to the development of catalytic hydrogenation, many methods were developed for the hydrogenation of unsaturated substrates. Many of these methods are only of historical and pedagogical interest. One prominent transfer hydrogenation agent is diimide or (NH)2, also called diazene. This becomes oxidized to the very stable N2:
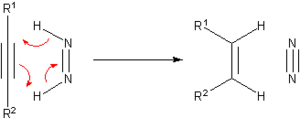
The diimide can be generated from hydrazine or certain other organic precursors.
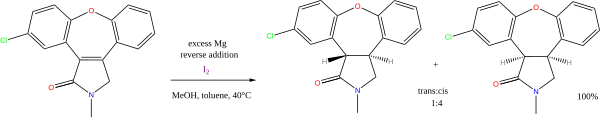
Two hydrocarbons that can serve as hydrogen donors are cyclohexene or cyclohexadiene. In this case, an alkane is formed, along with a benzene. The gain of aromatic stabilization energy when the benzene is formed is the driving force of the reaction. Pd can be used as a catalyst and a temperature of 100 °C is employed. More exotic transfer hydrogenations have been reported, including this intramolecular one:

Many reactions exist with alcohol or
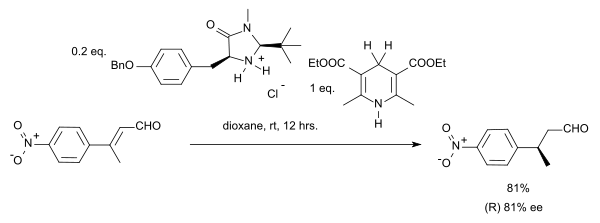
Organocatalytic transfer hydrogenation
In this particular reaction the substrate is an
In a case of
Extending the scope of this reaction towards
With another organocatalyst altogether, hydrogenation can also be accomplished for imines. One cascade reaction is catalyzed by a chiral phosphoric acid:[15]
The reaction proceeds via a chiral
See also
- Meerwein–Ponndorf–Verley reduction
- Oppenauer oxidation
- Dehydrogenation
- Hydrogenation
- Hydrogenolysis
- Borrowing hydrogen
References
- ISSN 0009-2665.
- ISBN 0-8247-1915-8.
- PMID 16187395.
- ^ T. Ikariya, K. Murata, R. Noyori "Bifunctional Transition Metal-Based Molecular Catalysts for Asymmetric Syntheses" Org. Biomol. Chem., 2006, volume 4, 393-406.
- PMID 11169691
- S2CID 106394152.
- .
- .
- PMID 15540245.
- PMID 15631434.
- S2CID 12456921.
- PMID 16639754.