Vinylogy
pKa
of such vinylogs, which approach that of the analogous carboxylic acid.In
acidic properties of formylacetone and related ketoaldehydes. Formylacetone, technically CH3(C=O)CH2CH=O, only exists in the ionized form CH3(C−O−)=CH−CH=O or CH3(C=O)−CH=CH−O−.[2] Its adjectival form, vinylogous, is used to describe functional groups in which the standard moieties of the group are separated by a carbon–carbon double bond
.
For example, a
3-dimethylaminoacrolein is the vinylogous-amide analog of dimethylformamide
.
Due to the transmission of electronic information through conjugation, vinylogous functional groups often possess "analogous"
enols (pKa ~ 12). Vitamin C (ascorbic acid
, see below) is a biologically important example of a vinylogous carboxylic acid.
The insertion of a o- or p-
phenylene (i.e., a benzene ring in the 1,2- or 1,4-orientation) also results in some similarities in reactivity (called "phenylogy"), although the effect is generally weaker, as conjugation through the aryl ring requires consideration of resonance forms or intermediates in which aromaticity is disrupted.[3][4]
Vinylogous reactions are believed to occur when
conjugated). Electron delocalization
enables the EWG to receive electron density through participation of the conjugated system.
Vinylogous reactivity
A classic example of vinylogy is the relatively high acidity of the γ-hydrogen in CH3CH=CHC(O)R. The acidity of the terminal methyl group is similar to that for the methyl ketone CH3C(O)R.[5]
Vinylogous reactions also include
Allylic
electrophiles often react by vinylogous attack of a nucleophile rather than direct addition.
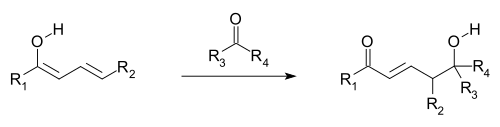
A further example of vinylogous reactivity:
conjugate base of ascorbic acid (center and right structures in last image), analogous to the two resonance structures that stabilize the negative charge on the anion that results from removal of a proton from a simple carboxylic acid (cf. first image). Analogously, sorbic acid
derivatives, extended by another "vinyl" moiety show vinylogous behaviour as well.
Electron pushing
for major resonance structures in conjugate base of ascorbic acidFurther reading
- Lisboa, Marilda P.; Hoang, Tung T.; Dudley, Gregory B. (2011). "Tandem Nucleophilic Addition / Fragmentation of Vinylogous Acyl Triflates: 2-Methyl-2-(1-Oxo-5-Heptynyl)-1,3-Dithiane". Organic Syntheses. 88: 353. .
References
- ISSN 0040-4020.
- PMID 20055373.
- ISBN 978-0-471-72091-1
