Tree swallow
| Tree swallow | |
|---|---|

| |
| Adult in Queens, New York
| |
| Scientific classification | |
| Domain: | Eukaryota |
| Kingdom: | Animalia |
| Phylum: | Chordata |
| Class: | Aves |
| Order: | Passeriformes |
| Family: | Hirundinidae |
| Genus: | Tachycineta |
| Species: | T. bicolor
|
| Binomial name | |
| Tachycineta bicolor (Vieillot, 1808)
| |
| |
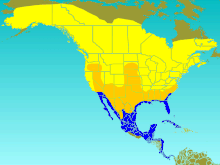
| Range of T. bicolor Breeding summer visitor Migration visitor Winter visitor
| |
| Synonyms | |
|
Hirundo bicolor Vieillot, 1808 | |
The tree swallow (Tachycineta bicolor) is a
The tree swallow nests either in isolated pairs or loose groups, in both natural and artificial cavities. Breeding can start as soon as early May, although this date is occurring earlier because of
An aerial
Taxonomy and etymology
The tree swallow was described as Hirundo bicolor by Louis Pierre Vieillot in his Histoire naturelle des oiseaux de l'Amérique Septentrionale, published in 1807.[2] It was then placed in its current genus Tachycineta when Jean Cabanis established it in 1850.[3] In 1878, Elliott Coues suggested that the tree swallow, at the very least, be put in its own subgenus, Iridoprocne, on the basis of its plumage, along with the white-winged swallow, Chilean swallow, white-rumped swallow, and mangrove swallow.[4] By 1882, he had upgraded this to a full genus.[5] Some authors continued to use this classification, with the addition of Tumbes swallow; however, genetic evidence supports the existence of a single genus, Tachycineta.[6] The tree swallow is also called the white-bellied swallow for its white underparts.[7]
The generally accepted genus name is from Ancient Greek takhykinetos, "moving quickly", and the specific bicolor is Latin and means "two-coloured".[8] The other genus name, Iridoprocne, comes from the Greek iris, meaning rainbow, and Procne, a figure who supposedly turned into a swallow.[5]
How exactly the tree swallow is related to other members of Tachycineta is unresolved. In studies based on mitochondrial DNA, it was placed basal (meaning it was the first offshoot in the species tree) within the North American-Caribbean clade consisting of the violet-green swallow, golden swallow, and Bahama swallow.[9][10] Although mitochondrial DNA is advocated as a better indicator of evolutionary changes because it evolves quickly, analyses based on it can suffer because it is only inherited from the mother, making it worse than nuclear DNA from multiple loci at representing the phylogeny of a whole group.[11] A study based on such nuclear DNA placed the tree swallow in the most basal position within Tachycineta as a whole (as a sister group to the rest of the genus).[12]
Description

The tree swallow has a length between about 12 and 14 cm (4.7 and 5.5 in) and a weight of approximately 17 to 25.5 g (0.60 to 0.90 oz). Wingspan ranges from 30 to 35 cm (12 to 14 in).[13] The male has mostly glossy blue-green upperparts, the wings and tail being blackish. The underparts[14] and the cheek patch are white,[7] although the underwing coverts are grey-brown.[14] The bill is black, the eyes dark brown, and the legs and feet pale brown.[7] The female is duller in colour than the male, and sometimes has a brown forehead.[14] The second-year[note 1] female also has brown upperparts, with a variable number of blue feathers; some third-year females also retain a portion of this subadult plumage.[15] According to a 1987 study, this likely allows a younger female to explore nest sites, as the resident male is usually less aggressive to a subadult female.[16] A 2013 study found that the resident female was less aggressive towards second-year female models when they were presented separately from older models. Why the female eventually replaces its subadult plumage is unknown; it may allow males to assess female quality, as pairs mate assortatively based on plumage brightness.[17] The juvenile tree swallow can be distinguished by its brown upperparts and grey-brown-washed breast.[14]
Voice

The tree swallow's song consists of three parts: the chirp, the whine, and the gurgle. These sections may be repeated or omitted, and all can stand alone. The first, as the chirp call (sometimes divided into the contact call and solicitation call), is made by the female during copulation[18] and in both sexes to stimulate the nestlings to beg[19] or (in some populations) when their mate leaves or enters the nest cavity. The whine, generally consisting of a downward shift in frequency followed by an upward shift, may be given alone as the anxiety call,[18] occasionally made in response to certain predators.[20] The gurgle, as when it appears at the end of the song, is usually uttered twice. It is likely involved in pair bonding. The chatter call is used to advertise nest sites (the reason it is also known as the "nest-site advertising call") and is also given to intruding conspecifics. A short high-pitched submission call is sometimes uttered after an aggressive encounter with another tree swallow. While being physically restrained or in pain, a distress call may be given. The male often utters a ticking (or rasping) aggression call during copulation, and both sexes use it at the end of mobbing dives. The alarm call is given in reaction to predators and other intruders,[18] and can serve to induce older nestlings to crouch and stop begging when a predator is near.[21]
Communication between parents and offspring can be disrupted by human-generated noise. A 2014 study, for example, found that broods for whom white noise was played were less likely to crouch or stop begging in response to alarm calls. Parents did not alter their calls to compensate, likely increasing predation risk.[22] Noise can also disrupt whether parents respond to begging, but this may be balanced out by the louder calls nestlings give when exposed to it. Increased begging effort, however, may be ineffective or costly for louder levels of noise.[23]
Distribution and habitat
The tree swallow breeds in North America. Its range extends to north-central Alaska and up to the
The breeding habitat of this bird is primarily in open and wooded areas, especially those near water.[7] It roosts every night during the non-breeding season, preferring to rest in cane or reed beds over water, but it is also found over land and on trees and wires. Roosting sites are generally 100 to 150 kilometres (62 to 93 mi) apart.[26]
Behaviour

Because of the large amount of research on the tree swallow and how it willingly breeds in nest boxes, biologist Jason Jones recommended that it be considered a model organism.[27] Although it is aggressive during the breeding season, this swallow is sociable outside of it, forming flocks sometimes numbering thousands of birds.[7]
Breeding

The tree swallow nests in structures with pre-existing holes, both natural and artificial. These were once found only in forested regions, but the building of nest boxes has allowed this bird to expand into open habitats.[7] This swallow usually nests in the area it bred the year before; only about 14% of females and 4% of males disperse to breed at a new site per year. Most do not go far, usually breeding at sites less than 10 kilometres (6.2 mi) away from their original grounds. Dispersal is influenced by breeding success; of the adult females that fail to fledge a chick, about 28% disperse, compared to 5% of successful breeders.[25] Natal dispersal (when a bird does not return to the site it was born at to breed) is common in the tree swallow and occurs more frequently than breeding dispersal.[28] It nests both in loose groups and isolated pairs. When nesting in loose groups, nests are usually spaced at least 10 to 15 metres (33 to 49 ft) apart,[14] and those that are closer in distance are usually further apart in terms of laying date.[29] In natural cavities, the tree swallow nests about 27 metres (89 ft) apart from its neighbor. The nest hole in these situations is, on average, 3.4 metres (11 ft) above ground level, although about 45% of them are less than 2 metres (6.6 ft) above the ground. Higher cavities are likely favoured because they reduce predation, while lower nest holes may be chosen to avoid competition. Entrance widths are often between 4 and 5 centimetres (1.6 and 2.0 in), whereas entrance heights are more variable: a 1989 study found openings ranging from 3.5 to 26 centimetres (1.4 to 10.2 in). Cavity volume is generally below 1,000 cm3 (61 cu in).[30] After finding a suitable place to nest, the male perches near it and calls frequently. A lack of sites can cause fights between birds, sometimes resulting in deaths. This swallow usually defends an area around the nest with a radius of about 4.6 metres (15 ft),[7] as well as extra nests inside of that territory,[14] by blocking the entrance to the nest and chasing intruders.[7] The nest cup itself is made from grass, moss, pine needles, and aquatic plants collected mostly by the female,[14] and is lined with feathers gathered primarily by the male in fights.[31] The feathers may function to insulate the nest, decreasing incubation time[32] and likely preventing hypothermia in chicks. In addition to faster growth for chicks,[31] eggs cool slower in nests with feathers than those without.[33] However, a study published in 2018 did not find a significant correlation between the number of feathers in nests that were artificially warmed versus those that were not. Additionally, it found that nests in St. Denis, Saskatchewan used significantly less feathers than those in Annapolis Valley, despite the former being further north. However, temperatures in Nova Scotia (where Annapolis Valley is) are generally lower than those in Saskatchewan, possibly explaining the unexpected result.[34]

During courtship, a male tree swallow attacks an unknown female. This can be stimulated through wing-fluttering flight by the female, which may be an invitation to court. The male may then take a vertical posture, with a raised and slightly spread tail and wings flicked and slightly drooped. This prompts the female to try to land on the male's back, but he flies to prevent this; this is repeated. After courting the female, the male flies to his chosen nest site, which the female inspects. During copulation, the male hovers over the female, and then mounts her, giving ticking calls. He then makes cloacal contact with the female while holding her neck feathers in his bill and standing on her slightly outstretched wings. Copulation occurs multiple times.[7]
Eggs are laid from early May to mid-June (although this is happening earlier due to

The tree swallow has high rates of extra-pair paternity, 38% to 69% of nestlings being a product of extra-pair paternity, and 50% to 87% of broods containing at least one nestling that was the result of an extra-pair copulation.[14] One factor that might contribute to this is that females have control over copulation, making paternity guards ineffective.[40] This may be mitigated by more frequent copulations just before egg laying, according to a 2009 study which found that within-pair copulation attempts peaked three to one days before the first egg was laid and that more successful attempts during this period increased the share of within-pair young males had. This latter finding contradicts those of a 1993 and a 1994 study.[41] Extra-pair paternity does not change the level of parental care the male contributes in the tree swallow.[42] A significant number of extra-pair fathers may be floaters (those present at breeding grounds that presumably do not breed). A 2001 study found that out of 35 extra-pair nestlings, 25 were sired by local residents, three by residents of nearby sites, and seven by male floaters. In the tree swallow, floating thus helps males in good condition produce more chicks, while allowing males in bad condition to be successful by investing in parental care.[43] There is also a significant population of female floaters; a 1985 study estimated that around 23% to 27% of females were floaters, of which about 47% to 79% were subadults.[44]
Why females engage in extra-pair copulation and how they choose extra-pair mates is controversial. One theory, called the genetic compatibility hypothesis, states that increased offspring fitness results from increased
Studies attempting to prove the adaptability of extra-pair paternity for females have been criticized for the lack of positive effect that increased offspring fitness would have when compared with the potential cost of decreased fitness for the female,[50] such as increased predation from searching for mates.[51] Thus, theories based on the non-adaptivity of extra-pair paternity for females have been postulated. These theories are based on genetic constraint, where an allele resulting in a maladaptive behaviour is maintained because it also contributes to a beneficial phenotype. The theory of intersexual antagonistic pleiotropy says that strong selection for extra-pair paternity in males (as seen in this bird[50]) overrides the weak selection against extra-pair paternity in females. The hypothesis of intrasexual antagonistic pleiotropy, meanwhile, argues that extra-pair paternity is present because the genes regulating it have pleiotropic effects on aspects of female fitness,[52] like within-pair copulation rate.[50]

The tree swallow lays a clutch of two to eight, although usually four to seven,[14] pure white, and translucent at laying, eggs that measure about 19 by 14 mm (0.75 by 0.55 in).[53] These eggs are incubated by the female,[14] usually after the second-to-last egg is laid,[54] for 11 to 20 days,[53] although most hatch after 14 to 15 days. About 88% of nests produce at least one nestling, but this can be lowered by poor weather and a younger breeding female.[14] The eggs generally hatch in the order they were laid. They also hatch slightly asynchronously, with an average of 28 hours between when the first and final nestling emerges. This can result in a weight hierarchy where earlier-hatched chicks weigh more (especially early in the nestling period) than those hatched later, allowing the female to prioritize which chick to give food to during food shortages. This likely has its greatest effect early in the nestling period, as by 12 days after hatching, there are generally no significant weight differences.[54] Infanticide of the chicks and eggs sometimes occurs when a male is replaced by another male. Infanticide usually does not occur when the clutch is not complete, as replacement males then have a chance to fertilize at least one egg. When the male arrives during incubation, it sometimes commits infanticide, but other times adopts the eggs, as there is a chance that some eggs were sired from the replacement male. If the replacement male arrives after the chicks hatch, infanticide is usually committed, though the female will sometimes prevent this.[55]
Nests produced by females of better condition often have sex ratios skewed towards high quality males. A 2000 study hypothesized this to be because males have more variable reproductive success, and therefore that a high quality male produces more offspring than a female of similar quality.[56]
The growth and survival of nestling tree swallows is influenced by their environment. In both younger and older nestlings (those between two and four days old and between nine and eleven days, respectively) growth is positively influenced by a higher maximum temperature, particularly in the former. A later hatching date negatively impacts growth, especially for younger nestlings. Older chicks grow somewhat faster when insects are abundant. Growth in younger nestlings increases with age, while in old nestlings, it decreases as they get older.[57] Young tree swallows are able to thermoregulate at least 75% as effectively as the adult at an average age of 9.5 days when out of the nest, and from four to eight days old when in the nest (depending on the size of the brood).[58] The nestlings fledge after about 18 to 22 days, with about 80% fledging success. Like hatching success, this is negatively affected by unfavourable weather and a younger female.[14] Chicks may be preyed on by snakes[59] and raccoons.[60] This predation can be exacerbated by begging calls.[61]
Feeding

The tree swallow forages up to 50 metres (160 ft) above the ground singly or in groups. Its flight is a mix of flapping and gliding. During the breeding season, this is mostly within 4 kilometres (2.5 mi) of the nest site. When it is foraging for nestlings, though, it usually goes up to 200 metres (660 ft) from the nest,[14] mostly staying in sight of it, and forages at a height up to 12 metres (39 ft).[62] As well as being caught in flight,[7] insects are sometimes taken from the ground, water, vegetation, and vertical surfaces.[14]
The tree swallow eats mostly insects, with some molluscs, spiders, and fruit. In North America, flies make up about 40% of the diet, supplemented with beetles and ants. Otherwise, the diet is about 90% flies.

Both sexes feed the nestlings (although the male feeds the chicks less than the females) resulting in about 10 to 20 feedings per hour.
Survival
The tree swallow has an average lifespan of 2.7 years[69] and a maximum of 12 years. About 79% of individuals do not survive their first year, and those that do face an annual mortality rate of 40% to 60%.[14] Most deaths are likely the result of cold weather, which reduces insect availability, leading to starvation.[7] Lifespan is associated with telomere length: a 2005 study that used return rates (to the breeding site of the previous year) as a proxy for survival found that those with the longest telomeres at one year of age had a predicted lifespan of 3.5 years, compared to the 1.2 years for those with the shortest telomeres.[70] Whether short telomeres cause a reduction in fitness or are simply an indicator of it is unknown. Regardless, a 2016 thesis found that measures of condition were positively correlated with telomere length. Males also generally had longer telomeres than females, as did smaller-winged birds. Individuals with shorter telomeres may compensate for potential losses in fitness by increasing reproductive effort, whereas those with longer telomeres may decrease their investment, as evidenced by the smaller proportion of chicks females with longer telomeres fledged.[71] Telomere length is highly heritable, and is especially dependent on that of the mother.[72]
Predation
The tree swallow is susceptible to a wide range of predators. Eggs, nestlings, and adults in the nest fall victim to
Parasites

The tree swallow is vulnerable to various parasites, such as the blood parasite
Immunology
In the breeding female tree swallow, humoral immunocompetence (HIC) is inversely correlated with laying date. This means that, on average, a bird that lays its eggs earlier has a stronger antibiotic response to an antigen than a bird that lays later. A tree swallow that is handicapped by wing-clipping generally has a lower HIC. These relationships could be interpreted as supporting the conclusion that a female that lays earlier acquires a higher HIC, but the authors of the study that found the correlations believed this unlikely, due to the colder temperatures near the start of the breeding season. Instead, they thought that HIC could be a measure of quality, and that a higher quality female is able to lay earlier. The authors also postulated that it is an indicator of workload, as shown by the lower HIC of handicapped birds.[81]
Higher quality female tree swallows (as measured by laying date) are able to maintain their reproductive effort while diverting resources to fight an immune challenge. Lower quality swallows are less able to do so; a 2005 study in Ithaca, New York, found that late-laying females with an artificially enlarged brood, although able to maintain offspring quality, had lower responses to an immune challenge than those that were of higher quality or did not have an enlarged brood.[82] Whether a female chooses to prioritize offspring quality or immunocompetence is likely related to survival probabilities; a 2005 study discovered that females with an enlarged brood in Alaska, where survival rates are lower, had weaker immune responses, but kept reproductive effort steady, whereas those in Tennessee, with higher survival rates, had a stronger response but lower quality offspring.[83]
In the tree swallow, some components of the immune system deteriorate with age. Acquired T cell-mediated immunity, for example, declines with age in the female tree swallow. But, the age of a female does not affect both the acquired and innate humoral immunity; the lack of deterioration in the former contrasts with studies on barn swallows and female collared flycatchers.[84] Because of this immunosenescence (a decrease in immune function with age), older females infected with a disease generally visit their nest less, resulting in their nestlings growing slower. They are also likely to lose weight because of an infection.[85]
Status
The tree swallow is considered to be
References
Notes
Citations
- ^ . Retrieved 12 November 2021.
- ^ Vieillot, Louis Pierre (1807). Histoire naturelle des oiseaux de l'Amérique Septentrionale: contenant un grand nombre d'espèces décrites ou figurées pour la première fois (in French). Vol. 1. Paris: Crapelet. p. 61.
- ^ Cabanis, Jean (1850). Museum Heineanum : Verzeichniss der ornithologischen Sammlung des Oberamtmann Ferdinand Heine auf Gut St. Burchard vor Halberstatdt (in German). Vol. 1. Halberstadt: Independently commissioned by R. Frantz. p. 48.
- ^ Coues, Elliott (1878). Birds of the Colorado Valley: a repository of scientific and popular information concerning North American ornithology. Washington, DC: Government Printing Office.
- ^ a b Coues, Elliott (1882). A check list of North American birds (2 ed.). Boston: Estes and Lauriat. p. 42.
- S2CID 216180193. Retrieved 14 January 2017.
- ^ ISBN 978-1-4081-3172-5.
- ISBN 978-1-4081-2501-4.
- ^ Cerasale, David J.; Dor, Roi; Winkler, David W.; Lovette, Irby J. (2012). "Phylogeny of the Tachycineta genus of New World swallows: Insights from complete mitochondrial genomes" (PDF). Energy balance in migratory birds: Insights from mitochondrial genomes and leptin receptor biology (PhD). Cornell University. pp. 91–122.
- PMID 11884168.
- PMID 28565131.
- PMID 22750631.
- ^ "Tree Swallow Identification, All About Birds, Cornell Lab of Ornithology". www.allaboutbirds.org. Retrieved 28 September 2020.
- ^ S2CID 216382529. Retrieved 10 December 2017.
- ^ a b Hussell, David J. T. (1983). "Age and plumage color in female tree swallows". Journal of Field Ornithology. 54 (3): 312–318.
- ISSN 0004-8038.
- S2CID 85030082.
- ^ JSTOR 2426582.
- ^ JSTOR 4089286.
- ^ S2CID 45389696.
- S2CID 86754716.
- S2CID 53173370.
- S2CID 53160897.
- ISSN 0012-9615.
- ^ S2CID 42958338.
- .
- S2CID 36003514.
- S2CID 28251342.
- JSTOR 1367216.
- JSTOR 1368072.
- ^ a b Winkler, David W. (1993). "Use and importance of feathers as nest lining in tree swallows (Tachycineta bicolor)". The Auk. 110 (1): 29–36.
- S2CID 55119375.
- ISSN 0908-8857.
- S2CID 46971093.
- PMID 10693819.
- ^ S2CID 4995233.
- S2CID 84919928.
- ISSN 1045-2249.
- ISSN 0004-8038.
- ^ S2CID 53195131.
- S2CID 13722818.
- S2CID 24760456.
- S2CID 25483760.
- ISSN 0004-8038.
- S2CID 22513713.
- ISSN 0008-4301.
- S2CID 53170228.
- ^ Hallinger, Kelly Kristen (2017). Context-dependent fitness consequences of extra-pair paternity in tree swallows (Tachycineta bicolor) (PhD). Cornell University.
- S2CID 91253157.
- ^ S2CID 32810854.
- S2CID 4975256.
- PMID 24909948.
- ^ ISBN 978-0-226-05781-1.
- ^ ISSN 0908-8857.
- ISBN 978-3-540-51759-7.
- S2CID 3720108.
- ISSN 0019-1019.
- ISSN 0043-5643.
- S2CID 19454000.
- ISSN 0008-4301.
- ISSN 1045-2249.
- ^ JSTOR 1369987.
- PMID 29217628.
- ISSN 1465-7279.
- ISSN 0908-8857.
- S2CID 4993059.
- S2CID 25507198.
- ^ S2CID 87271779.
- S2CID 25321642.
- PMID 17148169.
- ^ Belmaker, Amos (2016). "Patterns of co-variation of telomere length, condition, life history and fitness in a short-lived bird species". The Role of Telomere Length in Tree Swallow Behavior and Life History (PhD). Cornell University.
- PMID 31380080.
- ^ a b c d e Winkler, D. W.; Hallinger, K. K.; Ardia, D. R.; Robertson, R. J.; Stutchbury, B. J.; Chohen, R. R. (2011). Poole, A. F., ed. "Tree Swallow (Tachycineta bicolor)". The Birds of North America. Ithaca, New York: Cornell Lab of Ornithology.
- JSTOR 1939142.
- ^ Chapman, L. B. (1955). "Studies of a tree swallow colony". Bird-Banding. 6 (2): 45–70.
- ^ Errington, P. L. (1932). "Food habits of southern Wisconsin raptors. Part I. Owls". The Condor. 34 (4): 176–186.
- ^ a b Maass, Natalia May (2019). Perceived predation risk and the responses of adult and nestling tree swallows (Tachycineta bicolor) (MS). Eastern Kentucky University.
- ISSN 0008-4301.
- ^ a b Roby, Daniel D.; Brink, Karen L.; Wittmann, Karin (1992). "Effects of bird blowfly parasitism on eastern bluebird and tree swallow nestlings" (PDF). Wilson Bulletin. 104 (4): 630–643.
- ISSN 0908-8857.
- ISSN 1465-7279.
- ISSN 0021-8790.
- S2CID 33354318.
- PMID 17251097.
- PMID 21661557.
- ^ "Migratory Bird Treaty Act Protected Species (10.13 List)". US Fish & Wildlife Service. 2013. Retrieved 4 June 2018.
- ^ "Birds protected under the Migratory Birds Convention Act". Government of Canada. 2017. Archived from the original on 20 May 2019. Retrieved 21 July 2018.
- ISSN 0004-8038.
- JSTOR 1368686.
- S2CID 85573498.
- PMID 16297515.
- ISSN 0008-4301.
- S2CID 42492417.
- PMID 23468236.
- S2CID 222159036.
External links


