Viral vector
Viral vectors are tools commonly used by molecular
In addition to their use in molecular biology research, viral vectors are used for gene therapy and the development of vaccines. Vectors can either integrate into a cell's genome or transiently express a gene with non-integrative vectors.[2]: 50
Key properties of a viral vector
Viral Vectors are tailored to their specific applications but generally share a few key properties.[citation needed]
- Safety: Although viral vectors are occasionally created from virions.
- Low toxicity: The viral vector should have a minimal effect on the physiology of the cell it infects.
- Stability: Some viruses are genetically unstable and can rapidly rearrange their genomes. This is detrimental to predictability and reproducibility of the work conducted using a viral vector and is avoided in their design.
- Cell type specificity: Most viral vectors are engineered to infect as wide a range of cell types as possible. However, sometimes the opposite is preferred. The viral vector can be modified to target the virus to a specific kind of cell. Viruses modified in this manner are said to be pseudotyped.
- Identification: Viral vectors are often given certain genes that help identify which cells took up the viral genes. These genes are called markers. A common marker is resistance to a certain antibiotic. The cells can then be isolated easily, as those that have not taken up the viral vector genes do not have antibiotic resistance, and so cannot grow in a culture with the relevant antibiotic present.
Applications
Basic research
Viral vectors were originally developed as an alternative to
Protein coding genes can be expressed using viral vectors, commonly to study the function of the particular protein. Viral vectors, especially retroviruses, stably expressing marker genes such as GFP are widely used to permanently label cells to track them and their progeny, for example in xenotransplantation experiments, when cells infected in vitro are implanted into a host animal.[citation needed]
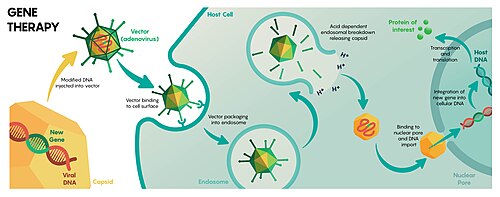
Gene therapy
Gene therapy is a technique for correcting defective genes responsible for disease development. In the future, gene therapy may provide a way to cure genetic disorders, such as severe combined immunodeficiency, cystic fibrosis or even haemophilia A. Because these diseases result from mutations in the DNA sequence for specific genes, gene therapy trials have used viruses to deliver unmutated copies of these genes to the cells of the patient's body. There have been a huge number of laboratory successes with gene therapy. However, several problems of viral gene therapy must be overcome before it gains widespread use. Immune response to viruses not only impedes the delivery of genes to target cells but can cause severe complications for the patient. In one of the early gene therapy trials in 1999 this led to the death of Jesse Gelsinger, who was treated using an adenoviral vector.[3]
Some viral vectors, for instance gamma-retroviruses, insert their genomes at a seemingly random location on one of the host chromosomes, which can disturb the function of cellular genes and lead to cancer. In a severe combined immunodeficiency retroviral gene therapy trial conducted in 2002, four of the patients developed leukemia as a consequence of the treatment;[4] three of the patients recovered after chemotherapy.[5] Adeno-associated virus-based vectors are much safer in this respect as they always integrate at the same site in the human genome, with applications in various disorders, such as Alzheimer's disease.[6]
Vaccines
A live vector vaccine is a
Unlike attenuated vaccines, viral vector vaccines lack other pathogen genes required for replication, so infection by the pathogen is impossible. Adenoviruses are being actively developed as vaccine vectors.
Medicine delivery
A strain of
Types
Retroviruses
Retroviral vectors can either be replication-competent or replication-defective. Replication-defective vectors are the most common choice in studies because the viruses have had the coding regions for the genes necessary for additional rounds of virion replication and packaging replaced with other genes, or deleted. These virus are capable of infecting their target cells and delivering their viral payload, but then fail to continue the typical
Conversely, replication-competent viral vectors contain all necessary genes for virion synthesis, and continue to propagate themselves once infection occurs. Because the viral genome for these vectors is much lengthier, the length of the actual inserted gene of interest is limited compared to the possible length of the insert for replication-defective vectors. Depending on the viral vector, the typical maximum length of an allowable DNA insert in a replication-defective viral vector is usually about 8–10 kB.[verification needed][10] While this limits the introduction of many genomic sequences, most cDNA sequences can still be accommodated.
The primary drawback to use of retroviruses such as the Moloney retrovirus involves the requirement for cells to be actively dividing for
There is concern that
Lentiviruses

For safety reasons, lentiviral vectors never carry the genes required for their replication. To produce a lentivirus, several
Adenoviruses
As opposed to lentiviruses, adenoviral DNA does not integrate into the genome and is not replicated during cell division.
Adeno-associated viruses

Adeno-associated virus (AAV) is a small virus that infects humans and some other primate species. AAV is not currently known to cause disease, and causes a very mild immune response. AAV can infect both dividing and non-dividing cells and may incorporate its genome into that of the host cell. Moreover, AAV mostly stays as episomal (replicating without incorporation into the chromosome); performing long and stable expression.[21] These features make AAV a very attractive candidate for creating viral vectors for gene therapy.[1] However, AAV can only bring up to 5kb which is considerably small compared to AAV's original capacity.[21]
Adeno-associated viral vectors have been engineered to evade virus recognition by
Furthermore, because of its potential use as a gene therapy vector, researchers have created an altered AAV called self-complementary adeno-associated virus (scAAV). Whereas AAV packages a single strand of DNA and requires the process of second-strand synthesis, scAAV packages both strands which anneal together to form double stranded DNA. By skipping second strand synthesis scAAV allows for rapid expression in the cell.[23] Otherwise, scAAV carries many characteristics of its AAV counterpart.
Plant viruses
Hybrids
Hybrid vectors are
Challenges in application
The choice of a
Pre-existing
Some shortcomings of viral vectors (such as genotoxicity and low transgenic expression) can be overcome through the use of
See also
References
- ^ S2CID 41788896.
- OCLC 1163431307.
- ^ Beardsley T (February 2000). "A tragic death clouds the future of an innovative treatment method". Scientific American.
- ^ McDowell N (15 January 2003). "New cancer case halts US gene therapy trials". New Scientist.
- PMID 20660403.
- S2CID 52978228.
- ^ "Live-Vector Vaccine". American Institute of Chemical Engineers. 17 December 2014. Archived from the original on 2021-02-03. Retrieved 2021-02-03.
- ^ "EPAR summary for the public: Oncept IL-2 (Feline interleukin-2 recombinant canary pox virus) [EMA/151380/2013 EMEA/V/C/002562]" (PDF). European Medical Agency. 2013.
- PMID 10784449.
- PMID 21433340.
- PMID 11961146.
- S2CID 9100335.
- S2CID 974969.
- S2CID 8715798.
- S2CID 8966580.
- PMID 30320151.
- OCLC 1163431307.
- PMID 33558455.
- PMID 26260012.
- S2CID 21927486.
- ^ ISBN 978-1-4377-0696-3.
- PMID 33568518.
- PMID 11509958.
- ^ S2CID 219588089.
- PMID 30677208.
- ^ magnICON
- PMID 23070017.
- ^ PMID 19907498.
- S2CID 17622444.
- ISBN 978-1420044379.
- PMID 12477888.
- PMID 22432020.
Further reading
- Torashima T, Koyama C, Higashida H, Hirai H (2007). "Production of neuron-preferential lentiviral vectors". Protocol Exchange. .
- Okada Y, Ikawa M (2007). "Placenta specific gene manipulation by transducing zona-free blastocyst using lentiviral vector". Protocol Exchange. .
- Fry JW, Wood KJ (8 June 1999). "A comparison of vectors in use for clinical gene transfer". Expert Reviews in Molecular Medicine.