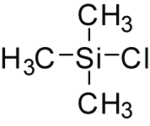
Trimethylsilyl chloride
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Chlorotri(methyl)silane | |||
| Other names
Trimethylsilyl chloride
Chlorotrimethylsilane TMSCl Trimethylchlorosilane TMCS | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
ECHA InfoCard
|
100.000.819 | ||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| UN number | 1298 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H9SiCl | |||
| Molar mass | 108.64 g/mol | ||
| Appearance | Colorless liquid, fumes in moist air | ||
| Density | 0.856 g/cm3, liquid | ||
| Melting point | −40 °C (−40 °F; 233 K) | ||
| Boiling point | 57 °C (135 °F; 330 K) | ||
| Reacts | |||
| −77.36·10−6 cm3/mol | |||
| Structure | |||
| Tetrahedral at Si | |||
| Hazards | |||
| GHS labelling: | |||
    
| |||
| Danger | |||
| H225, H301, H312, H314, H331, H351 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P280, P281, P301+P310, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P310, P311, P312, P321, P322, P330, P363, P370+P378, P403+P233, P403+P235, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −28 °C (−18 °F; 245 K) | ||
| 400 °C (752 °F; 673 K) | |||
| Related compounds | |||
Related halosilanes
|
Trimethylsilyl fluoride Trimethylsilyl bromide Trimethylsilyl iodide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Trimethylsilyl chloride, also known as chlorotrimethylsilane is an
Preparation
TMSCl is prepared on a large scale by the
Typically about 2–4% of the product stream is the monochloride, which forms an azeotrope with MeSiCl3.
Reactions and uses
TMSCl is reactive toward nucleophiles, resulting in the replacement of the chloride. In a characteristic reaction of TMSCl, the nucleophile is water, resulting in hydrolysis to give the hexamethyldisiloxane:
Silylation in organic synthesis
By the process of
Trimethylsilyl chloride also reacts with carbanions to give trimethylsilyl derivatives.
In the presence of
Dehydrations
Dehydration of metal chlorides with trimethylsilyl chloride in THF gives the solvate as illustrated by the case of
Other reactions
Trimethylsilyl chloride is used to prepare other trimethylsilyl halides and
Reduction of trimethylsilyl chloride give hexamethyldisilane:
References
- ISBN 978-3527306732.)
{{cite encyclopedia}}: CS1 maint: multiple names: authors list (link - ^ Such as in Norbert Zander and Ronald Frank (2005). "The use of polystyrylsulfonyl chloride resin as a solid supported condensation reagent for the formation of esters: Synthesis of N-[(9-fluorenylmethoxy)carbonyl]-L-aspartic acid; α tert-butyl ester, β-(2-ethyl[(1E)-(4-nitrophenyl)azo]phenyl]amino]ethyl ester". Organic Syntheses. 81: 235.
- .
- ^ Yoshihiko Ito, Shotaro Fujii, Masashi Nakatuska, Fumio Kawamoto, and Takeo Saegusa (1979). "One-Carbon Ring Expansion of Cycloalkanones to Conjugated Cycloalkenone: 2-Cyclohepten-1-one". Organic Syntheses. 59: 113
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 1, p. 327. - )
- ^ L. Birkofer and P. Wegner (1970). "Trimethylsilyl azide". Organic Syntheses. 50: 107; Collected Volumes, vol. 6, p. 1030.
- ^ Brook, Michael A. (2000). Silicon in Organic, Organometallic, and Polymer Chemistry. New York: John Wiley & Sons. pp. 193–194.




![{\displaystyle x\ {\ce {MeCl + Si}}\longrightarrow {\begin{cases}{\ce {Me3SiCl}},\\[2pt]{\ce {Me2SiCl2}},\\[2pt]{\ce {MeSiCl3}},\\[2pt]{\text{etc.}}\end{cases}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/1690fa91cc5b4389913c67077f81c5b4428e56cb)




