Tropane
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
N-Methyl-8-azabicyclo[3.2.1]octane
| |||
| Other names
2,3-Dihydro-8-methylnortropidine
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 6379695 | |||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.156.627 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C8H15N | |||
| Molar mass | 125.211 g/mol | ||
| Density | 0.9259 at 15 °C | ||
| Boiling point | 163 to 169 °C (325 to 336 °F; 436 to 442 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
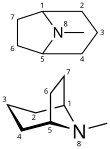
Tropane is a
Structurally, tropane is cycloheptane with a nitrogen bridge between carbons 1 and 5 and an additional methyl group attached to the nitrogen. While carbons 1 and 5 are asymmetric carbons, tropane itself is optically inactive due to mirror symmetry.
8-Azabicyclo[3.2.1]octane (tropane without the N-methyl group) is known as nortropane or nor-tropane.
See also
References
- ^ Merck Index, 11th Edition, 9689.
- ^ "Atropine content of plants". USDA, ARS, National Genetic Resources Program. Phytochemical and Ethnobotanical Databases. [Online Database] National Germplasm Resources Laboratory, Beltsville, Maryland. Archived from the original on November 7, 2004. Retrieved July 25, 2005.
- ^ "Cocaine content of plants". USDA, ARS, National Genetic Resources Program. Phytochemical and Ethnobotanical Databases. [Online Database] National Germplasm Resources Laboratory, Beltsville, Maryland. Archived from the original on November 7, 2004. Retrieved July 25, 2005.