Ventricular fibrillation
| Ventricular fibrillation | |
|---|---|
Electrocardiogram[1] | |
| Differential diagnosis | Torsades de pointes[1] |
| Treatment | Cardiopulmonary resuscitation (CPR) with defibrillation[3] |
| Prognosis | Survival rate 17% (out of hospital), 46% (in hospital)[4][5][1] |
| Frequency | ~10% of people in cardiac arrest[1] |
Ventricular fibrillation (V-fib or VF) is an
Ventricular fibrillation can occur due to
Treatment is with
Signs and symptoms
Ventricular fibrillation is a cause of
It has an appearance on
Causes
Ventricular fibrillation has been described as "chaotic asynchronous fractionated activity of the heart" (Moe et al. 1964). A more complete definition is that ventricular fibrillation is a "turbulent, disorganized electrical activity of the heart in such a way that the recorded
Ventricular fibrillation most commonly occurs within diseased hearts, and, in the vast majority of cases, is a manifestation of underlying ischemic heart disease. Ventricular fibrillation is also seen in those with cardiomyopathy, myocarditis, and other heart pathologies. In addition, it is seen with electrolyte imbalance, overdoses of cardiotoxic drugs, and following near drowning or major trauma.[8] It is also notable that ventricular fibrillation occurs where there is no discernible heart pathology or other evident cause, the so-called idiopathic ventricular fibrillation.[citation needed]
Idiopathic ventricular fibrillation occurs with a reputed incidence of approximately 1% of all cases of out-of-hospital arrest, as well as 3–9% of the cases of ventricular fibrillation unrelated to
The relevance of this is that theories of the underlying pathophysiology and electrophysiology must account for the occurrence of fibrillation in the apparent "healthy" heart. It is evident that there are mechanisms at work that we do not fully appreciate and understand. Investigators are exploring new techniques of detecting and understanding the underlying mechanisms of sudden cardiac death in these patients without pathological evidence of underlying heart disease.[11]
Familial conditions that predispose individuals to developing ventricular fibrillation and sudden cardiac death are often the result of gene mutations that affect cellular transmembrane ion channels. For example, in Brugada Syndrome, sodium channels are affected. In certain forms of long QT syndrome, the potassium inward rectifier channel is affected.[citation needed]
In 1899, it was also found that ventricular fibrillation was, typically, the ultimate cause of death when the electric chair was used.[12]
Pathophysiology

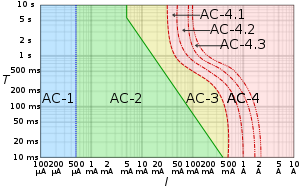
AC-1: imperceptible
AC-2: perceptible but no muscle reaction
AC-3: muscle contraction with reversible effects
AC-4: possible irreversible effects
AC-4.1: up to 5% probability of ventricular fibrillation
AC-4.2: 5-50% probability of fibrillation
AC-4.3: over 50% probability of fibrillation
Abnormal automaticity
Automaticity is a measure of the propensity of a fiber to initiate an impulse spontaneously. The product of a
Most myocardial cells with an associated increased propensity to arrhythmia development have an associated loss of membrane potential. That is, the maximum diastolic potential is less negative and therefore exists closer to the threshold potential. Cellular depolarisation can be due to a raised external concentration of potassium ions K+, a decreased intracellular concentration of sodium ions Na+, increased permeability to Na+, or a decreased permeability to K+. The ionic basic automaticity is the net gain of an intracellular positive charge during diastole in the presence of a voltage-dependent channel activated by potentials negative to –50 to –60 mV.[citation needed]
Myocardial cells are exposed to different environments. Normal cells may be exposed to hyperkalaemia; abnormal cells may be perfused by normal environment. For example, with a healed myocardial infarction, abnormal cells can be exposed to an abnormal environment such as with a myocardial infarction with myocardial ischaemia. In conditions such as myocardial ischaemia, possible mechanism of arrhythmia generation include the resulting decreased internal K+ concentration, the increased external K+ concentration, norepinephrine release and acidosis.
Re-entry
The role of re-entry or circus motion was demonstrated separately by
It is possible to think of the advancing wave of depolarisation as a dipole with a head and a tail. The length of the refractory period and the time taken for the dipole to travel a certain distance—the propagation velocity—will determine whether such a circumstance will arise for re-entry to occur. Factors that promote re-entry would include a slow-propagation velocity, a short refractory period with a sufficient size of ring of conduction tissue. These would enable a dipole to reach an area that had been refractory and is now able to be depolarised with continuation of the wavefront.[citation needed]
In clinical practice, therefore, factors that would lead to the right conditions to favour such re-entry mechanisms include increased heart size through hypertrophy or dilatation, drugs which alter the length of the refractory period and areas of cardiac disease. Therefore, the substrate of ventricular fibrillation is transient or permanent conduction block. Block due either to areas of damaged or refractory tissue leads to areas of myocardium for initiation and perpetuation of fibrillation through the phenomenon of re-entry.[citation needed]
Triggered activity
Triggered activity can occur due to the presence of afterdepolarisations. These are depolarising oscillations in the membrane voltage induced by preceding action potentials. These can occur before or after full repolarisation of the fiber and as such are termed either early (EADs) or delayed afterdepolarisations (DADs). All afterdepolarisations may not reach threshold potential, but, if they do, they can trigger another afterdepolarisation, and thus self-perpetuate.[citation needed]
Power spectrum

The distribution of frequency and power of a waveform can be expressed as a power spectrum in which the contribution of different waveform frequencies to the waveform under analysis is measured. This can be expressed as either the dominant or peak frequency, i.e., the frequency with the greatest power or the median frequency, which divides the spectrum in two halves.[citation needed]
Frequency analysis has many other uses in medicine and in cardiology, including analysis of heart rate variability and assessment of cardiac function, as well as in imaging and acoustics.[17][18]
Histopathology

Myofibre break-up, abbreviated MFB, is associated with ventricular fibrillation leading to death.
Treatment
The precordial thump is a manoeuver promoted as a mechanical alternative to defibrillation. Some advanced life support algorithms advocate its use once and only in the case of witnessed and monitored V-fib arrests as the likelihood of it successfully cardioverting a patient are small and this diminishes quickly in the first minute of onset.[citation needed]
People who survive a "V-fib arrest" and who make a good recovery are often considered for an implantable cardioverter-defibrillator, which can quickly deliver this same life-saving defibrillation should another episode of ventricular fibrillation occur outside a hospital environment.[citation needed]
Epidemiology
Sudden cardiac arrest is the leading cause of death in the industrialised world. It exacts a significant mortality with approximately 70,000 to 90,000 sudden cardiac deaths each year in the United Kingdom, and survival rates are only 2%.[20] The majority of these deaths are due to ventricular fibrillation secondary to myocardial infarction, or "heart attack".[21] During ventricular fibrillation, cardiac output drops to zero, and, unless remedied promptly, death usually ensues within minutes.[citation needed]
History
Lyman Brewer suggests that the first recorded account of ventricular fibrillation dates as far back as 1500 BC, and can be found in the
Whether this is a description of ventricular fibrillation is debatable.
The significance and clinical importance of these observations and descriptions possibly of ventricular fibrillation were not recognised until John Erichsen in 1842 described ventricular fibrillation following the
In 1874,
The first
Later milestones include the work by W. J. Kerr and W. L. Bender in 1922, who produced an electrocardiogram showing ventricular tachycardia evolving into ventricular fibrillation.[28] The re-entry mechanism was also advocated by DeBoer, who showed that ventricular fibrillation could be induced in late systole with a single shock to a frog heart.[29] The concept of "R on T ectopics" was further brought out by Katz in 1928.[30] This was called the "vulnerable period" by Wiggers and Wegria in 1940, who brought to attention the concept of the danger of premature ventricular beats occurring on a T wave.[citation needed]
Another definition of VF was produced by Wiggers in 1940. He described ventricular fibrillation as "an incoordinate type of contraction which, despite a high
Spontaneous conversion of ventricular fibrillation to a more benign rhythm is rare in all but small animals.[
See also
- Atrial fibrillation
- Electric shock
- Flatline
- HMR 1883
- Osborn wave
- Re-entry ventricular arrhythmia
- Ventricular flutter
References
- ^ PMID 27484660.
- ^ a b c d e "Types of Arrhythmia". NHLBI. July 1, 2011. Archived from the original on 7 June 2015. Retrieved 7 September 2016.
- ^ PMID 26472989.
In the appendix
- ^ PMID 20828914.
- PMID 24258072.
- ISBN 9780781787635. Archivedfrom the original on 2017-08-08.
- PMID 699945.
- PMID 7710153.
- PMID 2202193.
- PMID 1309182.
- PMID 7586358. Archived from the originalon 2012-07-08. Retrieved 2008-07-21.
- ^ Juan, Stephen (20 October 2006). "What happens when you are executed by electrocution?". The Register. Retrieved 2019-02-01.
- ^ a b "UOTW #37 - Ultrasound of the Week". Ultrasound of the Week. 11 February 2015. Archived from the original on 9 May 2017. Retrieved 27 May 2017.
- ^ Weineng Wang, Zhiqiang Wang, Xiao Peng, Effects of the Earth Current Frequency and Distortion on Residual Current Devices Archived 2014-11-08 at the Wayback Machine, Scientific Journal of Control Engineering, Dec 2013, Vol 3 Issue 6 pp 417-422
- ^ Ho K 1993
- ^ Mines GR 1913, Garrey WE 1914
- PMID 10688324.
- PMID 10689248.
- PMID 15837088.
- National Institute for Health and Clinical ExcellenceGuidelines 2000
- ^ Myerburg RJ et al. 1995
- PMID 6344674.
- PMID 6344674.
- ^ Hoffa M et al. 1850
- ^ Vulpian A 1874
- ^ MacWilliam JA 1887
- ^ Hoffman A 1912
- ^ Kerr, WJ et al. 1922
- ^ De Boer S 1923
- ^ Katz LN 1928
- ^ Wiggers, CJ et al. 1940
External links
- Interactive models and information on ventricular fibrillation and other arrhythmias
