Tyrosine
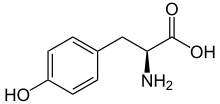 Skeletal formula of L-tyrosine
| |||
 L-Tyrosine at physiological pH
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
(S)-Tyrosine
| |||
| Other names
L-2-Amino-3-(4-hydroxyphenyl)propanoic acid
| |||
| Identifiers | |||
| |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.000.419 | ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C9H11NO3 | |||
| Molar mass | 181.191 g·mol−1 | ||
| Appearance | white solid | ||
| .0453 g/100 mL | |||
| -105.3·10−6 cm3/mol | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Supplementary data page | |||
| Tyrosine (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
L-Tyrosine or tyrosine (symbol Tyr or Y)
The one-letter symbol Y was assigned to tyrosine for being alphabetically nearest of those letters available. Note that T was assigned to the structurally simpler threonine, U was avoided for its similarity with V for valine, W was assigned to tryptophan, while X was reserved for undetermined or atypical amino acids.[6] The mnemonic tYrosine was also proposed.[7]
Functions
Aside from being a
Similar functionality is also presented in serine and threonine, whose side chains have a hydroxy group, but are alcohols. Phosphorylation of these three amino acids' moieties (including tyrosine) creates a negative charge on their ends, that is greater than the negative charge of the only negatively charged aspartic and glutamic acids. Phosphorylated proteins keep these same properties—which are useful for more reliable protein-protein interactions—by means of phosphotyrosine, phosphoserine and phosphothreonine.[8]
Binding sites for a signalling phosphoprotein may be diverse in their chemical structure.[9]
Phosphorylation of the hydroxyl group can change the activity of the target protein, or may form part of a signaling cascade via SH2 domain binding.[10]
A tyrosine residue also plays an important role in photosynthesis. In chloroplasts (photosystem II), it acts as an electron donor in the reduction of oxidized chlorophyll. In this process, it loses the hydrogen atom of its phenolic OH-group. This radical is subsequently reduced in the photosystem II by the four core manganese clusters.[11]
Dietary requirements and sources
The Dietary Reference Intake for tyrosine is usually estimated together with phenylalanine. It varies depending on an estimate method, however the ideal proportion of these two amino acids is considered to be 60:40 (phenylalanine:tyrosine) as a human body has such composition.[12] Tyrosine, which can also be synthesized in the body from phenylalanine, is found in many high-
Biosynthesis
In plants and most microorganisms, tyrosine is produced via
Mammals synthesize tyrosine from the essential amino acid phenylalanine (Phe), which is derived from food. The conversion of Phe to Tyr is catalyzed by the enzyme phenylalanine hydroxylase, a monooxygenase. This enzyme catalyzes the reaction causing the addition of a hydroxyl group to the end of the 6-carbon aromatic ring of phenylalanine, such that it becomes tyrosine.
Metabolism

Phosphorylation and sulfation
Some of the tyrosine residues can be tagged (at the hydroxyl group) with a phosphate group (phosphorylated) by protein kinases. In its phosphorylated form, tyrosine is called phosphotyrosine. Tyrosine phosphorylation is considered to be one of the key steps in signal transduction and regulation of enzymatic activity. Phosphotyrosine can be detected through specific antibodies. Tyrosine residues may also be modified by the addition of a sulfate group, a process known as tyrosine sulfation.[17] Tyrosine sulfation is catalyzed by tyrosylprotein sulfotransferase (TPST). Like the phosphotyrosine antibodies mentioned above, antibodies have recently been described that specifically detect sulfotyrosine.[18]
Precursor to neurotransmitters and hormones
In dopaminergic cells in the
The
are also derived from tyrosine.Precursor to other compounds
The latex of
Degradation
The decomposition of L-tyrosine (syn. para-hydroxyphenylalanine) begins with an α-ketoglutarate dependent
The next oxidation step catalyzes by
Thereby
Phloretic acid is also a urinary metabolite of tyrosine in rats.[26]
Ortho- and meta-tyrosine
Three
Medical use
Tyrosine is a precursor to neurotransmitters and increases plasma neurotransmitter levels (particularly dopamine and norepinephrine),[29] but has little if any effect on mood in normal subjects.[30][31][32]
A 2015 systematic review found that "tyrosine loading acutely counteracts decrements in working memory and information processing that are induced by demanding situational conditions such as extreme weather or cognitive load" and therefore "tyrosine may benefit healthy individuals exposed to demanding situational conditions".[33]
Industrial synthesis
L-tyrosine is used in
See also
- Albinism
- Alkaptonuria
- Betalain
- Iodinated tyrosine derivatives
- Pauly reaction
- Tyramine
- Tyrosine sulfation
- Tyrosinemia
References
- ^ .
- ^ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008. Retrieved 5 March 2018.
- ^ "Tyrosine". The Columbia Electronic Encyclopedia, 6th ed. Infoplease.com — Columbia University Press. 2007. Retrieved 2008-04-20.
- ^ Harper D (2001). "Tyrosine". Online Etymology Dictionary. Retrieved 2008-04-20.
- ^ "Amino Acids - Tyrosine". www.biology.arizona.edu. Retrieved 2018-01-31.
- .
- .
- PMID 22889903.
- PMID 34270697.
- PMID 22889907.
- PMID 25260243.
- PMID 17513429.
- ^ Nutient Ranking Tool. MyFoodData.com. https://tools.myfooddata.com/nutrient-ranking-tool/tyrosine/all/highest
- ^ "Tyrosine". University of Maryland Medical Center. Archived from the original on 2013-06-04. Retrieved 2011-03-17.
- ^ a b Top 10 Foods Highest in Tyrosine
- ^ Nutient Ranking Tool. MyFoodData.com. https://tools.myfooddata.com https://tools.myfooddata.com/nutrient-ranking-tool/tyrosine/meats/highest/ounces/common/no
- PMID 17046811.
- PMID 22406006.
- PMID 19948186.
- PMID 15860375.
- PMID 24374199.
- ISSN 0368-1769.
- PMID 20494114.
- PMID 27060254.
- .
- .
- PMID 16221230.
- S2CID 31154432.
- PMID 6885965.
- PMID 6764931.
- S2CID 33823121.
- PMID 4025206.
- S2CID 30331663.
- ^ S2CID 23088822.
- .