Urokinase
| PLAU | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
| Wikidata | ||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| ATC code | |
| Identifiers | |
| CAS Number | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C1376H2145N383O406S18 |
| Molar mass | 31126.65 g·mol−1 |
| | |
Urokinase, also known as urokinase-type plasminogen activator (uPA), is a
Urokinase is encoded in humans by the PLAU gene, which stands for "plasminogen activator, urokinase".[7] The same symbol represents the gene in other animal species.
Function
The PLAU gene encodes a serine protease (EC 3.4.21.73) involved in degradation of the extracellular matrix and possibly tumor cell migration and proliferation. A specific polymorphism in this gene may be associated with late-onset Alzheimer disease and also with decreased affinity for fibrin-binding. The protein encoded by this gene converts plasminogen to plasmin by specific cleavage of an Arg-Val bond in plasminogen. This gene's proprotein is cleaved at a Lys-Ile bond by plasmin to form a two-chain derivative in which a single disulfide bond connects the amino-terminal A-chain to the catalytically active, carboxy-terminal B-chain. This two-chain derivative is also called HMW-uPA (high molecular weight uPA). HMW-uPA can be further processed into LMW-uPA (low molecular weight uPA) by cleavage of chain A into a short chain A (A1) and an amino-terminal fragment. LMW-uPA is proteolytically active but does not bind to the uPA receptor.[8]
Structure
Urokinase is a 411-residue protein, consisting of three domains: the serine protease domain (consisting of residues 159-411), the kringle domain (consisting of residues 50-131), and the EGF-like domain (consisting of residues 1-49). The kringle domain and the serine protease domain are connected by an interdomain linker or connecting peptide (consisting of residues 132-158). Urokinase is synthesized as a zymogen form (prourokinase or single-chain urokinase), and is activated by proteolytic cleavage between Lys158 and Ile159. The two resulting chains are kept together by a disulfide bond between Cys148 and Cys279.[9]
In comparison to the mammalian system,
Interaction partners
The most important inhibitors of urokinase are the serpins plasminogen activator inhibitor-1 (PAI-1) and plasminogen activator inhibitor-2 (PAI-2), which inhibit the protease activity irreversibly. In the extracellular matrix, urokinase is tethered to the cell membrane by its interaction to the urokinase receptor.
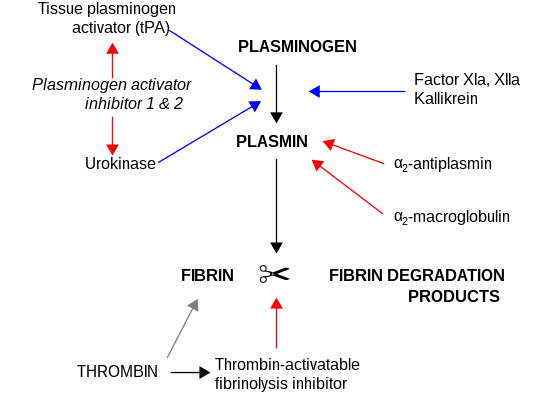
uPa also interacts with protein C inhibitor.[11][12]
zfuPA-a and zfuPA-b are poor activators of human plasminogen, while human uPA is a poor activator of salmon plasminogen. With the primary difference between the zebrafish uPA and human uPA being in the EGF domain.[10]
Urokinase and cancer
Elevated
uPA breakdown of the extracellular matrix is crucial for initiating the angiogenesis which is associated with cancer growth.[14]
uPA antigen is elevated in breast cancer tissue, which correlates with poor prognosis in breast cancer patients.[14] For this reason, uPA can be used as a diagnostic biomarker in breast cancer.[14]
Through its interaction with the
As of December 7, 2012, Mesupron (upamostat), a small molecule serine protease inhibitor developed by the WILEX pharmaceutical company, has completed phase II trials.[17] Mesupron appears to be safe when combined with chemotherapeutic drug Capecitabine for the progression-free survival in human breast cancer.[18]
Clinical applications
Urokinase is effective for the restoration of flow to intravenous catheters blocked by clotted blood or fibrin (catheter clearance). Catheters are used extensively to administer treatments to patients for such purposes as dialysis, nutrition, antibiotic treatment and cancer treatment. Approximately 25% of catheters become blocked, meaning that affected patients cannot receive treatment until the catheter has been cleared or replaced. Urokinase is also used clinically as a
All plasminogen activators (urokinase, tPA) catalyze the production of plasmin, which in turn leads to the breakdown of the fibrin mesh structure in blood clots. While there are commonalities in the mode of action for urokinase and tPA, urokinase has some advantages for treatment of peripheral clots (Pulmonary Embolism, Deep Vein Thrombosis, Peripheral arterial occlusive disease).
Unlike tPA, which is activated by binding to the fibrin within clots, urokinase is not sequestered by fibrin and therefore does not specifically attack hemostatic clots. This makes urokinase less likely to break down such hemostatic clots that are essential for ongoing blood vessel repair throughout the body. Dissolution of these “good” clots can lead to serious adverse events through hemorrhagic bleeding. Years of clinical study have confirmed the safety advantage of using urokinase.
A revolutionary method for the production of urokinase was patented by Evelyn Nicol in 1976 (U.S. Patent No. 3,930,944). Nicol was believed to be the first African American woman to receive a molecular biology patent.[21]
Society and culture
The presence of a
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000122861 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000021822 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 21711231.
- PMID 23201006.
- PMID 2415429.
- ^ "Entrez Gene: PLAU plasminogen activator, urokinase".
- PMID 19273310.
- ^ PMID 22733817.
- PMID 2752144.
- PMID 2551064.
- ^ PMID 30419600.
- ^ PMID 29484286.
- PMID 10738907.
- S2CID 205943879.
- ^ "Gemcitabine With or Without WX-671 in Treating Patients With Locally Advanced Pancreatic Cancer That Cannot Be Removed By Surgery". ClinicalTrials.gov. 28 March 2012.
- ^ "Fox Chase Cancer Center : New Small Molecule Inhibitor Could be a Safe and First-Line Treatment for Metastatic Breast Cancer". Press Release. Temple University Health System.
- PMID 10735422.
- S2CID 470599.
- ^ "Evelyn Nicol 1930 - 2020 - Obituary". www.legacy.com. Retrieved 2020-08-28.
- S2CID 4125748.
- ^ Sobel GW, Mohler SR, Jones NW, Dowdy ABC, Guest MM. Urokinase: an activator of plasma profibrinolysin extracted from urine. Am J Physiol 1952; 171: 768-69.
- .
Normal human and dog urine contains fibrinolysin (plasmin) and a potent activator of profibrinolysin (plasminogen). The activator, which we have designated urokinase, can be concentrated and partially purified by acetone or alcohol fractionation methods.
- PMID 13808740.
Further reading
- Ploug M, Gårdsvoll H, Jørgensen TJ, Lønborg Hansen L, Danø K (April 2002). "Structural analysis of the interaction between urokinase-type plasminogen activator and its receptor: a potential target for anti-invasive cancer therapy". Biochemical Society Transactions. 30 (2): 177–183. PMID 12023847.
- Alfano M, Sidenius N, Blasi F, Poli G (November 2003). "The role of urokinase-type plasminogen activator (uPA)/uPA receptor in HIV-1 infection". Journal of Leukocyte Biology. 74 (5): 750–756. S2CID 8526093.
- Harbeck N, Kates RE, Gauger K, Willems A, Kiechle M, Magdolen V, Schmitt M (March 2004). "Urokinase-type plasminogen activator (uPA) and its inhibitor PAI-I: novel tumor-derived factors with a high prognostic and predictive impact in breast cancer". Thrombosis and Haemostasis. 91 (3): 450–456. S2CID 19904733.
- Gilabert-Estelles J, Ramon LA, España F, Gilabert J, Castello R, Estelles A (2006). "Expression of the fibrinolytic components in endometriosis". Pathophysiology of Haemostasis and Thrombosis. 35 (1–2): 136–140. S2CID 29270171.