Virus
| Virus | |
|---|---|
| |
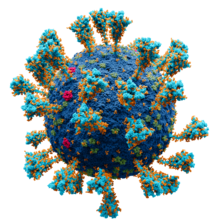
Coronavirinae
| |
| Virus classification | |
| (unranked): | Virus |
| Realms | |
A virus is a submicroscopic
When infected, a
The origins of viruses in the
Viral infections in animals provoke an
Etymology
The English word "virus" comes from the
Origins
Viruses are found wherever there is life and have probably existed since living cells first evolved.
There are three main hypotheses that aim to explain the origins of viruses:[25]
- Regressive hypothesis
- Viruses may have once been small cells that parasitised larger cells. Over time, genes not required by their parasitism were lost. The bacteria rickettsia and chlamydia are living cells that, like viruses, can reproduce only inside host cells. They lend support to this hypothesis, as their dependence on parasitism is likely to have caused the loss of genes that enabled them to survive outside a cell. This is also called the "degeneracy hypothesis",[26][27] or "reduction hypothesis".[28]
- Cellular origin hypothesis
- Some viruses may have evolved from bits of DNA or RNA that "escaped" from the genes of a larger organism. The escaped DNA could have come from transposons (molecules of DNA that replicate and move around to different positions within the genes of the cell).[29] Once called "jumping genes", transposons are examples of mobile genetic elements and could be the origin of some viruses. They were discovered in maize by Barbara McClintock in 1950.[30] This is sometimes called the "vagrancy hypothesis",[26][31] or the "escape hypothesis".[28]
- Co-evolution hypothesis
- This is also called the "virus-first hypothesis"hepatitis delta virus of humans has an RNA genome similar to viroids but has a protein coat derived from hepatitis B virus and cannot produce one of its own. It is, therefore, a defective virus. Although hepatitis delta virus genome may replicate independently once inside a host cell, it requires the help of hepatitis B virus to provide a protein coat so that it can be transmitted to new cells.[35] In similar manner, the sputnik virophage is dependent on mimivirus, which infects the protozoan Acanthamoeba castellanii.[36] These viruses, which are dependent on the presence of other virus species in the host cell, are called "satellites" and may represent evolutionary intermediates of viroids and viruses.[37][38]
In the past, there were problems with all of these hypotheses: the regressive hypothesis did not explain why even the smallest of cellular parasites do not resemble viruses in any way. The escape hypothesis did not explain the complex capsids and other structures on virus particles. The virus-first hypothesis contravened the definition of viruses in that they require host cells.[28] Viruses are now recognised as ancient and as having origins that pre-date the divergence of life into the three domains.[39] This discovery has led modern virologists to reconsider and re-evaluate these three classical hypotheses.[39]
The evidence for an ancestral world of RNA cells[40] and computer analysis of viral and host DNA sequences give a better understanding of the evolutionary relationships between different viruses and may help identify the ancestors of modern viruses. To date, such analyses have not proved which of these hypotheses is correct.[40] It seems unlikely that all currently known viruses have a common ancestor, and viruses have probably arisen numerous times in the past by one or more mechanisms.[41]
Microbiology
Life properties
Scientific opinions differ on whether viruses are a form of life or organic structures that interact with living organisms.
Structure
Viruses display a wide diversity of sizes and shapes, called '
A complete virus particle, known as a virion, consists of nucleic acid surrounded by a protective coat of protein called a capsid. These are formed from protein subunits called capsomeres.[50] Viruses can have a lipid "envelope" derived from the host cell membrane. The capsid is made from proteins encoded by the viral genome and its shape serves as the basis for morphological distinction.[51][52] Virally-coded protein subunits will self-assemble to form a capsid, in general requiring the presence of the virus genome. Complex viruses code for proteins that assist in the construction of their capsid. Proteins associated with nucleic acid are known as nucleoproteins, and the association of viral capsid proteins with viral nucleic acid is called a nucleocapsid. The capsid and entire virus structure can be mechanically (physically) probed through atomic force microscopy.[53][54] In general, there are five main morphological virus types:
- Helical
- These viruses are composed of a single type of capsomere stacked around a central axis to form a helical structure, which may have a central cavity, or tube. This arrangement results in virions which can be short and highly rigid rods, or long and very flexible filaments. The genetic material (typically single-stranded RNA, but single-stranded DNA in some cases) is bound into the protein helix by interactions between the negatively charged nucleic acid and positive charges on the protein. Overall, the length of a helical capsid is related to the length of the nucleic acid contained within it, and the diameter is dependent on the size and arrangement of capsomeres. The well-studied tobacco mosaic virus[55] and inovirus[56] are examples of helical viruses.
- Icosahedral
- Most animal viruses are icosahedral or near-spherical with chiral icosahedral symmetry. A regular icosahedron is the optimum way of forming a closed shell from identical subunits. The minimum number of capsomeres required for each triangular face is 3, which gives 60 for the icosahedron. Many viruses, such as rotavirus, have more than 60 capsomers and appear spherical but they retain this symmetry. To achieve this, the capsomeres at the apices are surrounded by five other capsomeres and are called pentons. Capsomeres on the triangular faces are surrounded by six others and are called hexons.[57] Hexons are in essence flat and pentons, which form the 12 vertices, are curved. The same protein may act as the subunit of both the pentamers and hexamers or they may be composed of different proteins.[58]
- Prolate
- This is an icosahedron elongated along the fivefold axis and is a common arrangement of the heads of bacteriophages. This structure is composed of a cylinder with a cap at either end.[59]
- Enveloped
- Some species of virus
- Complex
- These viruses possess a capsid that is neither purely helical nor purely icosahedral, and that may possess extra structures such as protein tails or a complex outer wall. Some bacteriophages, such as Enterobacteria phage T4, have a complex structure consisting of an icosahedral head bound to a helical tail, which may have a hexagonal base plate with protruding protein tail fibres. This tail structure acts like a molecular syringe, attaching to the bacterial host and then injecting the viral genome into the cell.[62]
The poxviruses are large, complex viruses that have an unusual morphology. The viral genome is associated with proteins within a central disc structure known as a nucleoid. The nucleoid is surrounded by a membrane and two lateral bodies of unknown function. The virus has an outer envelope with a thick layer of protein studded over its surface. The whole virion is slightly pleomorphic, ranging from ovoid to brick-shaped.[63]
Giant viruses
Mimivirus is one of the largest characterised viruses, with a capsid diameter of 400 nm. Protein filaments measuring 100 nm project from the surface. The capsid appears hexagonal under an electron microscope, therefore the capsid is probably icosahedral.[64] In 2011, researchers discovered the largest then known virus in samples of water collected from the ocean floor off the coast of Las Cruces, Chile. Provisionally named Megavirus chilensis, it can be seen with a basic optical microscope.[65] In 2013, the Pandoravirus genus was discovered in Chile and Australia, and has genomes about twice as large as Megavirus and Mimivirus.[66] All giant viruses have dsDNA genomes and they are classified into several families: Mimiviridae, Pithoviridae, Pandoraviridae, Phycodnaviridae, and the Mollivirus genus.[67]
Some viruses that infect Archaea have complex structures unrelated to any other form of virus, with a wide variety of unusual shapes, ranging from spindle-shaped structures to viruses that resemble hooked rods, teardrops or even bottles. Other archaeal viruses resemble the tailed bacteriophages, and can have multiple tail structures.[68]
Genome
| Property | Parameters |
|---|---|
| Nucleic acid |
|
| Shape |
|
| Strandedness |
|
| Sense |
|
An enormous variety of genomic structures can be seen among
A virus has either a DNA or an RNA genome and is called a DNA virus or an RNA virus, respectively. The vast majority of viruses have RNA genomes. Plant viruses tend to have single-stranded RNA genomes and bacteriophages tend to have double-stranded DNA genomes.[73]
Viral genomes are circular, as in the
A viral genome, irrespective of nucleic acid type, is almost always either single-stranded (ss) or double-stranded (ds). Single-stranded genomes consist of an unpaired nucleic acid, analogous to one-half of a ladder split down the middle. Double-stranded genomes consist of two complementary paired nucleic acids, analogous to a ladder. The virus particles of some virus families, such as those belonging to the Hepadnaviridae, contain a genome that is partially double-stranded and partially single-stranded.[73]
For most viruses with RNA genomes and some with single-stranded DNA (ssDNA) genomes, the single strands are said to be either
Genome size
Genome size varies greatly between species. The smallest—the ssDNA circoviruses, family Circoviridae—code for only two proteins and have a genome size of only two kilobases;[75] the largest—the pandoraviruses—have genome sizes of around two megabases which code for about 2500 proteins.[66] Virus genes rarely have introns and often are arranged in the genome so that they overlap.[76]
In general, RNA viruses have smaller genome sizes than DNA viruses because of a higher error-rate when replicating, and have a maximum upper size limit.[24] Beyond this, errors when replicating render the virus useless or uncompetitive. To compensate, RNA viruses often have segmented genomes—the genome is split into smaller molecules—thus reducing the chance that an error in a single-component genome will incapacitate the entire genome. In contrast, DNA viruses generally have larger genomes because of the high fidelity of their replication enzymes.[77] Single-strand DNA viruses are an exception to this rule, as mutation rates for these genomes can approach the extreme of the ssRNA virus case.[78]
Genetic mutation and recombination
Viruses undergo genetic change by several mechanisms. These include a process called
Segmented genomes confer evolutionary advantages; different strains of a virus with a segmented genome can shuffle and combine genes and produce progeny viruses (or offspring) that have unique characteristics. This is called reassortment or 'viral sex'.[83]
Genetic recombination is a process by which a strand of DNA (or RNA) is broken and then joined to the end of a different DNA (or RNA) molecule. This can occur when viruses infect cells simultaneously and studies of viral evolution have shown that recombination has been rampant in the species studied.[84] Recombination is common to both RNA and DNA viruses.[85][86]
Coronaviruses have a single-strand positive-sense RNA genome. Replication of the genome is catalyzed by an RNA-dependent RNA polymerase. The mechanism of recombination used by coronaviruses likely involves template switching by the polymerase during genome replication.[87] This process appears to be an adaptation for coping with genome damage.[88]
Replication cycle
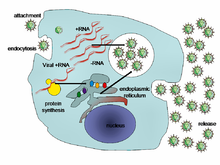

Viral populations do not grow through cell division, because they are acellular. Instead, they use the machinery and metabolism of a host cell to produce multiple copies of themselves, and they assemble in the cell.[89] When infected, the host cell is forced to rapidly produce thousands of copies of the original virus.[90]
Their life cycle differs greatly between species, but there are six basic stages in their life cycle:[91]
Attachment is a specific binding between viral capsid proteins and specific receptors on the host cellular surface. This specificity determines the host range and type of host cell of a virus. For example, HIV infects a limited range of human
Penetration or viral entry follows attachment: Virions enter the host cell through receptor-mediated endocytosis or membrane fusion. The infection of plant and fungal cells is different from that of animal cells. Plants have a rigid cell wall made of cellulose, and fungi one of chitin, so most viruses can get inside these cells only after trauma to the cell wall.[93] Nearly all plant viruses (such as tobacco mosaic virus) can also move directly from cell to cell, in the form of single-stranded nucleoprotein complexes, through pores called plasmodesmata.[94] Bacteria, like plants, have strong cell walls that a virus must breach to infect the cell. Given that bacterial cell walls are much thinner than plant cell walls due to their much smaller size, some viruses have evolved mechanisms that inject their genome into the bacterial cell across the cell wall, while the viral capsid remains outside.[95]
Uncoating is a process in which the viral capsid is removed: This may be by degradation by viral enzymes or host enzymes or by simple dissociation; the end-result is the releasing of the viral genomic nucleic acid.[96]
Replication of viruses involves primarily multiplication of the genome. Replication involves the synthesis of viral messenger RNA (mRNA) from "early" genes (with exceptions for positive-sense RNA viruses), viral protein synthesis, possible assembly of viral proteins, then viral genome replication mediated by early or regulatory protein expression. This may be followed, for complex viruses with larger genomes, by one or more further rounds of mRNA synthesis: "late" gene expression is, in general, of structural or virion proteins.[97]
Assembly – Following the structure-mediated self-assembly of the virus particles, some modification of the proteins often occurs. In viruses such as HIV, this modification (sometimes called maturation) occurs after the virus has been released from the host cell.[98]
Release – Viruses can be released from the host cell by lysis, a process that kills the cell by bursting its membrane and cell wall if present: this is a feature of many bacterial and some animal viruses. Some viruses undergo a lysogenic cycle where the viral genome is incorporated by genetic recombination into a specific place in the host's chromosome. The viral genome is then known as a "provirus" or, in the case of bacteriophages a "prophage".[99] Whenever the host divides, the viral genome is also replicated. The viral genome is mostly silent within the host. At some point, the provirus or prophage may give rise to the active virus, which may lyse the host cells.[100] Enveloped viruses (e.g., HIV) typically are released from the host cell by budding. During this process, the virus acquires its envelope, which is a modified piece of the host's plasma or other, internal membrane.[101]
Genome replication
The genetic material within virus particles, and the method by which the material is replicated, varies considerably between different types of viruses.
- DNA viruses
- The genome replication of most DNA viruses takes place in the cell's nucleus. If the cell has the appropriate receptor on its surface, these viruses enter the cell either by direct fusion with the cell membrane (e.g., herpesviruses) or—more usually—by receptor-mediated endocytosis. Most DNA viruses are entirely dependent on the host cell's DNA and RNA synthesising machinery and RNA processing machinery. Viruses with larger genomes may encode much of this machinery themselves. In eukaryotes, the viral genome must cross the cell's nuclear membrane to access this machinery, while in bacteria it need only enter the cell.[102]
- RNA viruses
- Replication of RNA replicase enzymes to create copies of their genomes.[103]
- Reverse transcribing viruses
- reverse transcription into the host genome as a provirus as a part of the replication process; pararetroviruses do not, although integrated genome copies of especially plant pararetroviruses can give rise to infectious virus.[104] They are susceptible to antiviral drugs that inhibit the reverse transcriptase enzyme, e.g. zidovudine and lamivudine. An example of the first type is HIV, which is a retrovirus. Examples of the second type are the Hepadnaviridae, which includes Hepatitis B virus.[105]
Cytopathic effects on the host cell
The range of structural and biochemical effects that viruses have on the host cell is extensive.
Dormant and latent infections
Some viruses cause no apparent changes to the infected cell. Cells in which the virus is
Host range
Viruses are by far the most abundant biological entities on Earth and they outnumber all the others put together.
Novel viruses
A novel virus is one that has not previously been recorded. It can be a virus that is isolated from its natural reservoir or isolated as the result of spread to an animal or human host where the virus had not been identified before. It can be an emergent virus, one that represents a new virus, but it can also be an extant virus that has not been previously identified.[121] The SARS-CoV-2 coronavirus that caused the COVID-19 pandemic is an example of a novel virus.[122]
Classification
Classification seeks to describe the diversity of viruses by naming and grouping them on the basis of similarities. In 1962,
ICTV classification
The ICTV developed the current classification system and wrote guidelines that put a greater weight on certain virus properties to maintain family uniformity. A unified taxonomy (a universal system for classifying viruses) has been established.[129] Only a small part of the total diversity of viruses has been studied.[130] As of 2022, 6 realms, 10 kingdoms, 17 phyla, 2 subphyla, 40 classes, 72 orders, 8 suborders, 264 families, 182 subfamilies, 2,818 genera, 84 subgenera, and 11,273 species of viruses have been defined by the ICTV.[7]
The general taxonomic structure of taxon ranges and the suffixes used in taxonomic names are shown hereafter. As of 2022, the ranks of subrealm, subkingdom, and subclass are unused, whereas all other ranks are in use.[7]
- Realm (-viria)
Baltimore classification
The Nobel Prize-winning biologist David Baltimore devised the Baltimore classification system.[131][132] The ICTV classification system is used in conjunction with the Baltimore classification system in modern virus classification.[133][134][135]
The Baltimore classification of viruses is based on the mechanism of
- I: Poxviruses)
- II: Parvoviruses)
- III: Reoviruses)
- IV:Togaviruses)
- V: Rhabdoviruses)
- VI: ssRNA-RT viruses (+ strand or sense) RNA with DNA intermediate in life-cycle (e.g. Retroviruses)
- VII: Hepadnaviruses)
Role in human disease

Examples of common human diseases caused by viruses include the
Viruses have different mechanisms by which they produce disease in an organism, which depends largely on the viral species. Mechanisms at the cellular level primarily include cell lysis, the breaking open and subsequent death of the cell. In multicellular organisms, if enough cells die, the whole organism will start to suffer the effects. Although viruses cause disruption of healthy homeostasis, resulting in disease, they may exist relatively harmlessly within an organism. An example would include the ability of the herpes simplex virus, which causes cold sores, to remain in a dormant state within the human body. This is called latency[139] and is a characteristic of the herpes viruses, including Epstein–Barr virus, which causes glandular fever, and varicella zoster virus, which causes chickenpox and shingles. Most people have been infected with at least one of these types of herpes virus.[140] These latent viruses might sometimes be beneficial, as the presence of the virus can increase immunity against bacterial pathogens, such as Yersinia pestis.[141]
Some viruses can cause lifelong or
Epidemiology
Viral epidemiology is the branch of medical science that deals with the transmission and control of virus infections in humans. Transmission of viruses can be vertical, which means from mother to child, or horizontal, which means from person to person. Examples of vertical transmission include hepatitis B virus and HIV, where the baby is born already infected with the virus.[145] Another, more rare, example is the varicella zoster virus, which, although causing relatively mild infections in children and adults, can be fatal to the foetus and newborn baby.[146]
Epidemiology is used to break the chain of infection in populations during outbreaks of viral diseases.[150] Control measures are used that are based on knowledge of how the virus is transmitted. It is important to find the source, or sources, of the outbreak and to identify the virus. Once the virus has been identified, the chain of transmission can sometimes be broken by vaccines. When vaccines are not available, sanitation and disinfection can be effective. Often, infected people are isolated from the rest of the community, and those that have been exposed to the virus are placed in quarantine.[151] To control the outbreak of foot-and-mouth disease in cattle in Britain in 2001, thousands of cattle were slaughtered.[152] Most viral infections of humans and other animals have incubation periods during which the infection causes no signs or symptoms.[153] Incubation periods for viral diseases range from a few days to weeks, but are known for most infections.[154] Somewhat overlapping, but mainly following the incubation period, there is a period of communicability—a time when an infected individual or animal is contagious and can infect another person or animal.[154] This, too, is known for many viral infections, and knowledge of the length of both periods is important in the control of outbreaks.[155] When outbreaks cause an unusually high proportion of cases in a population, community, or region, they are called epidemics. If outbreaks spread worldwide, they are called pandemics.[156]
Epidemics and pandemics
A
Although viral pandemics are rare events, HIV—which evolved from viruses found in monkeys and chimpanzees—has been pandemic since at least the 1980s.[160] During the 20th century there were four pandemics caused by influenza virus and those that occurred in 1918, 1957 and 1968 were severe.[161] Most researchers believe that HIV originated in sub-Saharan Africa during the 20th century;[162] it is now a pandemic, with an estimated 37.9 million people now living with the disease worldwide.[163] There were about 770,000 deaths from AIDS in 2018.[164] The Joint United Nations Programme on HIV/AIDS (UNAIDS) and the World Health Organization (WHO) estimate that AIDS has killed more than 25 million people since it was first recognised on 5 June 1981, making it one of the most destructive epidemics in recorded history.[165] In 2007 there were 2.7 million new HIV infections and 2 million HIV-related deaths.[166]
Several highly lethal viral pathogens are members of the
Except for smallpox, most pandemics are caused by newly evolved viruses. These "emergent" viruses are usually mutants of less harmful viruses that have circulated previously either in humans or other animals.[169]
Severe acute respiratory syndrome (
A related coronavirus,
Cancer
Viruses are an established cause of cancer in humans and other species. Viral cancers occur only in a minority of infected persons (or animals). Cancer viruses come from a range of virus families, including both RNA and DNA viruses, and so there is no single type of "
Host defence mechanisms
The body's first line of defence against viruses is the innate immune system. This comprises cells and other mechanisms that defend the host from infection in a non-specific manner. This means that the cells of the innate system recognise, and respond to, pathogens in a generic way, but, unlike the adaptive immune system, it does not confer long-lasting or protective immunity to the host.[185]
RNA interference is an important innate defence against viruses.[186] Many viruses have a replication strategy that involves double-stranded RNA (dsRNA). When such a virus infects a cell, it releases its RNA molecule or molecules, which immediately bind to a protein complex called a dicer that cuts the RNA into smaller pieces. A biochemical pathway—the RISC complex—is activated, which ensures cell survival by degrading the viral mRNA. Rotaviruses have evolved to avoid this defence mechanism by not uncoating fully inside the cell, and releasing newly produced mRNA through pores in the particle's inner capsid. Their genomic dsRNA remains protected inside the core of the virion.[187][188]
When the
Antibodies can continue to be an effective defence mechanism even after viruses have managed to gain entry to the host cell. A protein that is in cells, called
A second defence of vertebrates against viruses is called
Not all virus infections produce a protective immune response in this way. HIV evades the immune system by constantly changing the amino acid sequence of the proteins on the surface of the virion. This is known as "escape mutation" as the viral epitopes escape recognition by the host immune response. These persistent viruses evade immune control by sequestration, blockade of antigen presentation, cytokine resistance, evasion of natural killer cell activities, escape from apoptosis, and antigenic shift.[194] Other viruses, called 'neurotropic viruses', are disseminated by neural spread where the immune system may be unable to reach them due to immune privilege.[195]
Prevention and treatment
Because viruses use vital metabolic pathways within host cells to replicate, they are difficult to eliminate without using drugs that cause toxic effects to host cells in general. The most effective medical approaches to viral diseases are
Vaccines
Vaccination is a cheap and effective way of preventing infections by viruses. Vaccines were used to prevent viral infections long before the discovery of the actual viruses. Their use has resulted in a dramatic decline in morbidity (illness) and mortality (death) associated with viral infections such as
Antiviral drugs
Antiviral drugs are often
Hepatitis C is caused by an RNA virus. In 80% of people infected, the disease is chronic, and without treatment, they are infected for the remainder of their lives. There are effective treatments that use direct-acting antivirals.[210] The treatment of chronic carriers of the hepatitis B virus has also been developed by using similar strategies that include lamivudine and other anti-viral drugs.[211]
Infection in other species
Viruses infect all cellular life and, although viruses occur universally, each cellular species has its own specific range that often infects only that species.[212] Some viruses, called satellites, can replicate only within cells that have already been infected by another virus.[36]
Animal viruses
Viruses are important pathogens of livestock. Diseases such as foot-and-mouth disease and
Plant viruses

There are many types of plant viruses, but often they cause only a loss of
Originally from Peru, the potato has become a staple crop worldwide.[219] The potato virus Y causes disease in potatoes and related species including tomatoes and peppers. In the 1980s, this virus acquired economical importance when it proved difficult to control in seed potato crops. Transmitted by aphids, this virus can reduce crop yields by up to 80 per cent, causing significant losses to potato yields.[220]
Plants have elaborate and effective defence mechanisms against viruses. One of the most effective is the presence of so-called resistance (R) genes. Each R gene confers resistance to a particular virus by triggering localised areas of cell death around the infected cell, which can often be seen with the unaided eye as large spots. This stops the infection from spreading.[221] RNA interference is also an effective defence in plants.[222] When they are infected, plants often produce natural disinfectants that kill viruses, such as salicylic acid, nitric oxide, and reactive oxygen molecules.[223]
Plant virus particles or virus-like particles (VLPs) have applications in both biotechnology and nanotechnology. The capsids of most plant viruses are simple and robust structures and can be produced in large quantities either by the infection of plants or by expression in a variety of heterologous systems. Plant virus particles can be modified genetically and chemically to encapsulate foreign material and can be incorporated into supramolecular structures for use in biotechnology.[224]
Bacterial viruses

Bacteriophages are a common and diverse group of viruses and are the most abundant biological entity in aquatic environments—there are up to ten times more of these viruses in the oceans than there are bacteria,
The major way bacteria defend themselves from bacteriophages is by producing enzymes that destroy foreign DNA. These enzymes, called
Some bacteriophages are called "
Archaeal viruses
Some viruses replicate within
Role in aquatic ecosystems
Viruses are the most abundant biological entity in aquatic environments.
Microorganisms constitute more than 90% of the biomass in the sea. It is estimated that viruses kill approximately 20% of this biomass each day and that there are 10 to 15 times as many viruses in the oceans as there are bacteria and archaea.[246] Viruses are also major agents responsible for the destruction of phytoplankton including harmful algal blooms,[247] The number of viruses in the oceans decreases further offshore and deeper into the water, where there are fewer host organisms.[245]
In January 2018, scientists reported that 800 million viruses, mainly of marine origin, are deposited daily from the Earth's atmosphere onto every square meter of the planet's surface, as the result of a global atmospheric stream of viruses, circulating above the weather system but below the altitude of usual airline travel, distributing viruses around the planet.[248][249]
Like any organism,
In December 2022, scientists reported the first observation of
Role in evolution
Viruses are an important natural means of transferring genes between different species, which increases
Applications
Life sciences and medicine
Viruses are important to the study of
Geneticists often use viruses as
Virotherapy
Virotherapy involves the use of genetically modified viruses to treat diseases.
Materials science and nanotechnology
From the viewpoint of a materials scientist, viruses can be regarded as organic
Synthetic viruses
Many viruses can be synthesised de novo ("from scratch"). The first synthetic virus was created in 2002.
Weapons
The ability of viruses to cause devastating epidemics in human societies has led to the concern that viruses could be weaponised for biological warfare. Further concern was raised by the successful recreation of the infamous 1918 influenza virus in a laboratory.[270] The smallpox virus devastated numerous societies throughout history before its eradication. There are only two centres in the world authorised by the WHO to keep stocks of smallpox virus: the State Research Center of Virology and Biotechnology VECTOR in Russia and the Centers for Disease Control and Prevention in the United States.[271] It may be used as a weapon,[271] as the vaccine for smallpox sometimes had severe side-effects, it is no longer used routinely in any country. Thus, much of the modern human population has almost no established resistance to smallpox and would be vulnerable to the virus.[271]
See also
- Cross-species transmission
- Glossary of virology
- Law of declining virulence– Disproved hypothesis of epidemiologist Theobald Smith
- Non-cellular life
- Retrozyme
- Smallest organisms
- Theory of virulence– Theory by biologist Paul W. Ewald
- Viral metagenomics
- Viroplasm
- Zoonosis
References
Notes
- ^ Wu KJ (15 April 2020). "There are more viruses than stars in the universe. Why do only some infect us? – More than a quadrillion quadrillion individual viruses exist on Earth, but most are not poised to hop into humans. Can we find the ones that are?". National Geographic Society. Archived from the original on 15 April 2020. Retrieved 18 May 2020.
- ^ PMID 16984643.
- ^ Zimmer C (26 February 2021). "The Secret Life of a Coronavirus - An oily, 100-nanometer-wide bubble of genes has killed more than two million people and reshaped the world. Scientists don't quite know what to make of it". The New York Times. Archived from the original on 28 December 2021. Retrieved 28 February 2021.
- ^ PMID 19158076.
- S2CID 8059643.
- ^ a b Dimmock p. 4
- ^ a b c "Virus Taxonomy: 2022 Release". talk.ictvonline.org. International Committee on Taxonomy of Viruses. Retrieved 27 April 2023.
- ^ PMID 15936660.
- ^ PMID 12941415.
- ^ a b Rybicki EP (1990). "The classification of organisms at the edge of life, or problems with virus systematics". South African Journal of Science. 86: 182–86.
- ^ PMID 26965225.
- PMID 25567225.
- ^ Shors pp. 123–124
- ^ "Virus, n.". OED Online. Oxford University Press. March 2015.
- ^ a b Harper D (2011). "virus". The Online Etymology Dictionary. Retrieved 19 December 2014.
- ^ "Virulent, adj.". OED Online. Oxford University Press. March 2015.
- ^ Harper D (2011). "virulent". The Online Etymology Dictionary. Retrieved 19 December 2014.
- ^ Buschard K, Thon R (2003). "Diabetic Animal Models". In Hau J, Van Hoosier Jr GL (eds.). Handbook of Laboratory Animal Science. Animal Models. Vol. II (2nd ed.). CRC Press. pp. 163, 166.
- ^ William T. Stearn: Botanical Latin. History, Grammar, Syntax, Terminology and Vocabulary. David & Charles, 3rd ed., 1983. Quote: "Virus: virus (s.n. II), gen. sing. viri, nom. pl. vira, gen. pl. vīrorum (to be distinguished from virorum, of men)."
- ^ Harper D (2011). "viral". The Online Etymology Dictionary. Retrieved 19 December 2014.
- ^ Harper D (2011). "virion". The Online Etymology Dictionary. Retrieved 19 December 2014.
- ISBN 978-0-12-375146-1.
- PMID 16494962.
- ^ PMID 20660197.
- S2CID 256744818.
- ^ a b Dimmock p. 16
- ^ Collier p. 11
- ^ ISBN 978-0-12-375146-1.
- ^ Shors p. 810
- PMID 15430309.
- ^ Collier pp. 11–12
- ^ Dimmock p. 55
- ^ Shors pp. 791
- S2CID 221581424.
- ^ Shors pp. 460
- ^ S2CID 4422249.
- ^ Collier p. 777
- ^ Dimmock pp. 55–57
- ^ ISBN 978-0-12-375146-1.
- ^ ISBN 978-0-12-375146-1.
- ^ Dimmock pp. 15–16
- PMID 17914905.
- PMID 20010599.
- PMID 18473699.
- PMID 19016440.
- ^ Krasner p. 98
- ^ a b Collier pp. 33–55
- ^ Collier pp. 33–37
- PMID 1715774.
- ^ Collier p. 40
- PMID 14019094.
- S2CID 5740221.
- PMID 9138585.
- PMID 11514711.
- ^ Collier p. 37
- PMID 29900501.
- ^ Collier pp. 40, 42
- PMID 27045511.
- ISBN 978-0-12-375146-1.
- PMID 32580969.
- ^ Collier pp. 42–43
- PMID 15093831.
- PMID 4322005.
- PMID 16181700.
- PMID 21987820.
- ^ S2CID 16877147.
- PMID 31052218.
- ^ S2CID 9915859.
- ^ a b Dimmock p. 49
- ^ "NCBI Viral Genome database". ncbi.nlm.nih.gov. Retrieved 15 January 2017.
- PMID 21436418.
- S2CID 1198891.
- ^ a b c Collier pp. 96–99
- ISBN 978-0-470-02387-7.
- PMID 20861255.
- PMID 27209091.
- PMID 6433032.
- PMID 19264617.
- PMID 22143798.
- PMID 20375034.
- S2CID 17069567.
- PMID 17578210.
- ISBN 978-0-19-512496-5
- PMID 10573145.
- S2CID 26000112.
- S2CID 43110533.
- ^ Su S, Wong G, Shi W, Liu J, Lai ACK, Zhou J, Liu W, Bi Y, Gao GF. Epidemiology, Genetic Recombination, and Pathogenesis of Coronaviruses. Trends Microbiol. 2016 Jun;24(6):490-502. doi: 10.1016/j.tim.2016.03.003. Epub 2016 Mar 21. PMID 27012512; PMCID: PMC7125511
- ^ Barr JN, Fearns R. How RNA viruses maintain their genome integrity. J Gen Virol. 2010 Jun;91(Pt 6):1373-87. doi: 10.1099/vir.0.020818-0. Epub 2010 Mar 24. PMID 20335491
- PMID 26119571.
- PMID 29592895.
- ^ Collier pp. 75–91
- PMID 23737062.
- ^ Dimmock p. 70
- PMID 16172094.
- ^ Dimmock p. 71
- PMID 27174165.
- S2CID 11448687.
- PMID 11451488.
- ^ Shors pp. 836
- ^ Dimmock, Chapter 15, Mechanisms in virus latency, pp. 243–59
- ^ Dimmock 185–87
- ^ Shors p. 118; Collier p. 78
- ^ Collier p. 79
- PMID 16949329.
- ^ Collier pp. 88–89
- ^ Collier pp. 115–46
- ^ Collier p. 115
- PMID 10547702.
- PMID 18637511.
- PMID 18035323.
- S2CID 1399003.
- PMID 18164651.
- PMID 6326635.
- PMID 12076064.
- ISBN 978-0-19-957485-8.
- ^ Shors p. 643
- ^ Shors p. 631
- ^ Dimmock p. 272
- PMID 20122382. Retrieved 19 December 2014.
- PMID 27698618.
- PMID 22966141.
- ^ PMID 32143502.
- PMID 14467544.
- PMID 13931895.
- PMID 16105179.
- PMID 32341570.
- PMID 34226482.
- S2CID 5642060.
- PMID 32341570.
- PMID 17295196.
- PMID 4348509.
- PMID 4377923.
- PMID 15078590.
- S2CID 33422303.
- PMID 15183049.
- ISBN 978-0-7817-8215-9.
- PMID 17276367.
- S2CID 19830976.
- PMID 17686862.
- S2CID 9854903.
- S2CID 4425405.
- S2CID 13386004.
- PMID 17664817.
- S2CID 29885790.
- PMID 17825648.
- S2CID 7973561.
- ^ PMID 28289251.
- PMID 15627236.
- PMID 16544901.
- ^ Shors p. 264
- ^ Shors pp. 894
- PMID 19091686.
- ^ Shors p. 170
- ^ a b Shors pp. 170–72
- ^ Shors p. 272
- ^ Shors pp. 891
- ^ Collier pp. 409–15
- PMID 2021692.
- S2CID 22974230.
- PMID 29460740.
- PMID 30180422.
- S2CID 4432185.
- ^ "Fact Sheet" (PDF). UNAIDS.org. 2018. Retrieved 12 December 2019.
- ^ "UN AIDS DATA2019". UNAIDS.org. 2019. Retrieved 5 December 2019.
- PMID 16517997. Archived from the original(PDF) on 4 March 2016. Retrieved 19 December 2014.
- ^ "Status of the global HIV epidemic" (PDF). UNAIDS. 2008. Archived from the original (PDF) on 22 November 2015. Retrieved 19 December 2014.
- PMID 16775337.
- ^ "World Health Organisation report, 24 September 2014" (PDF).
- ^ "Virology Journal". Virology Journal.
- PMID 22094080.
- PMID 28475794.
- PMID 32093211.
- PMID 32109444.
- ^ Londoño E, Ortiz A (16 March 2020). "Coronavirus Travel Restrictions, Across the Globe". The New York Times.
- ^ "US takes more big pandemic response steps; Europe COVID-19 cases soar". CIDRAP. 15 March 2020.
- PMID 19467474.
- PMID 18812503.
- S2CID 36110778.
- S2CID 25399220.
- PMID 17672044.
- PMID 17726237.
- S2CID 20196938.
- PMID 17322915.
- S2CID 9482506.
- ISBN 0-8153-3218-1.
- PMID 17693253.
- PMID 15579070.
- PMID 15010218.
- PMID 8903801.
- PMID 9086126.
- PMID 21045130.
- PMID 17725500.
- PMID 11256746.
- PMID 15297608.
- PMID 25015488.
- PMID 17068034.
- PMID 16989262.
- PMID 16364754.
- PMID 17892169.
- PMID 16494719.
- PMID 33933145.
- PMID 1090805.
- PMID 3018891.
- S2CID 40821818.
- S2CID 19352868.
- ^ PMID 27281742.
- PMID 15592828.
- PMID 6355051.
- PMID 21430338.
- PMID 28319996.
- PMID 32102898.
- ^ Dimmock p. 3
- PMID 18035428.
- PMID 16316389.
- PMID 15579317.
- ISBN 978-0123611604. Retrieved 21 February 2022.
- ^ Shors p. 802
- ^ Shors pp. 799–807
- S2CID 33074838.
- PMID 31559020.
- PMID 11121079.
- ^ Shors pp. 809
- S2CID 27311732.
- ISBN 978-1-904455-75-2.
- PMID 10704475.
- S2CID 4271861.
- ^ Shors pp. 834–35
- PMID 8336674.
- S2CID 3888761.
- PMID 18703739.
- S2CID 42827598.
- ^ PMID 28550453.
- PMID 34966370.
- PMID 36839434.
- S2CID 20018642.
- S2CID 27481111.
- PMID 16545108.
- PMID 24909109.
- PMID 31749771.
- S2CID 53564176.
- ^ Weitz JS, Wilhelm SW (2013). "An ocean of viruses". The Scientist. 27 (7): 35–39.
- S2CID 4370363.
- JSTOR 1313569.
- .
- ^ S2CID 4658457.
- S2CID 52829633.
- S2CID 21017882.
- ^ Robbins J (13 April 2018). "Trillions Upon Trillions of Viruses Fall From the Sky Each Day". The New York Times. Retrieved 14 April 2018.
- PMID 29379178.
- .
- S2CID 255219850.
- ^ Irving M (28 December 2022). "First "virovore" discovered: An organism that eats viruses". New Atlas. Archived from the original on 29 December 2022. Retrieved 29 December 2022.
- PMID 31032941.
- PMID 11536914.
- ^ Collier p. 8
- ^ Lodish H, Berk A, Zipursky SL, Matsudaira P, Baltimore D, Darnell J (2000). "Viruses: Structure, Function, and Uses". Molecular Cell Biology (4th ed.). New York: W. H. Freeman.
- S2CID 8107934.
- ISBN 978-1-904455-75-2.
- PMID 25900073.
- S2CID 38864550.
- ^ "FDA approves Amgen's injected immunotherapy for melanoma". Reuters. 27 October 2015. Retrieved 24 January 2020.
- PMID 25989094.
- PMID 27446051.
- PMID 17348058.
- PMID 16608355.
- PMID 17193509.
- S2CID 5810309.
- PMID 18583614.
- ^ "NIH viral genome database". Ncbi.nlm.nih.gov. Retrieved 28 June 2021.
- PMID 30152475.
- ^ S2CID 33855724.
Bibliography
- Collier L, Balows A, Sussman M (1998). Mahy B, Collier LA (eds.). Topley and Wilson's Microbiology and Microbial Infections. Virology. Vol. 1 (9th ed.). ISBN 0-340-66316-2.
- Dimmock NJ, Easton AJ, Leppard K (2007). Introduction to Modern Virology (6th ed.). Blackwell Publishing. ISBN 978-1-4051-3645-7.
- Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B, Straus SE (2007). Fields Virology. Lippincott Williams & Wilkins. ISBN 978-0-7817-6060-7.
- Krasner R (2014). The microbial challenge: a public health perspective. Burlington, Mass: Jones & Bartlett Learning. OCLC 794228026.
- Shors T (2017). Understanding Viruses. Jones and Bartlett Publishers. ISBN 978-1-284-02592-7.