Tumor necrosis factor
Ensembl |
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| UniProt | |||||||||
| RefSeq (mRNA) | |||||||||
| RefSeq (protein) | |||||||||
| Location (UCSC) | Chr 6: 31.58 – 31.58 Mb | Chr 17: 35.42 – 35.42 Mb | |||||||
| PubMed search | [3] | [4] | |||||||
| View/Edit Human | View/Edit Mouse |
Tumor necrosis factor (TNF, cachexin, or cachectin; formerly known as tumor necrosis factor alpha or TNF-α[5][6]) is an adipokine and a cytokine. TNF is a member of the TNF superfamily, which consists of various transmembrane proteins with a homologous TNF domain.
TNF signaling occurs through two receptors:
TNF-α exists as a transmembrane form (mTNF-α) and as a soluble form (sTNF-α). sTNF-α results from enzymatic cleavage of mTNF-α,[10] by a process called substrate presentation. mTNF-α is mainly found on monocytes/macrophages where it interacts with tissue receptors by cell-to-cell contact.[10] sTNF-α selectively binds to TNFR1, whereas mTNF-α binds to both TNFR1 and TNFR2.[11] TNF-α binding to TNFR1 is irreversible, whereas binding to TNFR2 is reversible.[12]
The primary role of TNF is in the regulation of
As an adipokine, TNF promotes insulin resistance, and is associated with obesity-induced type 2 diabetes.[20] As a cytokine, TNF is used by the immune system for cell signaling. If macrophages (certain white blood cells) detect an infection, they release TNF to alert other immune system cells as part of an inflammatory response.[20] Certain cancers can cause overproduction of TNF. TNF parallels parathyroid hormone both in causing secondary hypercalcemia and in the cancers with which excessive production is associated. Under the name tasonermin, TNF is used as an immunostimulant drug in the treatment of certain cancers. Drugs that counter the action of TNF are used in the treatment of various inflammatory diseases such as rheumatoid arthritis.
Discovery
The theory of an
The
Research in the laboratory led by Mark Mattson has shown that TNF can prevent the death/apoptosis of neurons by a mechanism involving activation of the transcription factor NF-κB which induces the expression of antioxidant enzymes and Bcl-2.[31][32]
Gene
The human TNF gene was cloned in 1985.[33] It maps to chromosome 6p21.3, spans about 3 kilobases and contains 4 exons. The last exon shares similarity with lymphotoxin alpha (LTA, once named as TNF-β).[34] The three prime untranslated region (3'-UTR) of TNF contains an AU-rich element (ARE).
Structure
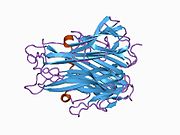
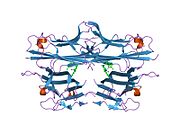
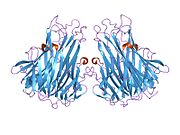
TNF is primarily produced as a 233-amino acid-long type II transmembrane protein arranged in stable homotrimers.[35][36] From this membrane-integrated form the soluble homotrimeric cytokine (sTNF) is released via proteolytic cleavage by the metalloprotease TNF alpha converting enzyme (TACE, also called ADAM17).[37] The soluble 51 kDa trimeric sTNF tends to dissociate at concentrations below the nanomolar range, thereby losing its bioactivity. The secreted form of human TNF takes on a triangular pyramid shape, and weighs around 17-kDa. Both the secreted and the membrane bound forms are biologically active, although the specific functions of each is controversial. But, both forms do have overlapping and distinct biological activities.[38]
The common house mouse TNF and human TNF are structurally different.
Cell signaling
TNF can bind two receptors,

Upon contact with their ligand, TNF receptors also form trimers, their tips fitting into the grooves formed between TNF monomers. This binding causes a conformational change to occur in the receptor, leading to the dissociation of the inhibitory protein SODD from the intracellular death domain. This dissociation enables the adaptor protein TRADD to bind to the death domain, serving as a platform for subsequent protein binding. Following TRADD binding, three pathways can be initiated.[41][42]
- Activation of apoptoticfactors.
- Activation of the apoptotic.
- Induction of death signaling: Like all death-domain-containing members of the TNFR superfamily, TNFR1 is involved in death signaling..
The myriad and often-conflicting effects mediated by the above pathways indicate the existence of extensive cross-talk. For instance, NF-κB enhances the transcription of
In animal models TNF selectively kills autoreactive T cells.[46]
There is also evidence that TNF-α signaling triggers downstream epigenetic modifications that result in lasting enhancement of pro-inflammatory responses in cells.[47][48][49][50]
Enzyme regulation
This protein may use the morpheein model of allosteric regulation.[51]
Clinical significance
TNF was thought to be produced primarily by
It has a number of actions on various organ systems, generally together with IL-1 and
- On the hypothalamus:
- On the insulin receptor substrate-1(IRS-1), which impairs insulin signaling
- It is a potent chemoattractant for neutrophilsmigrate.
- On macrophages: stimulates phagocytosis, and production of IL-1 oxidants and the inflammatory lipid prostaglandin E2 (PGE2)
- On other tissues: increasing insulin resistance. TNF phosphorylates insulin receptor serine residues, blocking signal transduction.
- On metabolism and food intake: regulates bitter taste perception.[55]
A local increase in concentration of TNF will cause the cardinal signs of Inflammation to occur: heat, swelling, redness, pain and loss of function.
Whereas high concentrations of TNF induce shock-like symptoms, the prolonged exposure to low concentrations of TNF can result in cachexia, a wasting syndrome. This can be found, for example, in cancer patients.
Said et al. showed that TNF causes an IL-10-dependent inhibition of CD4 T-cell expansion and function by up-regulating PD-1 levels on monocytes which leads to IL-10 production by monocytes after binding of PD-1 by PD-L.[56]
The research of Pedersen et al. indicates that TNF increase in response to
In the brain TNF can protect against excitotoxicity.[32] TNF strengthens synapses.[7] TNF in neurons promotes their survival, whereas TNF in macrophages and microglia results in neurotoxins that induce apoptosis.[32]
TNF-α and IL-6 concentrations are elevated in obesity.[59][60][61] Use of monoclonal antibodies against TNF-α is associated with increases rather than decreases in obesity, indicating that inflammation is the result, rather than the cause, of obesity.[61] TNF and IL-6 are the most prominent cytokines predicting COVID-19 severity and death.[20]
TNFα in Liver Fibrosis
TNFα mediates the inflammation that activates resident
While TNFα treatment suppresses collagen α1 gene expression, apoptosis, and proliferation in activated HSCs in vitro, an activity that should ameliorate fibrosis, it has also been shown to inhibit apoptosis in activated HSCs, an activity which should, in principle, induce fibrosis.[63] Specifically, TNFα produced by hepatic macrophages is known to support the survival of HSCs, the source of hepatic myofibroblasts.[64] TNFα is therefore believed to promote liver fibrosis through its pro-survival effect, despite its pleiotropic effects on HSCs.[65]
Yet another way TNFα contributes to the worsening of liver fibrosis is by stimulating the production of
It should furthermore be noted that CCR9+ macrophages, which play an essential role in the pathogenesis of liver fibrosis, are TNFα-dependent. When TNFα is attenuated using an anti-TNFα antibody, hepatic HSCs are not activated by CCR9+ macrophages.[67]
TNFα in NAFLD
TNFα has a dual role in the development of
Secondly, the binding of TNFα to TNFα receptor 1 (
It is additionally thought that TNFα increases the production of
The dual role of TNFα in the development of
TNFα is, therefore, thought to play a deleterious role in the progression of
Pharmacology
TNF promotes the inflammatory response, which, in turn, causes many of the clinical problems associated with autoimmune disorders such as rheumatoid arthritis, ankylosing spondylitis, inflammatory bowel disease, psoriasis, hidradenitis suppurativa and refractory asthma. These disorders are sometimes treated by using a TNF inhibitor. This inhibition can be achieved with a monoclonal antibody such as infliximab (Remicade) binding directly to TNF, adalimumab (Humira), certolizumab pegol (Cimzia) or with a decoy circulating receptor fusion protein such as etanercept (Enbrel) which binds to TNF with greater affinity than the TNFR.[76]
On the other hand, some patients treated with TNF inhibitors develop an aggravation of their disease or new onset of autoimmunity. TNF seems to have an immunosuppressive facet as well. One explanation for a possible mechanism is this observation that TNF has a positive effect on regulatory T cells (Tregs), due to its binding to the tumor necrosis factor receptor 2 (TNFR2).[77]
Anti-TNF therapy has shown only modest effects in cancer therapy. Treatment of renal cell carcinoma with infliximab resulted in prolonged disease stabilization in certain patients. Etanercept was tested for treating patients with breast cancer and ovarian cancer showing prolonged disease stabilization in certain patients via downregulation of IL-6 and CCL2. On the other hand, adding infliximab or etanercept to gemcitabine for treating patients with advanced pancreatic cancer was not associated with differences in efficacy when compared with placebo.[78]
Interactions
TNF has been shown to
Nomenclature
Because LTα is no longer referred to as TNFβ,[81] TNFα, as the previous gene symbol, is now simply called TNF, as shown in HGNC (HUGO Gene Nomenclature Committee) database.
References
- ^ a b c ENSG00000230108, ENSG00000223952, ENSG00000204490, ENSG00000228321, ENSG00000232810, ENSG00000228849, ENSG00000206439 GRCh38: Ensembl release 89: ENSG00000228978, ENSG00000230108, ENSG00000223952, ENSG00000204490, ENSG00000228321, ENSG00000232810, ENSG00000228849, ENSG00000206439 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024401 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 33086075.
- ISBN 978-0-323-09138-1.
The simplified name TNF is now preferred over the former designation TNF-α because the corresponding term TNF-β, an alternative name for LT, is now obsolete.
- ^ PMID 33192311.
- ^ PMID 33324405.
- PMID 33053859.
- ^ PMID 29234328.
- PMID 26117714.
- S2CID 40818956.
- S2CID 6544784.
- S2CID 7657797.
- S2CID 230209.
- PMID 12851985.
- PMID 12077089.
- PMID 17206706.
- PMID 27991935.
- ^ S2CID 238991468.
- PMID 5249808.
- PMID 5693925.
- PMID 1103152.
- PMID 6166564.
- S2CID 26446784.
- S2CID 4245957.
- S2CID 4339006.
- PMID 3895437.
- PMID 3764421.
- S2CID 4308324.
- PMID 16397579.
- ^ PMID 18774186.
- PMID 2413547.
- PMID 2995927.
- S2CID 31789769.
- PMID 8679576.
- S2CID 4251053.
- S2CID 1028523.
- PMID 17442953.
In human cells, contrary to results previously obtained in a rodent model, TNF seems not to be glycosylated and, thus, trafficking is carbohydrate independent. In an effort to localize the amino acid motif responsible for granule targeting, we constructed additional fusion proteins and analyzed their trafficking, concluding that granule-targeting sequences are localized in the mature chain of TNF and that the cytoplasmic tail is expendable for endocytotic sorting of this cytokine, thus excluding direct interactions with intracellular adaptor proteins
- ^ Theiss. A. L. et al. 2005. Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibrobasts via TNF Receptor 2. The Journal of Biological Chemistry. [Online] 2005. Available at: http://www.jbc.org/content/280/43/36099.long Accessed: 21/10/14
- PMID 12655295.
- S2CID 25321662.
- PMID 21979919.
- PMID 14555214.
- PMID 18755894.
- PMID 31840058.
- PMID 30824537.
- PMID 31611260.
- PMID 28218970.
- PMID 22182754.
- PMID 17442953.
- PMID 9218250.
- PMID 1709737.
- PMID 25911043.
- PMID 20208540.
- S2CID 30200779.
- PMID 19752112.
- PMID 11681809.
- PMID 30591653.
- ^ PMID 30165404.
- PMID 9247485.
- S2CID 85343893.
- PMID 23553591.
- PMID 26726307.
- S2CID 25137248.
- PMID 23460364.
- PMID 11474306.
- PMID 30090738.
- PMID 18339717.
- PMID 18339717.
- PMID 32235829.
- PMID 16691291.
- PMID 11027549.
- S2CID 25137248.
- PMID 18360618.
- PMID 29593717.
- PMID 26854213.
- S2CID 11683986.
- S2CID 17145731.
- S2CID 36721785.
External links
- Tumor Necrosis Factor-alpha at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Overview of all the structural information available in the PDB for UniProt: P01375 (Tumor necrosis factor) at the PDBe-KB.