Uranium hexachloride
| |
| Names | |
|---|---|
| IUPAC name
Uranium(VI) chloride
| |
| Other names
Uranium hexachloride
Peruranic chloride | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| UCl6 | |
| Molar mass | 450.745 g/mol |
| Appearance | dark green crystalline solid |
| Density | 3.6 g/cm3 |
| Melting point | 177 °C (351 °F; 450 K) |
| Related compounds | |
Other anions
|
Uranium hexafluoride |
Other cations
|
Tungsten hexachloride |
Related uranium chlorides
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
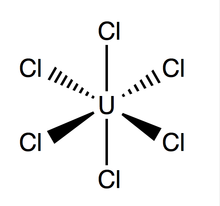
Uranium hexachloride (UCl6) is an
inorganic chemical compound of uranium in the +6 oxidation state.[1][2] UCl6 is a metal halide composed of uranium and chlorine. It is a multi-luminescent dark green crystalline solid with a vapor pressure between 1-3 mmHg at 373.15 K.[3] UCl6 is stable in a vacuum, dry air, nitrogen and helium at room temperature. It is soluble in carbon tetrachloride
(CCl4). Compared to the other uranium halides, little is known about UCl6.
Structure and Bonding
Uranium hexachloride has an
X-ray diffraction is 2.42 Å),[4]
and the distance between two adjacent chlorine atoms is 3.65 Å.
Chemical properties
Uranium hexachloride is a highly
hygroscopic compound and decomposes readily when exposed to ordinary atmospheric conditions.[5]
therefore it should be handled in either a vacuum apparatus or in a dry box.
Thermal decomposition
UCl6 is stable up to temperatures between 120 °C and 150 °C. The
.- 2 UCl6 (g) → 2 UCl5 (s) + Cl2 (g)
Solubility
UCl6 is not a very
soluble compound. It dissolves in CCl4 to give a brown solution. It is slightly soluble in isobutyl bromide and in fluorocarbon (C7F16).[6]
| Solvents | Temperature (°C) | Grams of UCl6/100g of solution |
|---|---|---|
| CCl4 | −18 | 2.64 |
| CCl4 | 0 | 4.9 |
| CCl4 | 20 | 7.8 |
| 6.6% Cl2 : 93.4% CCl4 | −20 | 2.4 |
| 12.5% Cl2 : 87.5% CCl4 | −20 | 2.23 |
| 12.5% Cl2 : 87.5% CCl4 | 0 | 3.98 |
| Liquid Cl2 | −33 | 2.20 |
| CH3Cl | −24 | 1.16 |
| Benzene | 80 | Insoluble
|
Freon 113
|
45 | 1.83 |
Reaction with hydrogen fluoride
When UCl6 is reacted with purified anhydrous liquid hydrogen fluoride (HF) at room temperature produces UF5.[6]
- 2 UCl6 + 10 HF → 2 UF5 + 10 HCl + Cl2
Synthesis
Uranium hexachloride can be synthesized from the reaction of
reactant (ideal temperature 100 °C - 125 °C). The reaction is carried out in a closed gas-tight vessel (for example a glovebox
) that can withstand the pressure that builds up.
Step 1: 2 UO3 + 5 Cl2 → 2 UCl5 + 3 O2
Step 2: 2 UCl5 + Cl2 → 2 UCl6
Overall reaction: 2 UO3 + 6 Cl2 → 2 UCl6 + 3 O2
This metal hexahalide can also be synthesized by blowing Cl2 gas over sublimed UCl4 at 350 °C.[8]
Step 1: 2 UCl4 + Cl2 → 2 UCl5
Step 2: 2 UCl5 + Cl2 → 2 UCl6
Overall Reaction: UCl4 + Cl2 → UCl6
References
- .
- .
- ^ Van Dyke, R. E.; Evers, E. C. (1955). "Preparation of Uranium Hexachloride". Google Patents: 2.
- PMID 15634063.
- ^ Lipkin, D.; Wessman, S. (1955). "Process and Apparatus for protecting Uranium hexachloride from Deterioration and Contamination". Google Patents: 2.
- ^ a b c Katz, J.J.; Rabinowitch,E. (1951). The Chemistry of Uranium. Ann Arbor: The McGraw-Hill Book Company.
- ^ Van Dyke, R. E.; Evers, E. C. (1955). "Preparation of Uranium Hexachloride". Google Patents: 2.
- doi:10.1063/1.437313.
