Triterpene
Triterpenes are a class of
Structures
Triterpenes exist in a great variety of structures. Nearly 200 different skeletons have been identified.[3] These skeletons may be broadly divided according to the number of rings present. In general pentacyclic structures (5 rings) tend to dominate.
| Number of rings | Examples |
|---|---|
| 0 | Squalene |
| 1 | Achilleol A [4] |
| 2 | Polypodatetraene |
| 3 | Malabaricane |
| 4 | Lanostane, Cucurbitacin |
| 5 | Hopane, Oleanane, Ursolic acid |
| 6 | Chamaecydin |
Squalene is biosynthesized through the head-to-head condensation of two farnesyl pyrophosphate units. This coupling converts a pair of C15 components into a C30 product. Squalene serves as precursor for the formation of many triterpenoids, including bacterial hopanoids and eukaryotic sterols.
Triterpenoids
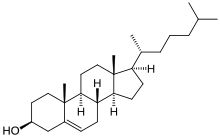
By definition triterpenoids are triterpenes that possess heteroatoms, usually oxygen. The terms triterpene and triterpenoid often are used interchangeably.
Triterpenoids possess a rich chemistry and pharmacology (e.g. cholesterol) with several pentacyclic motifs. Lupane, oleanane and ursane show particular promise as anti-cancer agents.[5][6]
Steroids
Steroids feature a
Triterpenoid saponins
References
- ISBN 9783527609949.
- ISBN 978-3-540-66573-1.
- PMID 14751299.
- ISSN 0040-4039.
- PMID 19742422.
- PMID 8847885.
- PMID 21333312.
- PMID 10571242.

