Tropinone

| |

| |
| Names | |
|---|---|
| IUPAC name
8-Methyl-8-azabicyclo[3.2.1]octan-3-one
| |
| Other names
3-Tropinone
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.007.756 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H13NO | |
| Molar mass | 139.195 g/mol |
| Appearance | Brown solid |
| Melting point | 42.5 °C (108.5 °F; 315.6 K) |
| Boiling point | (decomposes) |
| Hazards | |
| GHS labelling: | |
  [1] [1]
| |
| Danger | |
| H302, H314[1] | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tropinone is an
Synthesis
The first synthesis of tropinone was by Richard Willstätter in 1901. It started from the seemingly related cycloheptanone, but required many steps to introduce the nitrogen bridge; the overall yield for the synthesis path is only 0.75%.[5] Willstätter had previously synthesized cocaine from tropinone, in what was the first synthesis and elucidation of the structure of cocaine.[6]

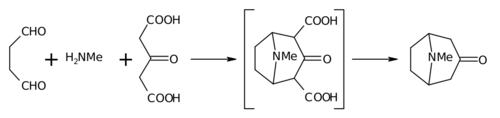
Robinson's "double Mannich" reaction
The 1917 synthesis by Robinson is considered a classic in
This reaction is described as an intramolecular "double Mannich reaction" for obvious reasons. It is not unique in this regard, as others have also attempted it in piperidine synthesis.[9][10]
In place of acetone, acetonedicarboxylic acid is known as the "
Reaction mechanism
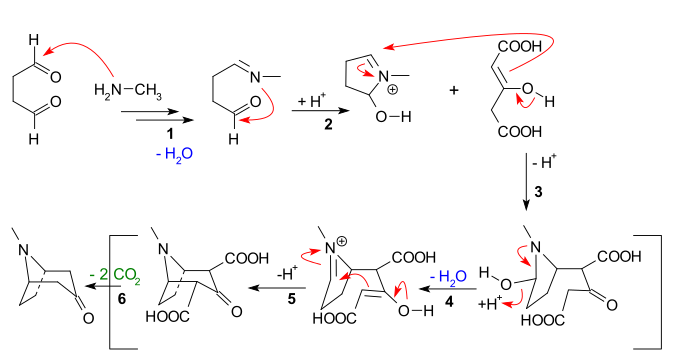
The main features apparent from the reaction sequence below are:
- Nucleophilic addition of methylamine to succinaldehyde, followed by loss of water to create an imine
- Intramolecular addition of the imine to the second aldehyde unit and first ring closure
- Intermolecular Mannich reaction of the enolateof acetone dicarboxylate
- New enolate formation and new imine formation with loss of water for
- Second intramolecular Mannich reaction and second ring closure
- Loss of 2 carboxylic groups to tropinone
Some authors have actually tried to retain one of the CO2H groups.[11]
CO2R-tropinone has 4 stereoisomers, although the corresponding ecgonidine alkyl ester has only a pair of enantiomers.
From cycloheptanone
Biochemistry method
This section is empty. You can help by adding to it. (April 2022) |
Reduction of tropinone
The reduction of tropinone is mediated by

See also
- Benztropine
- Daturaolone
- 2-Carbomethoxytropinone (2-CMT) an intermediate in the creation of ecgonine cocaine analogues
- Ecgonidine
References
- ^ a b "Tropinone". Substance Information. ECHA.
- .
- PMID 10649349.
- ^ Chemical Entities of Biological Interest Identification code: ChEBI:57851 "tropiniumone"
- ^ ISBN 978-0-85404-544-0.
- PMID 11699882.
- ISBN 978-0-12-372532-5.
- S2CID 143267467.
- PMID 10669562.
- PMID 11425577.
- .
- ^ U.S. patent 8,609,690
- PMID 11878978.
- PMID 30538251.
- .
- PMID 10626376.